1. Background
Obesity has a clear impact on many disorders, including cardiovascular diseases, osteoarthritis, and type-2 diabetes (1-3). An association based on the role of vitamin D in enhancing lipogenesis and inhibiting lipolysis has been reported in in-vitro studies (4). Obese people are more prone to vitamin D deficiency. This is probably due to the solubility of vitamin D in fat, making serum levels of this vitamin lower in obese people (5). 25-hydroxy vitamin D, a major form of circulating vitamin D, acts as a reservoir biologically for active vitamin D. Therefore, 25-hydroxy vitamin D is an indicator of the overall status of vitamin D (6). Physical activity is correlated with serum 25-hydroxy vitamin D levels (7). Although body weight as an interfering factor may not fully justify the association of physical activity with serum 25-hydroxy vitamin D levels, as observed after adjusting for fat percentage, the association of physical activity with serum 25-hydroxy vitamin D levels remains constant (7). One study reported that nine months of exercise (aerobic exercise, balance, and strength) with and without the use of Kulehkelsifrol elevated serum 25-hydroxy vitamin D levels in elderly people at the age of 70 years (8).
Measuring anthropometric characteristics can be described as a simple and practical way to predict obesity and the risk of cardiovascular diseases. Thus, paying attention to body composition is significant because of the high proportion of adipose tissue in the case of capacity to perform a work (9). Exercise is a good way to reduce fat, weight, and obesity, and thus, reduce the body index. It is also important to regulate body weight over time because it increases the metabolism level in the body while it is at rest (10). Previous studies have suggested that different types of exercise, including Pilates and aerobic exercise, have significant effects on anthropometric characteristics (11-13). Research evidence has shown that overweight and obesity are associated with increased serum lipids and lipoproteins (14). Understanding factors affecting the occurrence of cardiovascular diseases has a major role in preventing these diseases. The most important risk factors for these diseases are elevated total cholesterol (TC), triglyceride (TG), low density lipoprotein (LDL), very low density lipoprotein (VLDL), and high density lipoprotein (HDL) (15, 16). One study investigated the effect of Pilates training on body composition, body mass index (BMI), fat mass, and fat percentage in women. Pilates training was held for 12 weeks, two sessions per week, and one hour each session. The results showed that Pilates training decreased fat mass and fat percentage while increasing BMI, but had no change in body weight (17).
2. Objectives
Today, exercise training such as Pilates is being considered by some researchers. It should be noted that Pilates training does not require a high physical fitness and emphasizes muscles’ ability to maintain body balance (18). Performing Pilates requires no special skills and equipment; it is applicable to mattresses and for people with a normal fitness level (19, 20). Thus, this study aimed to examine the effect of Pilates training on body composition, lipid profile, and serum 25-hydroxy vitamin D levels in inactive overweight women.
3. Methods
3.1. Subjects, Study Design, and Exercise Protocol
The present study is a quasi-experimental study with a pretest-posttest design. The Ardabil University of Medical Sciences approved this study with the number IR.ARUMS.REC.1398.114. The statistical population consisted of overweight women in Ahwaz, Iran, with the age range of 25 to 35 years and BMI between 25 and 29 kg/m2, among whom 26 women were selected. The subjects were randomly divided into two groups: an exercise group with the age of 29.61 ± 3.61 years (n = 14) and a control group with the age of 30.12 ± 4.01 years (n = 14). There was no significant difference between the age (P = 0.435) and height (P = 0.259) of the two groups based on an independent t-test. The Pilates training program consisted of three 60-min sessions per week for 12 weeks. These exercises were divided into two parts: the first part on the mattress (the first six weeks) and the second part using bandages and ball (the second six weeks). The movements started from simple and then increased in intensity and complexity. The control group had no exercise training.
3.2. Blood Sampling
Blood samples were measured 48 hours before and after the last session of the training protocol on the 12-hours fasting status. Five ml of blood was drawn from each subject’s arm vein. After centrifugation for 10 min at a speed of 3000 rpm, serum was separated and shed in special microtubules and then held at -70°C. Serum 25-hydroxy vitamin D levels were determined using the IDS kit with the sensitivity of 1.6 ng/mL and the ELISA method. Also, cholesterol and triglyceride were measured using the Pars Azmun Company kit with the sensitivity of 3 mg/dL and 1 mg/dL, respectively. Moreover, LDL and HDL-C levels were measured using the Pars Azmun Company kit and the calorimetric enzymatic method with the sensitivity of 1 mg/dL.
