1. Background
Industrial use of metal oxide nanoparticles (i.e. titanium oxide, iron oxide, silicon, and etc.) has rapidly grown during the past decade. This has led to increase in the occupational and environmental exposure of humans and other species to nanoparticles [1]. Many in-vivo studies suggested that nanoparticles can be accumulated in the liver, kidney, lung, spleen, brain, and heart, and can generate various inflammatory responses [2-8].
There has been extensive research by scientists and experts on the broad applications of titanium dioxide (TiO2) nanoparticles in tumor therapy, drug and gene delivery to the cells and tissues, and labeling cells and macromolecules [9]. Therefore, few studies have been reported regarding their toxicity or side effects in vivo on cells and animals. Besides, the very limited numbers of studies that have been done in this field have had contradictory results.
For instance, Ge-yu et al. showed that transbronchial exposure to TiO2 nanoparticles (with a size of 50 nm, and 0.5, 5.0, and 50.0 mg/kg nano-TiO2) can induce oxidative stress in the liver and kidney but has no effect on the kidney and liver function that causes pathological changes [10].
In addition, Fabian et al. showed that TiO2 nanoparticles are well distributed through the liver, spleen, kidneys, and lungs, over time, without disruption of tissue. One month after the injection, their concentration in the tissues is reduced. Moreover, no change was observed in the cytokine and enzyme levels measured in blood samples; which means there was no considerable inflammatory response or tissue toxicity [11].
Chen et al. studied the acute toxicity of titanium nanoparticles in vivo in mature rats and the effects of nanoparticles were tested on the hepatic parameters, and liver, kidney, and lung tissues at different times (24 and 48 hours; 7 and 14 days). The results showed elevated levels of liver markers Alkalynphosphatase (ALP), glutamate oxaloacetate transaminase (SGOT) and glutamate pyruvate transaminase (SGPT). Furthermore, accumulation of the nanoparticle in the liver, kidney, and lung tissues has been observed. Hepatic necrosis, apoptosis, hepatic fibrosis, renal glomerular inflammation, pneumonia, and pulmonary thrombosis were other conclusions and observations of this study [12].
The difference in tissue distribution, penetration, and damage of nanoparticles in various studies is probably due to the difference in their synthesis method that leads to changes in size, shape, and other physical and chemical properties of nanoparticles. Therefore, the impact and interaction of nanoparticles with cells and tissues of animals will be different [13].
Because of the very characteristics of TiO2, a risk assessment of potential adverse effects of nanoparticles on human health and environmental pollution is necessary. Accordingly, in the present study, impact of intraperitoneal injection with different doses of spherical-shape TiO2 nanoparticles with a diameter of 10 - 15 nm, and inorganic nature, and wet synthesis in the liquid phase, on the function of liver, lung, kidney, and liver enzymes was studied in Wistar male rats.
2. Materials and Methods
2.1. Characterization of TiO2 Nanoparticles
Two thousand fifty mL TiO2 was purchased from Tehran Neutrinoy Co. (Iran) that imports nanoparticles from Spain. In order to make sure of the size of the nanoparticles, 1 g of them was sent to the department of Materials Engineering of the Islamic Azad University (Najafabad branch), and that center confirmed the validity of the nanoparticles size using X-ray tests.
The used TiO2 nanoparticles were dissolved with the following specifications: density of 3.84 g/mL, specific surface area (SSA) of 100 - 150 m2/g, diameter of 10 - 15 nm, 21.2% rutile and 78.8% anatase, purity of 99.9% and serum concentration of 1000 ppm.
2.2. Mother Solution Preparation
Mother solution was prepared through the following procedure for TiO2 nanoparticles:
A) 1 mL TiO2 nanoparticle with a concentration of 1000 ppm + 10 mL double distilled sterile water by Merck, Germany = 10 ppm concentration of titanium dioxide nanoparticles.
B) 10 mL TiO2 nanoparticles with a concentration of 1000 ppm + 100 mL double distilled sterile water by Merck, Germany = 100 ppm concentration of titanium dioxide nanoparticles.
