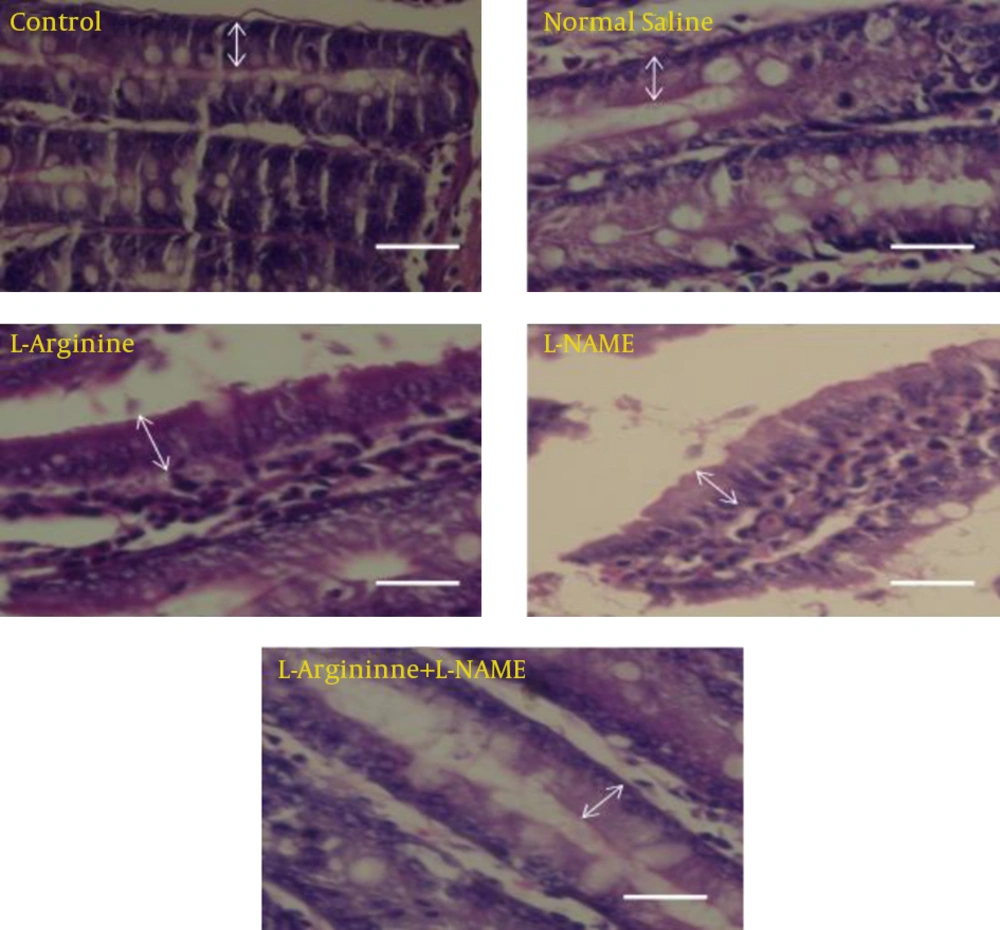
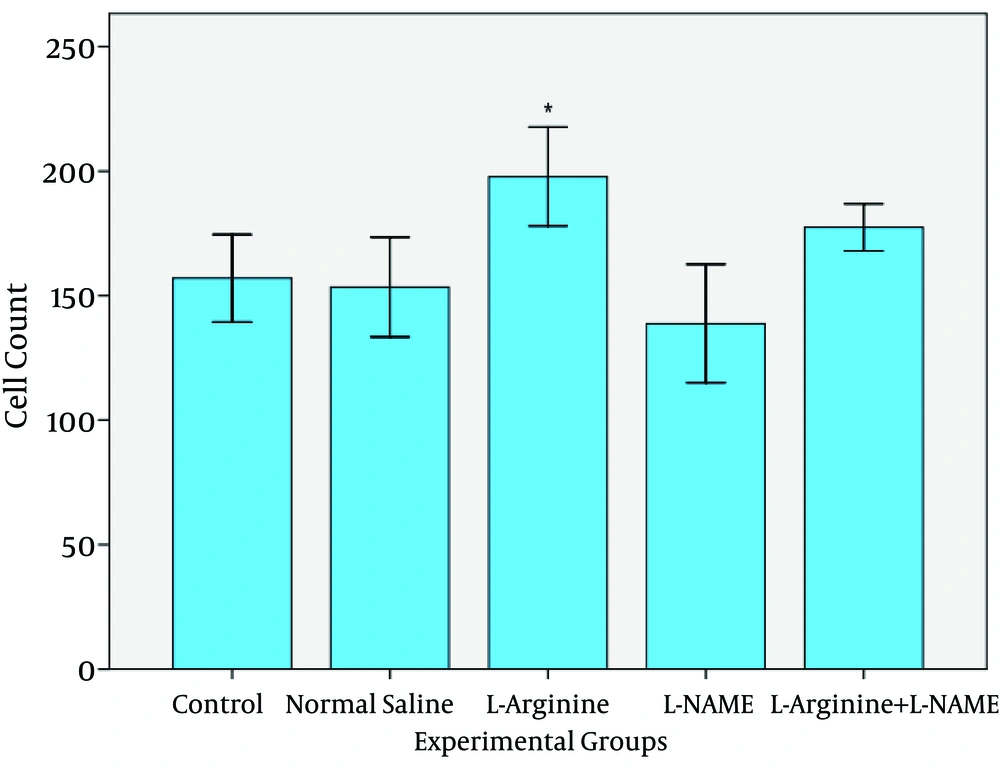
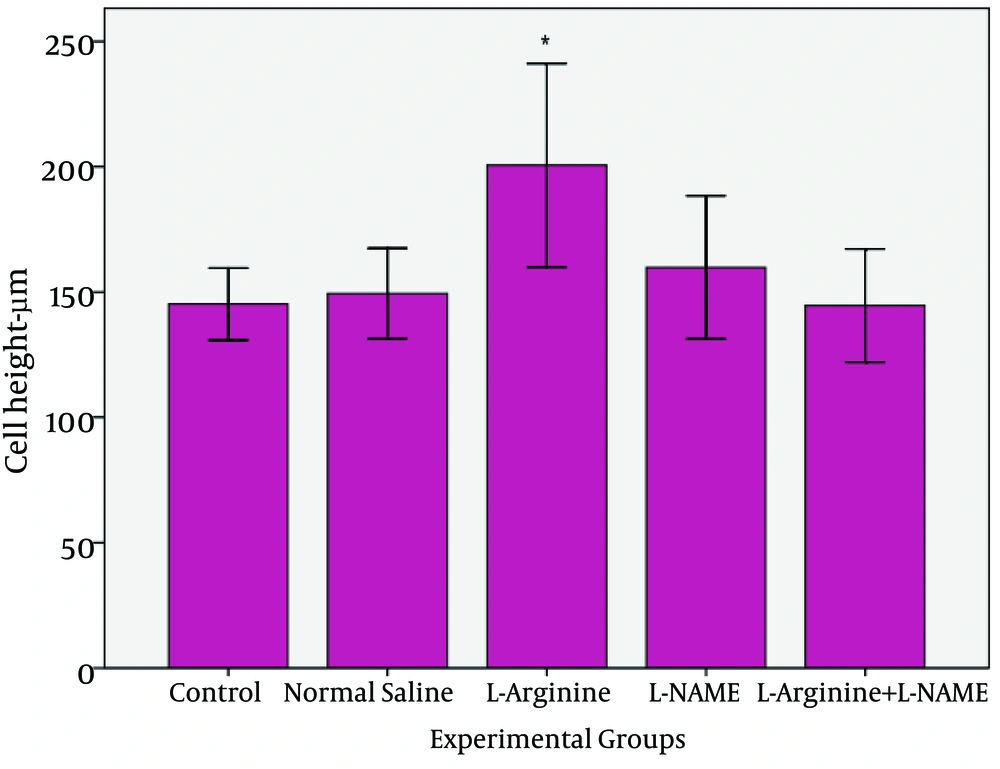
In this study, effect of L-Argentine as a NO precursor and L-NAME as a NO inhibitor on epithelial cells of jejunum was evaluated. The results demonstrated that L-Argentine and L-NAME administration caused increase and decrease of entrecotes number respectively. The height of epithelial cells did not have any significant difference between L-Argentine and L-NAME groups while a significant increase was observed in L-Argentine group in comparison with three other groups.