3.3. Statistical Analysis
The study data were processed using SPSS software version 23. The data distribution normality and the homogeneity of the variances were evaluated using the Shapiro-Wilk test and Levon’s test, respectively. Afterward, paired t-test and ANCOVA were used to examine within- and between-group differences, respectively.
4. Results
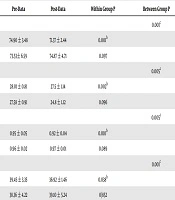
Table 1 shows the mean and standard deviation of the subjects’ anthropometric characteristics at baseline and the end of the excremental period in both groups. Based on ANCOVA, weight (P = 0.001), BMI (P = 0.005), the waist-to-hip ratio (WHR) (P = 0.005), and body fat percentage (P = 0.001) significantly decreased in the exercise group compared to the control group after 12 weeks of Pilates training. Table 2 shows the mean and standard deviation of serum 25-hydroxy vitamin D levels and lipid profile before and after 12 weeks in both groups. The ANCOVA results showed that the serum 25-hydroxy vitamin D levels (P = 0.005) and HDL-C (P = 0.028) significantly increased and triglyceride (P = 0.001) and cholesterol (P = 0.001) significantly decreased in the Pilates training group. However, there was no significant difference between the two groups regarding LDL-C levels (P = 0.435).
| Index | Pre-Data | Post-Data | Within Group P | Between Group P |
|---|---|---|---|---|
| Weight, kg | 0.001c | |||
| Training | 74.90 ± 3.48 | 71.37 ± 2.44 | 0.001b | |
| Control | 73.53± 6.59 | 74.87 ± 4.71 | 0.097 | |
| BMI, kg/cm2 | 0.005c | |||
| Training | 28.01 ± 0.81 | 27.5 ± 1.14 | 0.002b | |
| Control | 27.58 ± 0.91 | 24.8 ± 1.12 | 0.096 | |
| WHR, cm | 0.005c | |||
| Training | 0.95 ± 0.05 | 0.92 ± 0.04 | 0.010b | |
| Control | 0.96 ± 0.02 | 0.97 ± 0.01 | 0.089 | |
| Body fat percentage | 0.001c | |||
| Training | 39.45 ± 3.35 | 36.92 ± 1.46 | 0.038b | |
| Control | 38.36 ± 4.22 | 39.10 ± 5.24 | 0/852 |
Abbreviations: BMI, body mass index; WHR, waist-to-hip ratio.
aValues are expressed as mean ± SD.
bSignificant difference (P < 0.05) for within-group comparison.
cSignificant difference (P < 0.05) for between-group comparison.
| Index | Pre-Data | Post-Data | Within Group P | Between Group P |
|---|---|---|---|---|
| Triglyceride, mg/dL | 0.001b | |||
| Training | 158.35 ± 1.76 | 137.71 ± 2.80 | 0.001a | |
| Control | 155.77 ± 1.36 | 152.07 ± 2.47 | 0.108 | |
| Cholesterol, mg/dL | 0.001b | |||
| Training | 168.57 ± 2.35 | 149.42 ± 2.29 | 0.001a | |
| Control | 170.14 ± 2.75 | 166.35 ± 2.19 | 0.264 | |
| HDL-C, mg/dL | 0.028b | |||
| Training | 43.23 ± 0.84 | 47.30 ± 1.32 | 0.001a | |
| Control | 45.36± 1.58 | 43.69 ± 1.25 | 0.097 | |
| LDL-C, mg/dL | 0.435 | |||
| Training | 96.21 ± 2.45 | 93.57 ± 2.84 | 0.101 | |
| Control | 94.30 ± 1.95 | 95.57± 2.117 | 0.483 | |
| 25¬Hydroxy VD, ng/mL | 0.005b | |||
| Training | 12.32 ± 4.45 | 17.40 ± 3.78 | 0.011a | |
| Control | 13.17 ± 3.54 | 12.58 ± 2.28 | 0.094 |
aSignificant difference (P < 0.05) for within-group comparison.
bSignificant difference (P < 0.05) for between-group comparison.