C) 30 mL TiO2 nanoparticle with a concentration of 1000 ppm + 300 mL double distilled sterile water by Merck, Germany = 300 ppm concentration of titanium dioxide nanoparticles.
2.3. Animals, Treatment, and Blood Biomarker Assay
In an experimental study, 32 male Wistar rats (mean weight: 225 ± 25 g) were purchased from the Animal Center of Shahrekord university and housed in stainless steel cages in a ventilated animal room. Room temperature was maintained at 20 ± 2°C, with relative humidity of 60 ± 10%, and a 12 hour light/dark cycle.
Distilled water and sterilized food for rats were available ad libitum. They were acclimated to this environment for 7 days prior to dosing. After two weeks of accommodation, rats were randomly divided into 4 groups: control (treated with 0.5 mL normal saline) and 3 experimental groups. Group 1, 2, and 3 received 0.5 mL of solution containing 10, 100, and 300 ppm TiO2 via intraperitoneal (i.p.) injection for 7 successive days, respectively.
The effects of TiO2 nanoparticles on serum levels of glutamate oxaloacetate transaminase (SGOT) and glutamate pyruvate transaminase (SGPT) were evaluated at 2nd, 7th and 14th days after the treatment. All animals (at 14th day) were anesthetized by ether and sacrificed for histological assessment.
All animal handling and manipulation procedures were performed according to the guideline of the animal welfare act and the experimental protocols were approved by the office of research ethics committee of university of Shahrekord.
2.4. Biochemical Analysis of Liver Function
Serum was collected by centrifuging blood at 2,500 rpm for 10 minutes. Liver function was evaluated based on the serum levels of SGOT and SGPT. All biochemical assays were performed using a clinical automatic chemistry analyzer (Hitachi Automatic Analyzer 902, Roche, Germany) and Pars Azmon kits (Tehran, Iran).
2.5. Histopathological Examination
Histological observations were performed according to the standard laboratory procedures. A small piece of lung or liver fixed in formalin 10% (v/v) was embedded in a paraffin block, sliced into 5 μm thicknesses and then placed onto glass slides. The section was stained with hematoxylin-eosin (HE) and examined by light microscopy.
All data were analyzed by using the statistical package for social sciences (SPSS-19) software and were summarized and expressed as mean and standard error (mean ± SEM). Statistical analysis of data was performed by Multivariate Analyses of Covariance model (MANCOVA), while serum values at 2, 7, and 14 day after intervention using as dependant variables and baseline value of serum was controlled. We compare between groups. We using Wilk’s lambda or Roy largest Root for totally differences between groups, P Value about each depended variables was reported in last row of tables, the Bonferroni posttest was used to paired comparisons. P value less than 0.05 were considered significant.
3. Results
According to MANCOVA P = 0.023; the mean serum glutamic oxaloacetic transaminase (SGOT) was significantly greater in groups receiving TiO2 (10 ppm and 300 ppm) than the control group 2 days after the intervention; P = 0.002 and P = 0.009, respectively. Seven and 14 days after the injection of TiO2 10 ppm and TiO2 300 ppm the liver damage was returned (Table 1).
| Groups | SGOT | ||
|---|---|---|---|
| 289.3 ± 7.0 | 279.5 ± 12.8 | 201.6 ± 9.0 | |
| 275.4 ± 7.8 | 268.3 ± 11.8 | 204.6 ± 6.4 | |
| 312.5 ± 15.5 | 249.3 ± 13.6 | 194.9 ± 15.4 | |
| 220.4 ± 13.4 | 253.3 ± 9.4 | 213.5 ± 15.4 | |
| 0.001 | 0.294 | 0.886 | |
According to MANCOVA P = 0.001; intergroup difference was observed in mean serum glutamic pyruvic transaminase (SGPT). Bonferroni posttest was showed the mean SGPT 2 days after the intervention was greater in the group receiving 300 ppm TiO2 than the groups receiving 100 ppm (P = 0.027) and 10 ppm TiO2 (P < 0.001) (Table 2).