Effects of NO on enteric blood circulation and its protective role on intestinal mucosa were shown in 1992 [
7]. Then several studies were done and the researchers introduced it as a mediator of intestinal fluid and electrolyte transport [
8,
13]. In recent studies, NO has been introduced as a vascular regulator which increases intestinal blood circulation and stimulates cell proliferation, protein synthesis and cell survival [
6,
14,
15]. It has been demonstrated in many researches that NO is found in gastrointestinal vessels and endothelial myentric nerves. NO removes free oxygen radicals from digestive system. Moreover, it inhibits inflammation and improves mucosal blood flow and permeability which prevents injuries to intestinal mucosa [
16,
17]. NO protects epithelium against damages and also play an important role in epithelial reconstruction [
18]. In a research which was focused on the effect of NO on superior mesenteric artery (SMA) blood flow, the researchers reported that decrease in NO level causes vasoconstriction and defect in intestinal blood circulation which lead to intestinal mucosal atrophy [
19]. Another study represents that hypoxia results in intestinal ischemia because of decrease in SMA blood flow. Hypoxia and ischemia have physiological and pathological effects on integrity of epithelial barrier of intestine. NO improves mucosal permeability and acts as a noradrenergic and noncholinergic mediator to improve vascular tonicity. NO also protect intestine against toxins, and prevent apoptosis and necrosis, leading to survive epithelial cells and decrease mortality [
20].
Studies show that high concentration of NO was effective in progress of colon cancer in mouse and in pathogenesis of celiac disease. On the other hand, it has been reported that L-NAME (NOS inhibitor) induces decrease in cell proliferation of colon. Therefore, researchers suggest that it may be used as a prophylactic factor for colon cancer [
21-
23]. Our results are consistent with the findings of mentioned studies. In the current study, entrecote number of jejunum in L-NAME group was significantly decreased in comparison with L-Argentine group. But there were no significant difference in cell height between L-NAME group and other groups. Moreover, cell number in L-Argentine group was significantly increased. This suggests that NO can be considered as a mitotic factor which plays a role in intestinal cells proliferation [
21]. On the other hand, it has been reported that because of its mutagenic effect, NO can act as a protective factor against gastrointestinal ulcers induced by bacteria, stress and drugs like NSAIDs [
6,
15,
24]. There are inconsistent reports about the effects of L-NAME on intestinal epithelial cells. However, the results of the present study are in contrast with Akgun-dar et al. observations. According their report, L-NAME prescription induced significant increase in the height and proliferation of intestinal epithelial cells [
24].
The present study demonstrated that after NO precursor administration, the number and height of entrecotes increased in comparison with control, normal saline, and L-Arginine+L-NAME groups. It may be attributed to NO synthesis stimulation and the effect of NO on modulation of intestinal blood flow. Whereas L-NAME group had no statistically difference with control and normal saline group. L-NAME ineffectiveness may be related to its dose which could not inhibit any synthesis. In L-Arginine+L-NAME group the results were the same as normal saline and control groups. But there is a question: is the increase in cell number consequence of mitosis enhancement or is due to decrease in apoptosis? On the other hand it has been shown that celiac and malabsorption diseases as well as radiation induce entrecote injury will result in decrease of entrecote height [
9,
25,
26].
In the present study, L-Argentine increased entrecote height. It prevented cell damage which is due to improved jejuna blood flow. While there was not any significant difference in L-Arginine+L-NAME group compared to control and normal saline groups. In conclusion, the results of this experiment demonstrated that NO and its precursor increase number and height of jejuna entrecotes. Further studies are required for investigating the exact mechanism of NO and L-Argentine on the number and height of intestinal epithelial cells. Therefore, besides stereological and morph metrical studies, assessments of apoptosis, evaluation and measurement of serum level of eons and different doses of NO precursor and inhibitor is suggested.