Introduction
All-trans-retinoic acid (atRA), a bioactive vitamin A metabolite, is a signaling molecule indispensable for the formation of many organs, including eyes, heart, and kidneys [1]. atRA is an important physiological regulator of cellular differentiation, proliferation, apoptosis, reproduction and embryonic development in many species [2]. Retinoic action is mediated by specific nuclear retinoic acid receptors and retinoid receptors belonging to the steroid/thyroid super-family of transcription factors [3]. Inadequate levels of retinoids (excess or deficiency) may result in a set of defects denoted retinoic acid embryopathy which may provoke defects in the development of the neural crista [4], in addition to limb malformation [5] and other skeletal malformations [6].
atRA affects by serving as an activating ligand of nuclear atRA receptors (RAR α, β, and γ) and peroxisome proliferator-activated receptors (PPAR β/δ), which form heterodimers with retinoid X receptors [7]. The concentration of atRA during embryonic development is tightly controlled in a spatial and temporal manner, and in adult tissues, it is maintained within a very narrow range that is specific for each given tissue. If the control mechanisms fail and the concentration of atRA exceed or fall below the optimal range, tissues and cells undergo pathophysiological changes that in most severe cases can lead to disease [8].
Embryopathy due to RA is being intensely investigated in view of the teratogenic potential of retinols and of the crucial role played by their receptors in embryo development. The RA dose of 70 mg/kg was administered in mice on the eighth gestational day which caused neural tube defect (NTD), omphalocele, gastroschisis, limb defects, imperforated anus and tail agenesis/alteration. atRA increases the production of reactive oxygen species and oxidative stress [9].
On the other hand, a group of supplements that are good candidates for antioxidant therapy are flavonoids. Flavonoids are a class of polyphenolic compounds available in the fruits and vegetables. Epidemiological studies showed that the risk of cardiovascular disease in subjects, who had a high intake of flavonoids, has been reduced [10]. Quercetin (3, 30, 40, 5, 7-pentahydroxy-flavone) is a flavonoid commonly found in frequently consumed foods, including apples, berries, onion, tea, nuts, seed and vegetables that represent an integral part of the human diet. Quercetin is one of the most abundant representing the 60%-75% of the average polyphenol ingestion [11]. Quercetin has been reported to have biological, pharmacological, and medicinal activities [12] that are believed to arise from its antioxidant properties [13].
Quercetin was reported to have many beneficial effects on human health, including cardiovascular protection, anticancer activity, antiulcer activity, anti-allergic activity, cataract prevention, anti-inflammatory and antiviral activity [14]. Quercetin could prevent oxidant injury and cell death by several mechanisms, such as scavengering oxygen radicals, protecting against lipid peroxidation and chelating metal ions [15]. Quercetin directly scavenges the superoxide anion [16] and inhibits several superoxide-generating enzymes such as xanthine oxidase (XO) [17] or the neutrophil membrane NADPH oxidase complex [18]. As mentioned above atRA increases oxidative stress and quercetin acts as antioxidant; so, in present study, the preventive effect of quercetin on atRA-induced neural tube and skeletal malformations in rats was evaluated.
Results
No maternal deaths were observed throughout the course of this study. Likewise, the dose of atRA used in this investigation was well tolerated by the dams, as evidenced by no differences in food and water consumption.