5. Discussion
The present study’s main finding was significantly increased serum 25-hydroxy vitamin D levels after 12 weeks of Pilates training, which is consistent with AlMulhim et al.’s (21) and Hajinajaf et al.’s (5) study results. The results of Hajinajaf et al.’s study (5), examining the effect of aerobic exercise on serum 25-hydroxy vitamin D levels and anthropometric characteristics in obese and overweight women, showed a significant increase in serum 25-hydroxy vitamin D levels. Since obesity is associated with vitamin D deficiency (22), body weight, especially fat mass, is one of the factors adversely affecting vitamin D status and making it accessible to 25-hydroxy vitamin D. In contrast, physical activity results in weight loss and increases lipolysis and adipose tissue movement, thereby increasing serum vitamin D levels (23). Another notable factor is the effect of physical activity on bone mass, resulting in decreased calcium secretion and increased absorption efficiency. Increasing serum calcium can also save serum vitamin D levels (24). The present study had limitations such as lack of precise nutrition control, lack of sun exposure, and coverage of subjects. Nevertheless, one reason for the increase in serum vitamin D levels in the present study is subjects’ weight loss in the exercise group, although it was less than the normal value of 20 ng/mL. The main reason for vitamin D deficiency is body fat because vitamin D is soluble in fat and is readily absorbed by fat cells (25).
Other factors measured in the present study were changes in body parameters such as body weight, BMI, WHR, and body fat percentage, all of which significantly decreased after 12 weeks of Pilates training compared to the control group. Studies have shown that deep breathing during exercise increases the energy consumption of fats. The reason is that in addition to active muscles, involved respiratory muscles also consume energy (26). Since deep and diaphragmic breathing is one of the most important principles of Pilates training, Pilates training is expected to have significant effects on body fat mass, fat percentage, and weight (27, 28). This study’s results showed a positive effect of Pilates training on lipid profile. Concentrations of triglyceride, cholesterol, and HDL-C were significantly different in the training group compared to the control group, but the LDL-C levels were not significantly changed. Exercise more affects the lipid profile of people with higher TG and LDL-C levels or lower HDL-C levels (29). Studies with animal samples have also shown similar results. For example, 12 weeks of resistance training have been shown to improve lipid profile in healthy (30) and ovoctomized rats (31). Accordingly, eight weeks of resistance training improved lipid profile in rats with either fatty or normal diet (32). There was a significant decrease in total cholesterol and triglyceride levels in the exercise group compared to the control group after 12 weeks of Pilates training. It could be attributed to the intensity and duration of training, age, sex, and health status of the participants. Another measured factor was HDL-C level that significantly increased. There is sufficient consensus on positive changes in HDL-C, resulting from exercising in the research literature. Positive changes in HDL-C were observed even in a single session training. These results are consistent with Henderson et al.’s findings (33), but not with Arsalan et al.’s results (34). Major differences between those studies and the present study were the subjects’ characteristics and exercise protocols. For example, it appears that eight weeks of exercise are insufficient to improve some lipid profile indices. Muscle activity usually increases by subcutaneous aerobic activities to produce energy through fat metabolism, thereby increasing fat oxidation. In this type of exercise, the lipid oxidation rate is tripled due to an increase in epinephrine activity and a reduction in insulin stimulation.
The present study’s results also showed no significant change in LDL-C levels. One factor that could possibly justify the change of this parameter in the present study is the female subjects. It has been shown that changes in lipoprotein levels are usually less responsive to exercise in women than in men, partly due to lower cholesterol levels in women compared to men (35). The limitations of this study were lack of positive food intake, lack of nutritional status control, and lack of exposure to sunlight. Future studies should be carried out in a longitudinal fashion, taking into account the amount of sun exposure, nutrition status, and calorie intake of subjects.
5.1. Conclusions
According to the present study’s findings, it can be concluded that 12 weeks of Pilates training improved serum 25-hydroxy vitamin D levels and lipid profile in overweight women. It appears that Pilates training can be a suitable non-pharmacological approach for overweight people.