Introduction
Pompe disease, also known as glycogen storage disorder type II and acid maltase deficiency, is a rare progressive and often fatal neuromuscular disease caused by lysosomal acid alpha-glucosidase enzyme deficiency (GAA) [1, 2]. The disease was first described in 1932 by a Dutch pathologist JC Pompe in a 7-month old infant girl [3]. This disorder is inherited as an autosomal recessive manner and affects females and males equally [4]. The disease is manifested with continuous spectrum of clinical symptoms and based on age and the onset of symptoms is divided into two main groups of infantile-onset and late-onset Pompe disease [5]. The infantile form is characterized with massive deposition of glycogen in the heart, liver and skeletal muscles, which leads to rapidly progressive cardiomyopathy, hepatomegaly, generalized muscle weakness, prominent hypotonia, respiratory insufficiency and motor delay. Death occurs due to the cardiac or respiratory failure before the end of the first year of life [4, 6]. Cardiac hypertrophy is the key feature of this form of disease [7]. The late-onset group includes childhood and adult forms. Pompe disease incidence is estimated within 0.17 to 1.31 per 100,000 people of population based on the diagnosed cases [8]. Incidence of infantile-onset Pompe disease is estimated 1 per 138,000 people based on the frequency of GAA gene mutation carriers [9]. This study was conducted to characterize the demographic, clinical and laboratory characteristics, and the natural course of patients with infantile-onset Pompe disease.
Materials and Methods
This study is a retrospective review of clinical and paraclinical findings of Pompe disease patients admitted to Ali Asghar children's hospital in Zahedan between 2000 and 2012. During this period, 17 patients were registered diagnosed with infantile-onset Pompe disease, two of which were excluded due to the incomplete records. Fifteen patients were diagnosed based on clinical history, physical examination, paraclinical findings, including chest X-ray, electrocardiogram, echocardiogram, and muscle enzymes after ruling out the other diseases. All patients were put under laboratory examinations, including measurement of aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), blood sugar, electrolytes, creatine kinase (CK), Serum and urine amino acid chromatography, as well as cardiac assessment by echocardiography, electrocardiogram and chest radiograph. Due to the lack of availability, GAA enzyme activity measurement and the gene mutation determination for definitive diagnosis were not possible for any patient and the diagnosis was based on the available clinical and paraclinical findings. The data relating to the clinical history, physical examination and diagnostic evaluations were extracted from the patients' medical records. Due to the lack of availability of specific treatment, supportive care measures, including respiratory, cardiac and nutritional supports, were performed on all patients. In this retrospective review, patient’s secrets and details were not disclosed. The data were analyzed using SPSS-16 software. We used descriptive statistic and frequencies for all calculation in this article and data are shown as medians and percentage.
Results
Fifteen patients were diagnosed with Pompe disease between 2000 and 2012. Demographic characteristics of patients are shown in table 1.
| Patient no. | Gender | Age at diagnosis (days) | History of affected sibling | Consanguity of parents | Age at death (months) |
|---|---|---|---|---|---|
| 1 | F | 3 | Pos | Yes | 4 |
| 2 | M | 60 | Pos | Yes | 4 |
| 3 | M | 90 | Pos | Yes | 5.5 |
| 4 | F | 60 | Pos | Yes | 6 |
| 5 | M | 150 | Pos | Yes | 7 |
| 6 | F | 5 | Neg | Neg | 5.5 |
| 7 | M | 120 | Neg | Yes | 8 |
| 8 | M | 150 | Neg | Yes | 7 |
| 9 | F | 115 | Neg | Neg | 8 |
| 10 | M | 135 | Pos | Neg | 6 |
| 11 | F | 75 | Pos | Yes | 5 |
| 12 | M | 45 | Neg | Yes | 4 |
| 13 | M | 105 | Neg | Yes | 6 |
| 14 | F | 3 | Neg | Yes | 7 |
| 15 | M | 50 | Pos | Yes | 6.5 |
Demographic characteristics and outcome of patients
Sixty percent of patients were male and 40% were female. The mean age at the onset of symptoms was 78 days (range: 3-150 days). In 80% of patients, parents were close relatives and more than half the cases (53%) had a similar family history in brothers or sisters, 5 cases had the history of the death of brother and 3 cases had the history of the death of sister due to the similar cause. All patients died below one years of age and the mean age at the time of death was 5.96 months (range: 4-8 months). The clinical findings of patients are shown in table 2.