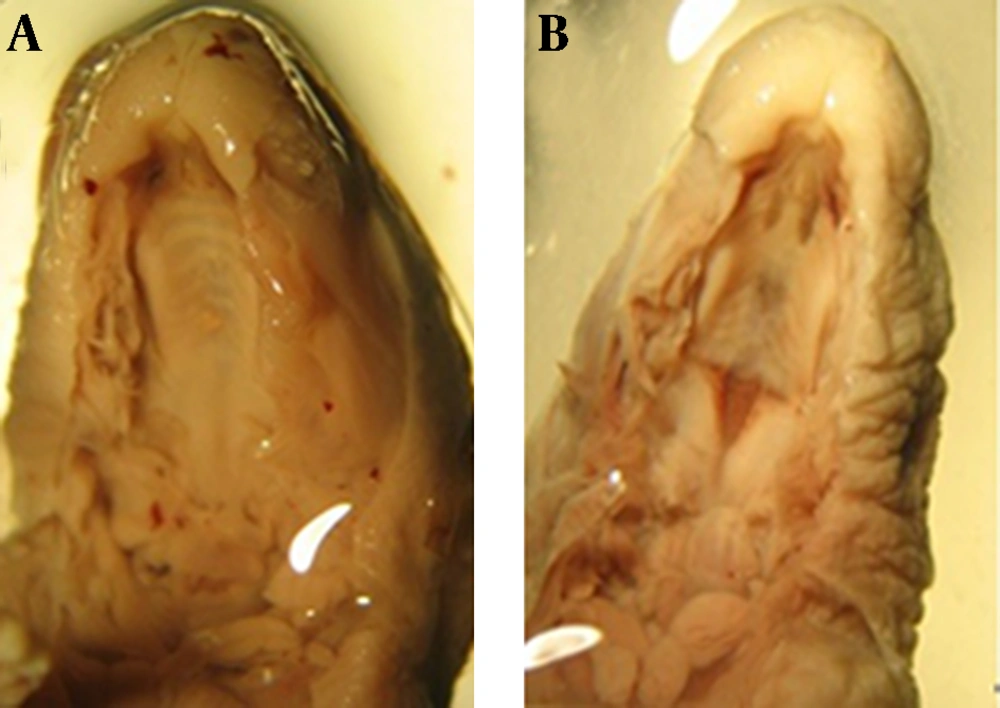
Fourty two fetuses were obtained from seven rats of control group. There were not observed macroscopic anomalies in the control animals. In the control group palatal closures of fetuses were normal (Fig. 1, 2-A) at gestational day 20 (i.e., palatal shelves had grown vertically on the sides of the tongue, then horizontally to meet and fuse).
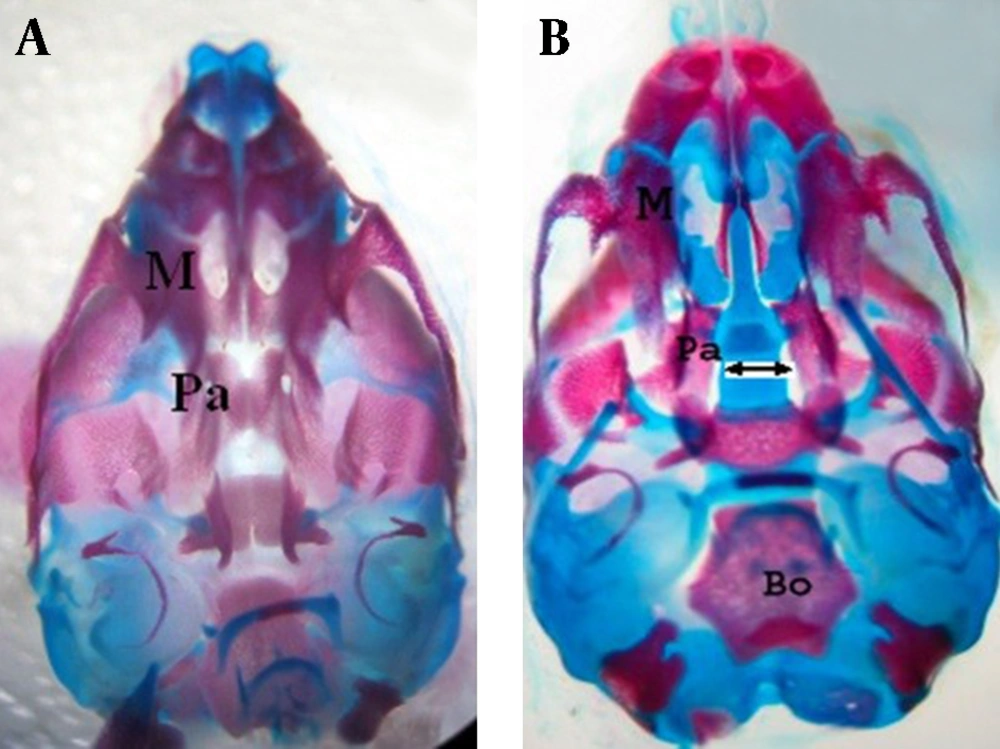
atRA induced cleft palate, exencephaly and spina bifida 30.76%, 61.53% and 30.76% incidence respectively. Quercetin with dose of 75 mg/kg reduced incidence of atRA-induced cleft palate, exencephaly and spina bifida to 11.11%, 16.66% and 5.55% respectively (Fig. 1, 2, 3). Quercetin 200 mg/kg reduced incidence of atRA-induced cleft palate, exencephaly and spina bifida to 10.52%, 10.52% and 0% respectively.