| Groups | SGPT | ||
|---|---|---|---|
| After 2 Days | After 7 Days | After 14 Days | |
| 129.3 ± 5.2 | 158.9 ± 12.2 | 146.0 ± 6.3 | |
| 116.8 ± 7.1 | 156.4 ± 12.9 | 154.8 ± 7.9 | |
| 171.3 ± 7.1 | 165.1 ± 13.9 | 166.6 ± 12.6 | |
| 158.3 ± 10.4 | 170.6 ± 12.0 | 174.0 ± 13.6 | |
| < 0.0001 | 0.811 | 0.309 | |
3.1. Liver Histopathological Evaluation
The histological photomicrographs of the liver sections are shown in Figures 1 - 3.
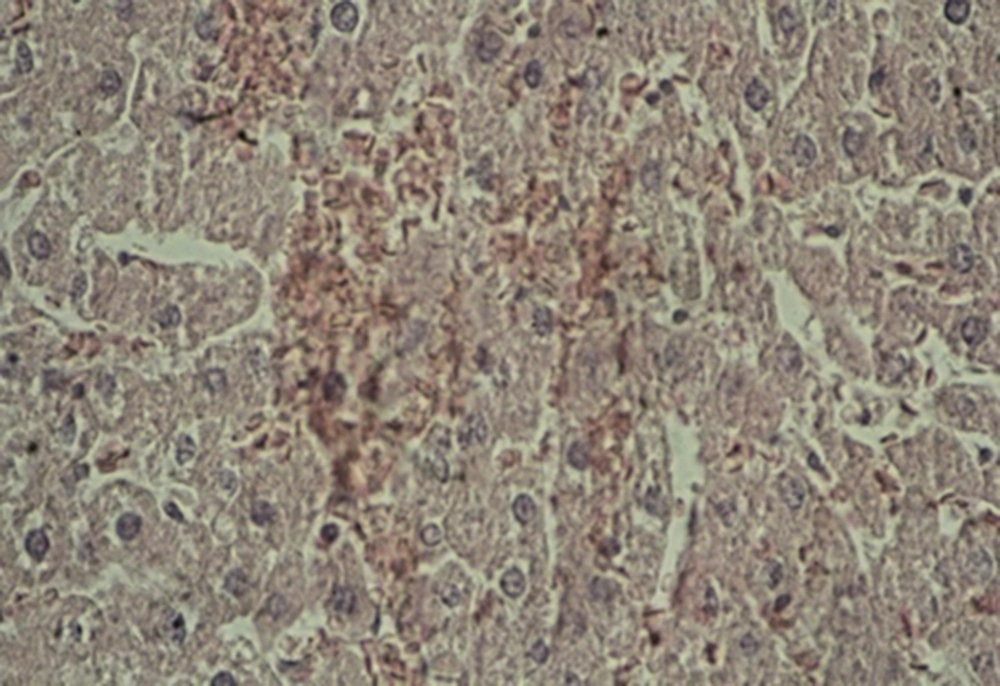
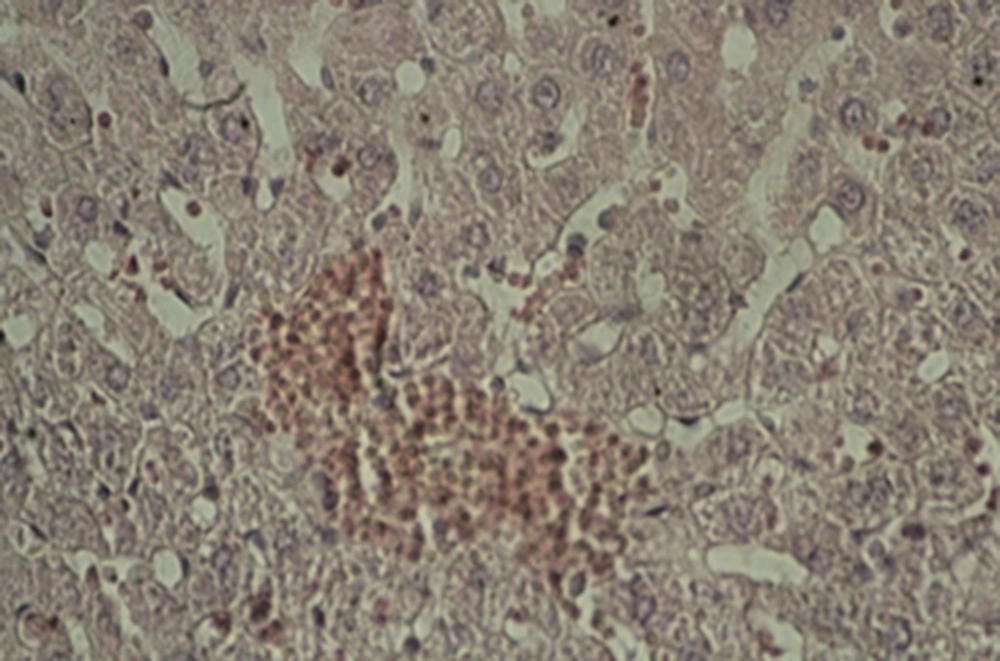
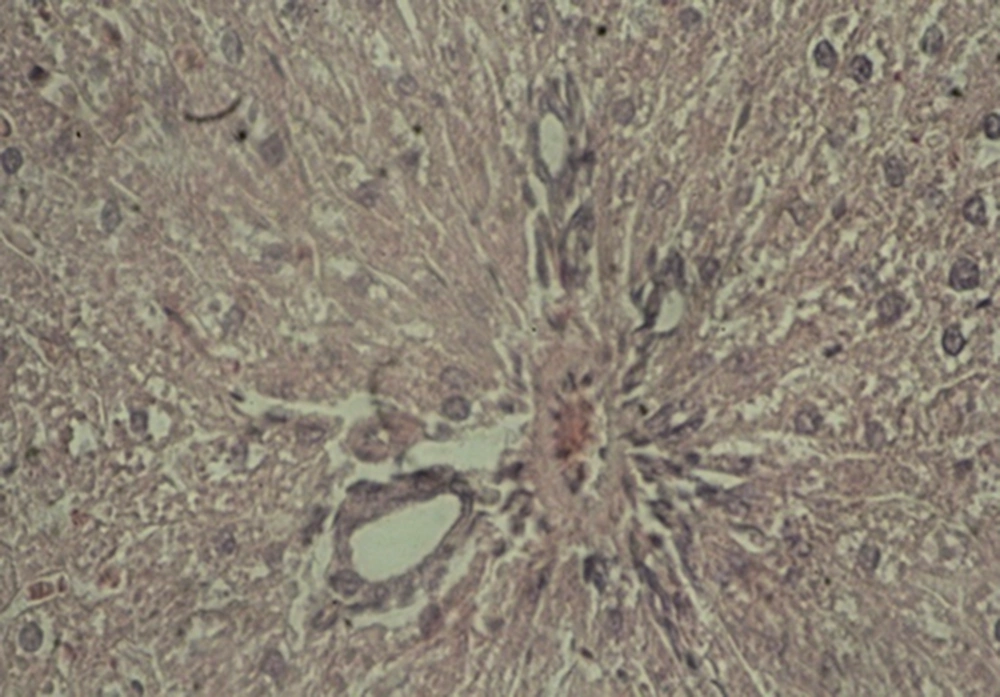
A: Experimental group 1 (10 ppm TiO2): hepatocyte vasculature-hypertrophy, approximate elimination of hepatic lobules, and weak acidophilic hepatocyte indicate the impact of the mentioned nanoparticles and prevalence of histopathological changes (Figure 1).
B: Experimental group 2 (100 ppm TiO2): decreased effect of hepatocyte acidophilic, elimination of hepatic lobules, and central venous hyperemia were considerable histopathological changes in this group (Figure 2).
C: Experimental group 3 (300 ppm TiO2): significant shrinkage of central veins, immediate hyperemia, elimination of hepatic lobules, hepatocyte hypertrophy, sinusoidal thinning, and decreased effect of hepatocyte acidophilic were seen in this group. Besides, a reduction was observed in the size of hepatic artery and portal vein in the portal triad (Figure 3).