| Sign / symptom | Frequency (%) |
|---|---|
| Failure to thrive | 13 (87) |
| Pallor | 7 (47) |
| Cyanosis | 6 (40) |
| Respiratory problems | 8 (53) |
| Feeding difficulties | 9 (60) |
| Macroglossia | 13 (87) |
| Hypotonia | 14 (93) |
| Hyporeflexia | 14 (93) |
| Heart murmer | 8 (53) |
| Hepatomegaly | 12 (80) |
Clinical signs and symptoms of patients
| Abnormal finding | Frequency (%) |
|---|---|
| Cardiomegaly | 15(100) |
| Short PR interval | 12(80) |
| Large QRS complex | 15(100) |
| Invert T wave | 8(53) |
| ST depression | 8(53) |
| LV hypertrophy | 15(100) |
| Increased thickness of ventricular walls | 15(100) |
| Interventricular septum hypertrophy | 15 (100) |
Chest X-ray, ECG and echocardiogram characteristic findings
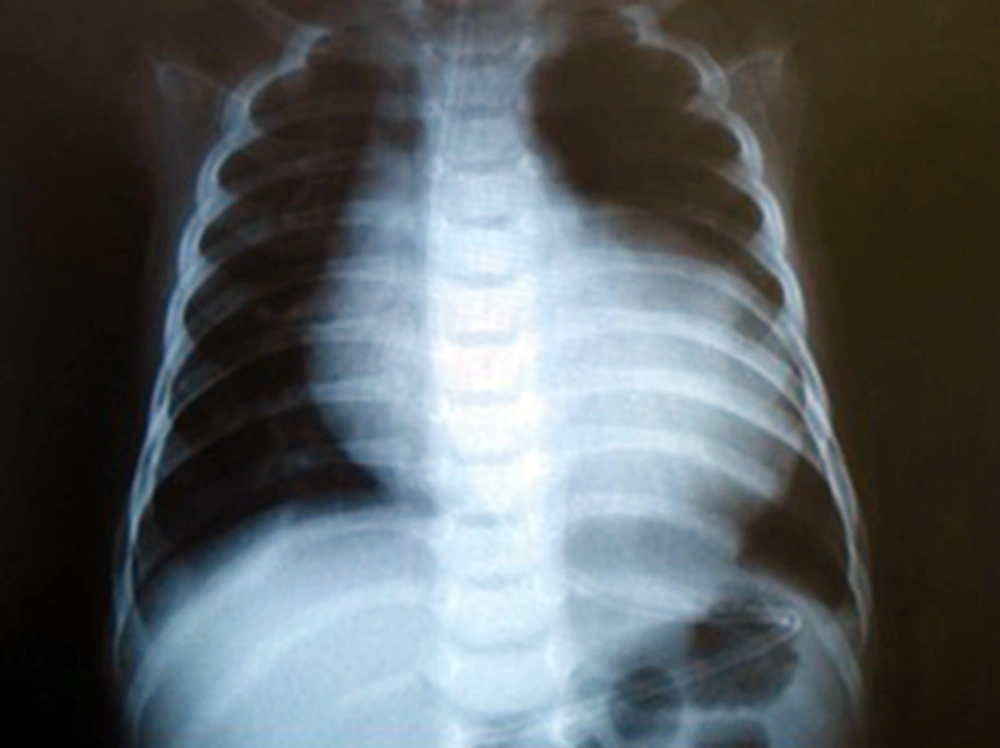
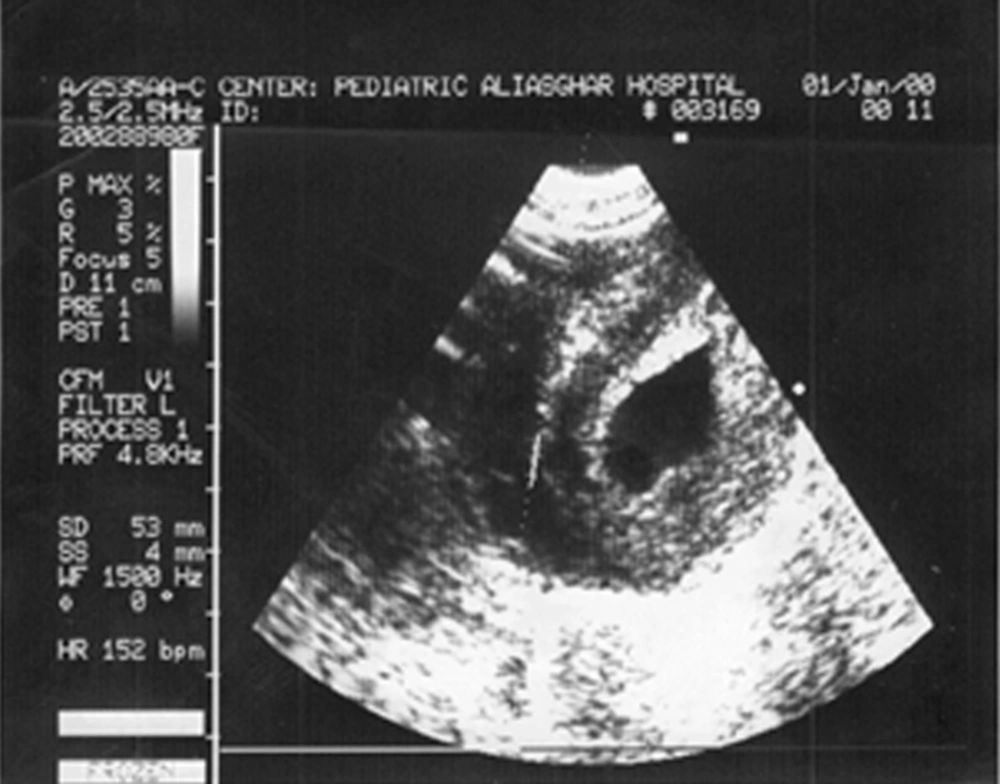
Most frequent signs and symptoms include hypotonicity, decreased deep tendon reflexes, macroglossia, growth failure and hepatomegaly. Cardiac findings of the Pompe disease are shown in table 3 which was the most common finding on chest X-ray and echocardiogram were cardiomegaly and hypertrophic cardiomyopathy, respectively (Fig. 1and 2).
The level of muscle enzymes including AST, ALT, CPK, and LDH was measured and it was been elevated for all patients. The average level of AST was 334 IU (range: 52-678 IU). The average level of ALT was 149 IU (range: 44-310 IU). The median value of LDH was 1661 IU (range: 693-4345 IU). The median value of CPK was 731 IU (range: 225-1700 IU). Serum and urine amino acid chromatography were normal in all patients.