Percentages of absorbed fetuses were 66.66, 47.05 and 45.71 in groups 2, 6 and 7, respectively, so quercetin decreased the resorption rate.
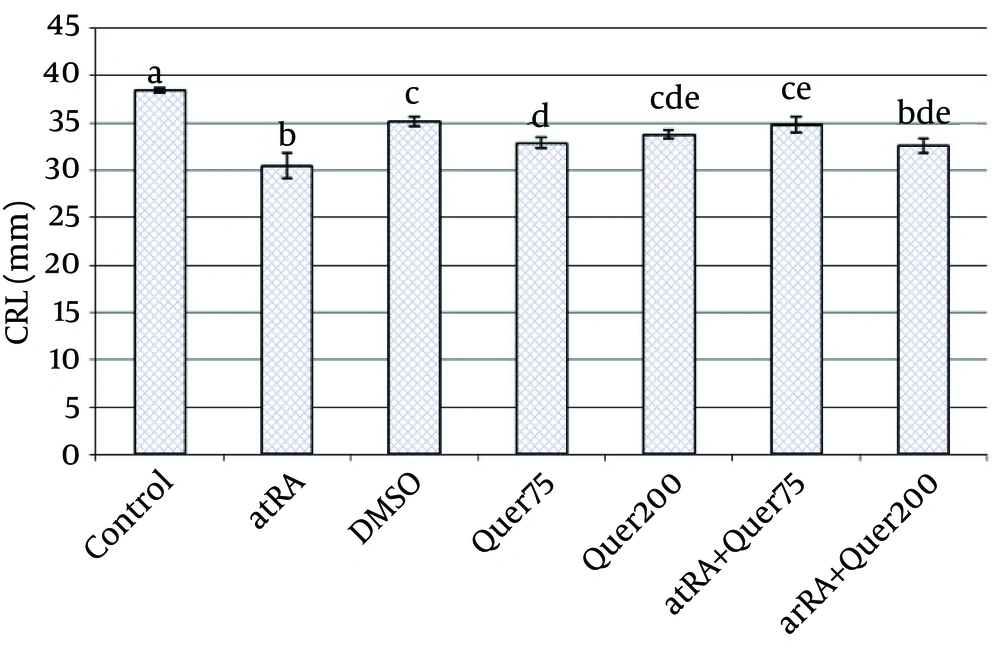
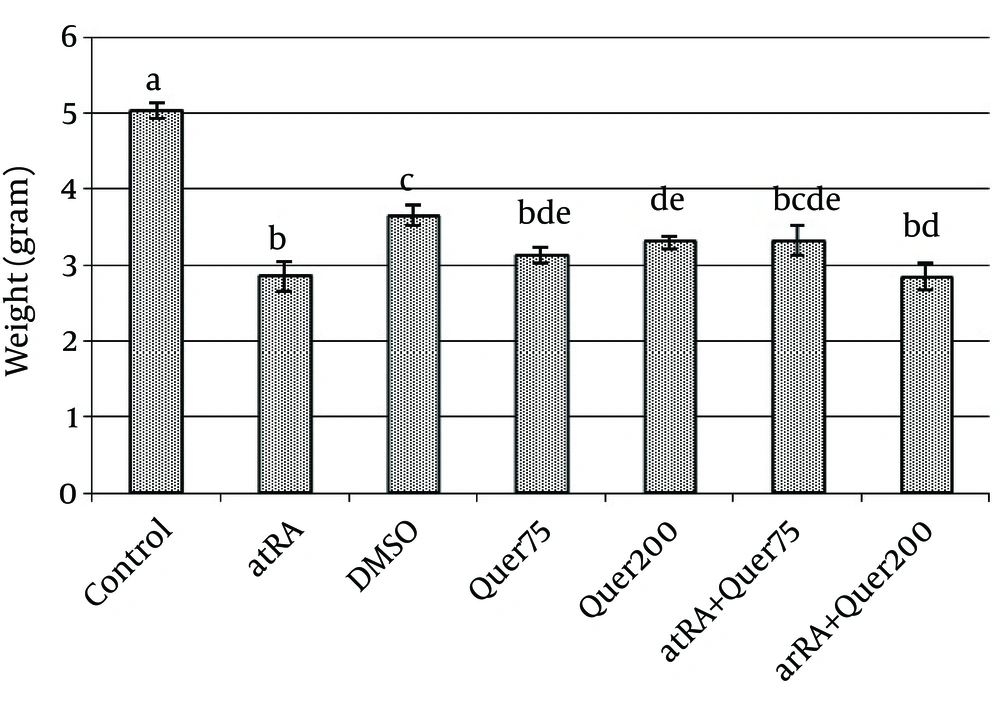
The mean of weight of animals' fetuses that received atRA in 8-10th days was significantly (p<0.0001) decreased in comparison with normal saline group. The mean of length of animals' fetuses that received atRA plus quercetin (75 mg/kg) in 8-10th days was significantly (p<0.0001) greater than the group received only atRA.
The mean weight and length in the group that received quercetin (200 mg/kg) did not differ significantly with atRA group.
The mean weight (p<0.0001) and length (p<0.0001) were significantly decreased in the group which received quercetin in comparison to the control group (Fig. 4, 5).
| Anomaly | Incidence (%) | ||
|---|---|---|---|
| Group 2 | Group 6 | Group 7 | |
| 30.76 | 11.11 | 10.52 | |
| 61.53 | 16.66 | 10.52 | |
| 30.76 | 5.55 | 0 | |
Group 2 received all-trans-retinoic acid (atRA) (25 mg/kg); Group 6 received atRA+quercetin (75 mg/kg); Group 7 received atRA+quercetin (200 mg/kg)
Discussion
We demonstrated atRA (at dose 25 mg/kg, i.p.) decreased weight and length and produced cleft palate (30.76% present of fetuses), exencephaly (61.53% present of fetuses) and spina bifida (30.76% present of fetuses).
The results presented here show that quercetin administration during the gestational period has a partial protective effect on atRA-induced teratogenesis (decreasing the frequencies of exencephaly, cleft palate, spina bifida). It is well established that atRA is an important physiological regulator of embryonic development; it