3.2. Lung Histopathological Evaluation
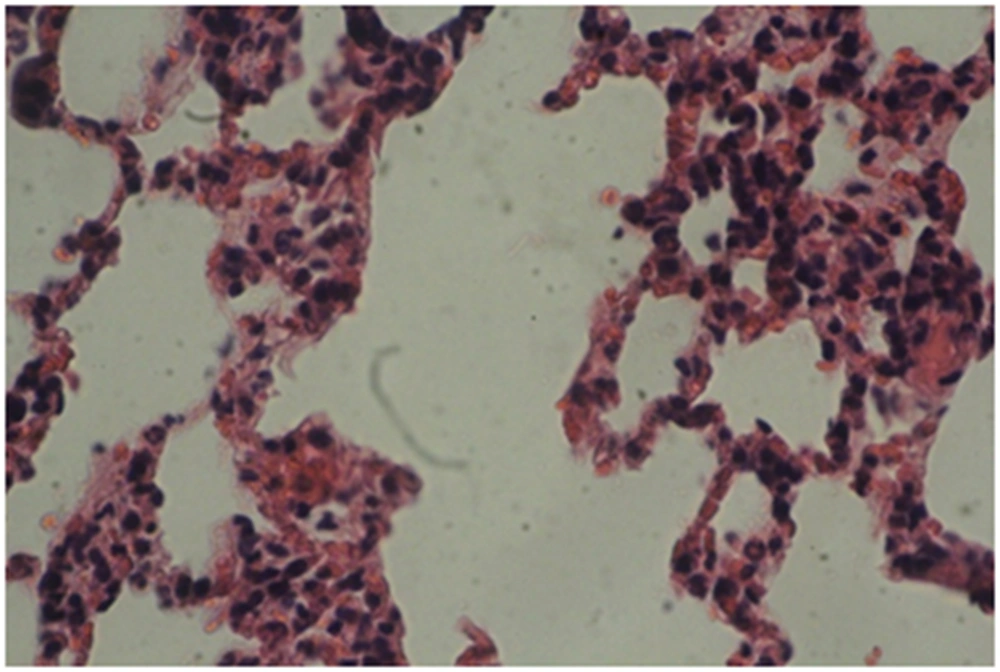
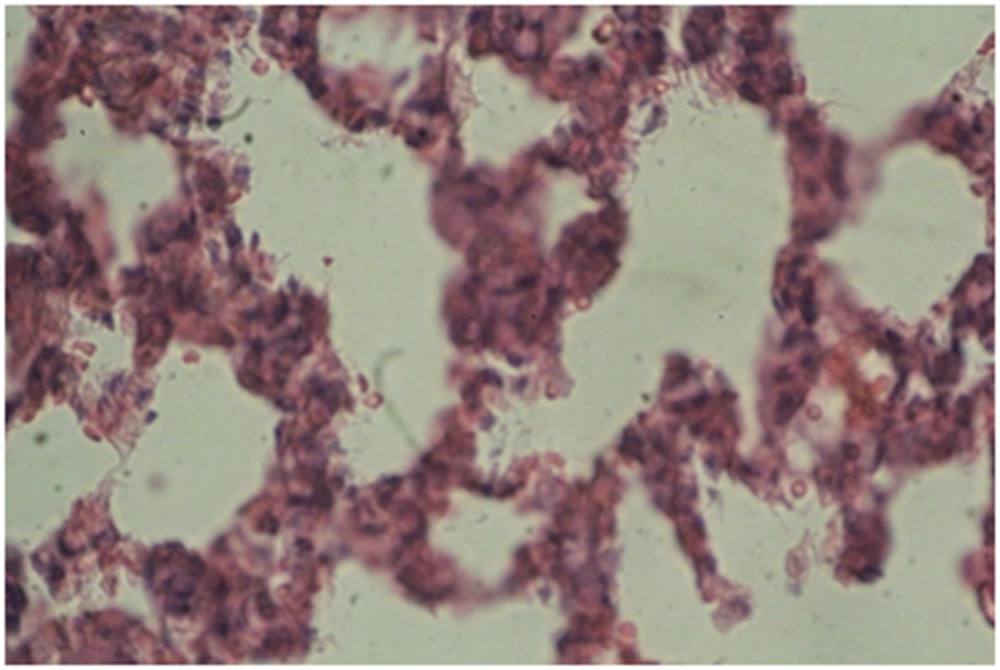
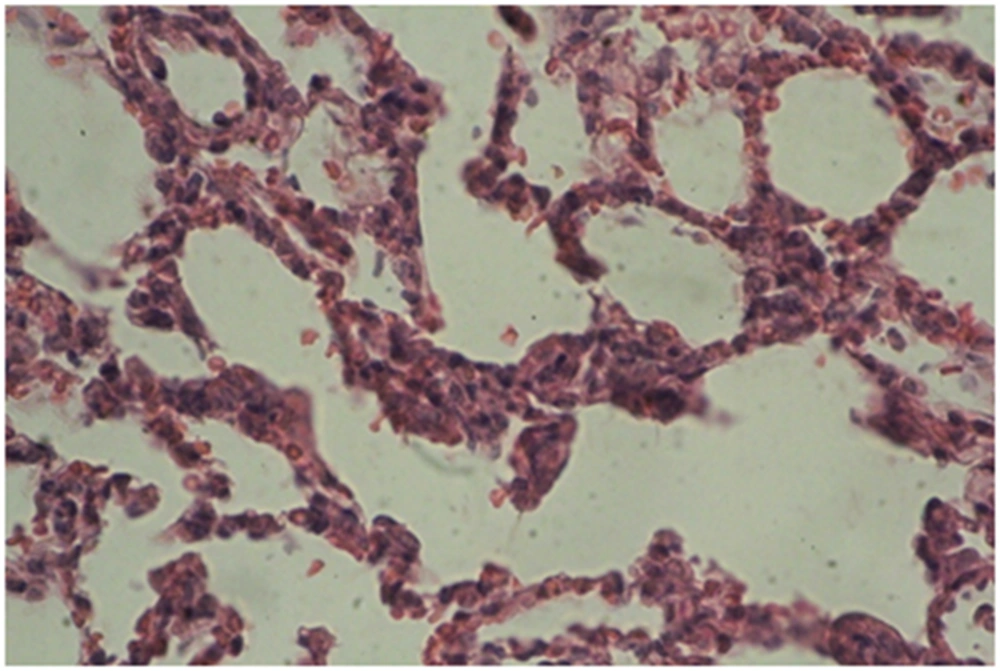
Photomicrographs of the lung sections are shown in Figures 4 - 6.
A: Experimental group 1 (10 ppm TiO2): according to Figure 4, destruction and overlapping of alveolus indicates atelectasis. Moreover, alveolar wall thickening and vasculature hyperemia were seen in this treatment group (Figure 4).
B: Experimental group 2 (100 ppm TiO2): according to Figure 5, destruction of alveolus, eliminating alveolus, and vasculature hyperemia are evident (Figure 5).
C: Experimental group 3 (300 ppm TiO2): according to Figure 6, increased thickness of alveolus walls and connective fibrous space are the most considerable changes in this group (Figure 6).
4. Discussion
The results of this study showed that mean liver enzyme SGOT was significantly greater in groups that received 10 ppm and 300 ppm TiO2 than the control group two days after the intervention. Seven and 14 days after the injection, the liver damage returned in both groups. Maybe, the other liver enzyme (such as metalothioneine) activated and neutralized the effects of TiO2 after 7 days. In addition, mean SGPT, two days after the intervention, in the group that received 300 ppm TiO2 was significantly greater than the groups that received 100 ppm TiO2 and 10 ppm TiO2. Higher concentrations of titanium dioxide (300 ppm TiO2) had a higher toxicity on hepatic enzymes than other concentrations. No difference was observed between the experimental and control groups in terms of mean SGPT. Furthermore, the histological results of liver in group 1 showed hepatocyte vasculature, hypertrophy approximate, in group 2: decreased effect of hepatocyte acidophilic, elimination of hepatic lobules, in group 3: significant shrinkage of central veins, immediate hyperemia showed and histological results in lung: destruction of alveolus and their overlapping (group 1) and vasculature hyperemia in all groups showed. The results indicated intense histopathological changes in lung and liver tissues by 10 - 15 nanometer spherical-shaped TiO2 nanoparticles in all three experimental groups.