The time interval between the diagnosis and death was 101 days (range: 45-207 days). Muscle biopsy was performed on one case (patient number 5). Also, postmortem heart muscle biopsy was performed on one case (patient number 11) and both cases revealed the increase in glycogen content in the muscle cell lysosome which was shown in the periodic acid Schiff staining as vacuoles with various severity.
Discussion
In our study the male to female ratio was 3:2. The median age at the onset of symptoms and death were 78 days and 5.96 months, respectively. Cardiomegaly, hypotonia, hyporeflexia, macroglossia, failure to thrive, hepatomegaly, and feeding problems were most common findings. All patients died before one year of age. The spectrum of clinical symptoms in Pompe disease is variable depending on the age of patient at presentation. Muscle weakness is a prominent feature in all forms of the disease which usually leads to respiratory failure. Cardiomyopathy is a prominent feature of infantile-onset Pompe disease which is rarely seen in the late-onset disease [4]. Differential diagnosis of infantile-onset Pompe disease including acute form of Werdnig-Hoffman, hypothyroidism, endocardial fibroelastosis, myocarditis, congenital muscular dystrophy, glycogen storage disease type III, Glycogen storage disease type IV (Anderson's disease), Danon disease, idiopathic hypertrophic cardiomyopathy and peroxisomal abnormalities [10].