regulates many processes in organogenesis such as development of important organs and systems including the heart, the cardiovascular system, the hindbrain, and the foregut, among others [6]. However, both its deficiency and excess can result in abnormal embryonic development. When atRA is administered in large doses during this critical period GD 8-10, it causes embryonic malformations in a dose-dependent manner [20]. Inappropriate gene expression has been proposed as a mechanistic basis for atRA teratogenicity. Morphological changes visible after atRA treatment of embryos could be explained by alterations in the spatial and temporal patterns of expression of genes controlling differentiation, proliferation, apoptosis, and morphogenesis in embryonic organization and in initial axial patterning [21].
Kistler reported that retinoic acid (120 mg/kg) was orally administered to pregnant females was highly embryolethal when administered on days 9 and 10 of gestation (96.2 and 100% resorptions). The earliest teratogenic effect of retinoic acid was noted on the 9th day of gestation. Severe multiple defects were produced by retinoic acid administration on days 9 and 11 of gestation, but more specific malformations involving the axial skeleton, the fore- and hind limbs, and cleft palate resulted from treatment on days 12-18 of gestation [22].
Tom et al. reported after maternal treatment of mouse with 5 mg/kg retinoic acid on 8.5 days of gestation, 53% of SELH/Bc mouse embryos had exencephaly, compared with 22% in ICR/Bc and 14% in normal strain (SWV/Bc) [23]. Dorko et al. reported that the application of atRA on 7-9th day of gestation influenced the weight of new-born rats [24].