Special features of nanoparticles, such as their small size, shape, high surface area, and their specific construction, are the reasons for the biomedical and industrial applications of these particles [14, 15]. However, in recent years, evidence of the adverse impact of nanoparticles has been reported, such as increased mortality, cardio-respiratory diseases, and malignant asthma [16].
Researchers showed that TiO2 nanoparticles can damage DNA and cause cell death by induced oxidative stress. In addition, these nanoparticles can produce reactive oxygen species (ROS) and reduce cell antioxidants such as glutathione and vitamin E [17]. ROS is the physiological products generated during aerobic metabolism in mammalian mitochondria. The intracellular ROS level is balanced through balancing the metabolism (by antioxidant enzymes and scavengers). A number of possible signaling pathways can describe the ROS association with apoptosis including death pathways involving cell-surface receptors (external) and mitochondrial pathways (internal) [18].
Ma et al. reviewed the acute liver damage in mice caused by anatase TiO2 nanoparticles. In this study, nano-anatase TiO2 (5 nm) were injected into the abdominal cavity of ICR mice for 14 days and the inflammatory responses in the mice’s liver was examined. Results of this study demonstrated the concentration of titanium observable in the liver DNA, histopathological changes, apoptosis in liver hepatocytes, and liver dysfunction with higher doses of nano-anatase TiO2 [19].
Afaq et al. investigated the titanium dioxide nanoparticle toxicity in rats induced by intratracheal instillation. The peroxidation of lipid and hydrogen peroxide radicals did not change with an increase in the activity of glutathione peroxidase, glutathione reductase, 6-phosphate dehydrogenase, and glutathione S-transferase. This indicates the induction of antioxidant enzymes in animals by nano-TiO2 (30 nm) [20].
Ma et al. showed that high doses of nano-anatase TiO2 (5 nm) can cause liver dysfunction, and induced invasion and oxidative stress in rat’s liver [21].
Although it is clear that TiO2 nanoparticles or other nanoparticles can induce serious toxicity in the liver, the molecular mechanisms and pathogenesis have not been identified yet. When TiO2 nanoparticles stimulate hepatocyte, they are able to induce inhibitory proteins such as phosphorylated IkBs (nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor) and analyze it, and then inhibit NF-kb (nuclear factor kappa-light-chain-enhancer of activated B cells) activity that ultimately leads to the transcription of proinflammatory and inflammatory cytokine genes in rat’s liver [22].
Liu et al. investigated the biochemical toxicity of nano-anatase TiO2 in mice. In order to evaluate the toxicity of these nanoparticles, they were injected into the abdominal cavity of ICR (imprinting control region) mice for 14 days and the coefficient of the organs and serum biochemical parameters were examined. The results showed that increased dose of these nanoparticles gradually increased the coefficients of liver, kidney, and spleen. Moreover, it gradually decreased the lung and brain coefficients, and the change in brain coefficients was not significant. The order of titanium concentration in organs included: heart < brain < lung < spleen < kidney. Reviewing serum biochemical parameters with a low dose of titanium dioxide nanoparticles, showed a small difference with the control group. However, a high dose of such nanoparticles significantly increased indicators of liver function, including alkaline phosphatase, alanine, leucine acid peptide, pseudocholinesterase, and total protein and albumin. Renal function indicators, including blood urea nitrogen and uric acid decreased. Indicators of heart function such as activity of aspartate aminotransferase, creatine kinase, dehydrogenase lactate, and alpha-hydroxy butyrate dehydrogenase increased [23].