In addition to the clinical history, a series of tests are helpful to assess cardiomyopathy including chest X-ray, electrocardiogram and echocardiogram, and a series of tests are helpful to assess myopathies such as electromyography (EMG), CPK, AST, ALT, and LDH. Various methods are available to confirm the clinical diagnosed disease such as GAA enzyme activity measurement in skin fibroblasts, lymphocytes, muscle biopsy and dried blood spot [4, 11]. GAA activity measurement at dried blood spot has the sensitivity and specificity of about 100% and is considered a suitable method for screening of newborns [12].
In a study that focused on the natural history of the disease, 20 Dutch patients with infantile-onset Pompe disease and 133 cases reported in the literature were examined. In this study, the median age of patients at the onset of symptoms in both groups was 1.6 months, and the median age at the time of death was 7.7 and 6 months, respectively. Most patients died before their first birthday and only one patient (5%) survived after 12 months [13]. In another retrospective study, the natural history of 168 patients with infantile-onset Pompe disease was investigated. The median age at the onset of symptoms was 2 months and the median age at the time of death was 8.7 months [14]. In two studies, cardiomegaly, cardiomyopathy, hypotonia, respiratory problems, nutrition disorders, and growth failure were the most common signs and symptoms and the natural history of the disease did not significantly change [13, 14]. In our study, the mean age at the onset of symptoms was 78 days and the average age at the time of death was 5.96 months. This relative difference could be due to a lag in the reference and the more advanced stage of the disease at the time of diagnosis. However, clinical symptoms of our patients were similar to the two previous studies, and all of them died before one year of age. In a study Willemsen et al. described three infants with infantile-onset Pompe disease whose clinical symptoms were mostly generalized hypotonia, reduced tendon reflexes, and hypertrophic cardiomyopathy, and all three died before one year of age [15]. The findings of present study were also similar to the findings of this study. In another study, hypotonia was the most common presenting symptom and the most common supportive measure for the patients was cardiac medication [16]. However, cardiomegaly was the most common finding in our study.
In the study of Zhang, 4 out of 17 patients had infantile-onset Pompe disease, whose onset of symptoms was between 2 and 6 months and in 3 cases, it led to death due to respiratory failure [17]. In our study, the age of symptoms onset in all patients was less than 5 months. In a review, 23% of 742 patients with Pompe disease had infantile-onset Pompe disease, and about 15% had no cardiomyopathy [18]. However, 100% of patients in present study had cardiomyopathy. There is no pharmacological treatment for this disease. Enzyme Replacement Therapy (ERT) and gene therapy are being evaluated [4]. The study of Levine et al. claims that 92 to 95% of patients with infantile-onset Pompe disease die in the first year of their life. In their study, the researchers showed cardiovascular system surprisingly rapidly responds to enzyme replacement therapy [19]. In the present study, due to lack of availability, none of the patients were treated with enzyme replacement. The study of Haskins mostly discusses gene therapy for lysosomal storage disease in animal models which had an important role in effectiveness and safety of the treatment [20]. The study of Koeberel et al. claims that gene therapy could be curative in patients with glycogen storage disease types I and II in the future [21].
The study of Sun et al., some evidence was also shown of effectiveness of gene therapy in GSD II mouse models [22]. Given that Pompe disease is a multi-system disorder, management of these patients at present includes multidisciplinary supportive measures and without ERT, all patients with classic form of infantile Pompe will die before the first year of life. Timely diagnosis of the disease and the availability of enzyme and start of therapy before 6 months of age will lead to early response and improves patients’ survival and justifies the recommendation of screening test for early detection of Pompe disease [23-26].
Like the other diseases with autosomal recessive pattern, genetic consultation for individuals with a family history of Pompe disease is of great importance to identify potential carriers. Prenatal diagnosis is also possible by measuring activity in amniotic cells or chorionic villus sampling (CVS) [26]. In sum, it can be concluded that although Pompe disease was diagnosed in our patients based on clinical and paraclinical findings, the prevalence of symptoms and the process of disease had no significant difference with other studies and in general, outcome of this disease will be dismal and disappointing if left without specific treatment. According to availability and possibility of definitive diagnosis of Pompe disease by determining GAA enzyme activity, at present, it is recommend that all patients suspected with Pompe disease should be under investigations for early diagnosis and necessary measures should be taken to provide the availability of ERT for all patients in all parts of the country before the disease progression.