A wide spectrum of congenital abnormalities, including exophthalmos, microphthalmia and anophthalmia, maxillo-mandibular dysostosis, micrognathia of both maxilla and mandible, cleft palate, subdevelopment of ear lobe, preauricular tags and macroglossia, were observed in the offspring of retinoic acid treated animals. The abnormalities were both time and dosage dependent and characteristic of Treacher-Collins syndrome when retinoic-acid was administered on the 11.5 days of gestational age. In contrast, when retinoic acid was administered were on gestational days 10-12, the defects were similar to those seen in the first and second pharyngeal arch syndrome, as well as in the oculo-auriculo-vertebral spectrum [25].
atRA plays important role in the control of cell differentiation and morphogenesis during prenatal development [26]. However atRA, used in the treatment of dermatological disorders, has been implicated in the production of congenital anomalies in infants born to mothers taking the drugs during the first trimester [27]. In mice, administration of RA leads to small embryo forebrain and hindbrain, exencephaly, little or no flexure of the brain, and optic vesicle aplasia [28]. Similar studies have shown that atRA given after the peri-implantational period induces abdominal wall and neural defects [29]. Our findings are consistent with previous research.
In one study, observed administration time-dependent changes in the teratological effects. Maternal administration of atRA in group II rats was associated with congenital malformations including microcephaly, exencephaly, hydrocephaly, gastrochisis, omphalocele, exophthalmus, mandibular hypoplasia, facial dysmorphia, limb reduction defects and reduction of the crown-rump length. Colakoğlu and Kükner conclude that atRA is more teratogenic before neurulation. The atRA administration during this period leads to severe developmental retardation and malformed stillborns. atRA is less teratogenic after neurulation, and causes only a few omphalocele [30].
Also, we observed protective effect of quercetin on atRA teratogenicity. This effect reported by some researchers. For example; Prater et al. reported that low-dose quercetin (66 mg/kg supplemented in rodent chow throughout gestation; approximately 70% of human dose), high-dose quercetin (333 mg/kg supplemented in rodent chow throughout gestation; approximately 3.5x daily human dose), impairs placental oxidative stress and fetal skeletal malformation induced by methylnitrosourea [31].
Song et al. reported that quercetin has protective effects on the spinal cord by the potential mechanism of inhibiting the activation of p38MAPK/iNOS signaling pathway and thus regulating secondary oxidative stress [32]. Gupta et al. reported that quercetin (10, 30 and 100 mg/kg for 5 consecutive days) ameliorates the diethylnitrosamine induced hepatotoxicity in rats and can be a candidate for a good chemoprotectant [33].
In one study, quercetin was administered at a dose of 10 mg/kg/day, i.p. for 14 days, which results call into question the ability of therapy with the antioxidant quercetin to reverse diabetic oxidative stress in an overall sense [34]. In another study, quercetin treatment prevents renal tubular damage and increased oxidative stress induced by chronic cadmium administration, most probably throughout its antioxidant properties [35].
Also, quercetin reduced abnormal development of mouse embryos produced by hydroxyurea [36]. Liang et al. demonstrated that quercetin (66 mg/kg supplemented diet) significantly improves high fatty saturated induced fetal skeletal maldevelopment, perhaps in part due to antioxidant effects of quercetin in placenta. This speculation is supported by previous reports that demonstrate quercetin prevention of oxidant injury and cell death by ROS scavenging and protection against lipid peroxidation [37].
Abdelmoaty et al. reported that quercetin could prevent hyperglycemia induced by stereptozotosin in rats [38]. In another study, quercetin with dose 50 mg/kg orally was most effective in preventing arsenic poisoning by reducing oxidative stress [39].
In conclusion, the present study showed the effects of quercetin for the first time on teratogenicty induced atRA in rat fetuses. The present results indicate that exposure 25 mg/kg of atRA in 8-10th days of gestation of rat decreases weight and length of embryos and influences on skeletal system. The protective effect of quercetin in atRA-induced teratogenesis in rat may, at least in part, be due to its antioxidant activity, which we believe deserves further investigation.