Olmedo et al. reported that six months after intraperitoneal injection of TiO2 nanoparticles, the deposition of the nanoparticles in organs such as liver, spleen, and lungs was observed [24]. Results of the study by Huggins and Froehlich showed that after the intravenous injection of 250 mg/kg TiO2 (size 0.2 - 0.4 nm) in rats, almost 69% of the injected nanoparticles of titanium dioxide at 5 minutes and 80% of injected TiO2 nanoparticles at 15 minutes were accumulate in the liver. The results indicated that liver deposition was the highest amount among the other tissues [25].
Wang et al. fed TiO2 nanoparticles to mature rats and evaluated them after two weeks. They observed that 25 and 80 nm nanoparticles in 5 g/kg dose had caused a toxic effect on them and influence biochemical parameters such as ALT, AST, ALP, and Liver pathology. They also did this through intravenous and intraperitoneal injection. Due to the agglomeration of the nanoparticles in liver and kidney cells, liver damage, renal pathological changes and renal inflammation were observed as glomeruli and nephron-like toxic damage to the liver hepatocytes around the central vein lobules. Accumulation of these substances caused abnormal pathological changes in the tissues of the heart, lung, testis, ovary, and spleen [26].
Alkaline phosphatase exists (is distributed) in the liver, bone, bile duct, and ALT and AST in the liver, heart, and other organs. When organs are damaged, the activity of ALP, ALT, and AST increases. It has been indicated that LDH is an important isozyme in glycolysis and glycogen which are present in many tissues and the heart and liver. When tissues are damaged, LDH is released into the blood serum from the organs or cells, leading to an increase in LDH activity and isozymes in similar organs. Pseudocholinesterase (PChE, acylcholine acyl hydrolase), which is found in many animal tissues, acts with low-density in the metabolism of lipids and lipoprotein. When the liver is damaged, PChE activity is dramatically increased which causes impaired lipid metabolism and low-density lipoprotein [27].
In order to find out that apoptotic hepatocyte is induced through the use of intragastric TiO2 NPs (nanoparticles) for 60 consecutive days, apoptosis of hepatocytes, oxidative stress parameters, and the amount of expression of genes associated with stress in the liver of mice were examined by Cui et al. Results showed that hepatocyte apoptosis, oxidative stress, and altered expression of genes are associated with regulated metabolism/detoxification of titanium dioxide nanoparticles and radical scavenging activities [27].
In this study, the toxic effect of spherical-shaped TiO2 nanoparticles with a diameter of 10 - 15 nm was evaluated on the function of liver and lung and SGPT and SGOT liver enzymes levels. We faced with increased concentration of such enzymes in high doses of nanoparticles. Due to damaged liver cells, such enzymes (which are inside the hepatocyte cell) are released into the blood. Therefore, a high amount of such enzymes indicates destruction of liver cells.
Various reports indicated that significant amounts of nanoparticles injected to the body are absorbed by the liver. The reticuloendothelial system in liver can gradually remove the accumulated nanoparticles from the body [28].
Understanding the specific mechanisms of nanoparticles and their reaction requires very extensive research in this area. When nanoparticles are accumulated in a tissue, they may either be absorbed into the cell or no absorption may occur. If these nanoparticles are absorbed, the final cellular replacement in the lysosomal and cytoplasm of the cell will depend on the characteristics of the nanoparticle. If nanoparticles are located in the cytoplasm, some large size material can cause direct damage or cell death by such interactions [29].
It seems that the main mechanism of toxicity is through oxidative stress causes damage to the lipids, carbohydrates, protein, and DNA. Probably the pathological changes in liver tissue are caused by accumulation and deposition of nanoparticles in this tissue [30].
On the other hand, the entry of TiO2 nanoparticles in this study shows their passing through blood and air barrier and damaging the lung tissue. Among the applications of nanoparticles that help in biological and medical fields is the influence and accumulation of effective nanoparticles in cells and various body organs after its injection [31]. Given the current findings, it can be concluded that application of 10 - 15 nanometer spherical-shaped TiO2 nanoparticles in vivo, even in small amounts for medical purposes, causes cell toxicity. Further researches are needed to study its effects on organs and blood factors.