1. Background
Uterine myomas (fibroids) are benign tumors originated from smooth muscle of the uterus. Myomas are typically found in 20% - 40% of females at reproductive age [1]. Given the heterogeneous nature of the disease, many genes are suggested to be involved in its pathogenesis, though much work is required to determine the molecular biology of these tumorigenesis [2]. Many thrombotic processes are activated by tumors, on the other hand, clot formation could protect tumor cells from the immune system in some stage of the tumorigenesis [3]. Because of the dependency of tumor growth to thrombosis, coagulation factors could be considered as a major risk factor in tumorigenesis [3, 4]. Several studies have investigated the role of thrombosis factors in tumors and have shown the importance of thrombosis management in tumor treatment [5, 6]. Although the coincidence of myoma and thrombosis is reported in several types of research [7, 8], published studies on the association between myoma and thrombosis-related mutations are limited. FXIII and FV are two important coagulation factors, which their deficiencies cause severe thrombotic disorders [9, 10].
FXIII is the last zymogen in the coagulation cascade, which consists of two non-covalently related subunits: A subunit is coded by F13A1 gene that is responsible for its catalytic activity and “B” subunit acts as a molecular carrier in plasma [11]. F13A1 gene has been mapped on chromosome 6p24-25, which spans 160 kb of the genome [12]. The protein products of this gene covalently cross-link the fibrin clot to increase resistance to proteolytic insults [13]. Valin (G allele) to Leucine (T allele) substitution at codon 34 in exon 2 of the F13A1 gene is known as a common mutation in this gene [14].
V34L mutation locates adjacent to the thrombin cleavage site of FXIII and results in a faster activation of F XIII by thrombin [15]. Then, this variant increases the resistance to clot formation and could decrease thrombosis risk [14].
Factor V is the other essential coagulation component required for normal clotting cascade. The gene for factor V is localized on chromosome 1q24.2 [16]. Activated thrombosis factor 5 (FV) forms a complex with Factor X, which in turn changes fibrinogen to fibrin. The most prevalent mutation in the F5 gene called FV Leiden (FVL, rs6025), occurred exactly at the cleavage site for activator protein C (APC). Consequently, the FV Leiden is resistant to APC and remains active and increases the rate of thrombin generation [17]. This mutation is associated with G-to-A substitution in exon 10 of F5 gene, which results in Arginine (Arg) to Glutamine (Gln) change at codon 506 (R506Q) [18].
In this study, we aimed to investigate the association of two genetic mutations, rs5985 (V34L) in A subunit of F13A1 gene and rs6025 (FVL) in F5 gene and the risk of myoma in women from the south of Iran.
2. Methods
2.1. Study Population
In total, 73 diagnosed women with uterine myomas with the age range 29 - 65 years (mean ± SD, 50.5 ± 7 years), and 73 healthy women (16 - 51 years, 38.4 ± 7 years) as control subjects were enrolled in this case- control study. Patients subdivided into three categories based on the age. The age range from 21 - 30 years old (12 cases: 16.4%), 31 - 40 years old (27 cases: 37%) and 41 - 50 years old (34 cases: 46.6%). Blood samples were provided by Hazrat-e-Zeynab hospital, Shiraz, Iran. Patients who their uterine myoma confirmed by laparoscopy were signed the informed consent. The inclusion criteria were: a, symptomatic uterine myoma; and b, indication for surgery (hysteroscopy or myomectomy). Exclusion criteria: a, pregnancy; b, breastfeeding; c, hormonal contraception or any hormonal therapy in the last 3 months; d, any genital tumors and e, unexplained genital bleeding. All clinical procedures were approved by the local Ethics Committee of Shiraz Medical School.
2.2. Genotyping
DNA from venous blood was isolated using the phenol-chloroform method and used for genetic analysis [19]. Two mutations, including FVL (rs6025) in F5 gene and rs5985 in F13A1 gene, were genotyped using amplification refractory mutation system-polymerase chain reaction (ARMS-PCR) technique. Two PCR reactions were done for each individual, each with the allele specific primer, common primer and a pair of primer for a gene as an internal control. In the present study, the gene for Hemoglobin gamma 1 (HBG1) was selected to amplify as an internal control. Primers for two variations were designed using gene runner software. Details including the location of each mutation in the respective genes, primer sequences, annealing temperature and product sizes are presented in Tables 1 and 2. The reaction details are as follows: Both mutations were determined by ARMS-PCR using a Bio-Rad t100 thermo cycler with a total volume of 25 µL, containing PCR buffer: 1x, MgCl2: 1.6 mM, Primers: 0.3 pM, dNTPs: 0.8 mM, Taq DNA polymerase: 2U/25µL, DNA: ~200 ng/25 µ. The reaction condition for FVL mutation was: initial denaturation at 94°C for 5 minutes, followed by 30 cycles of denaturation at 94°C for 30 s, annealing at 59°C for 50 seconds and extension at 72°C for 30 seconds, and a final extension step at 72°C for 5 minutes. The amplification condition for The F13A1 V34L polymorphism was: initial denaturation at 94°C for 5 minutes followed by 30 cycles of denaturation at 94°C for 30 seconds, annealing at 57°C for 45 seconds and extension at 72°C for 30 seconds, ended by a final extension at 72°C for 3 minutes. Amplified fragments were separated by 2% agarose gel electrophoresis stained with SYBER gold safe stain and visualized with a Light Transilluminator. DNA molecular weight marker (Fermentas, 50 bp Ladder) was used to assess the size of the PCR products.
| Gene | Mutation | Primer Sequence Temp | Annealing |
|---|---|---|---|
| F5 | FV Leiden, G/A, Exon 10 | Rv common: GGACTACTTGACAATTACTGTTCTCTTG | 59°C |
| Fw (G): GCAGATCCCTGGACAGACG | |||
| Fw (A): GCAGATCCCTGGACAGACA | |||
| F13 | V34L, G/T, Exon 2 | Rv common: GAACCCCAGTGGAGACAGAG | 57°C |
| Fw (G):ACAGTGGAGCTTCAGCGCG | |||
| Fw (T): ACAGTGGAGCTTCAGCGCT |
Mutations in F5 and F13A1 Genes, Their Locations, Primer Sequences, Annealing Temperatures and Product Sizes
| Gene | Primer Sequence | Annealing Temp |
|---|---|---|
| HBG1 | Fw: AACGGCTGACAAAAGAAGTCCTGG | 57°C |
| Rv: TGCCAGGCACAGGGTCCTTCC |
Characteristics of the Internal Control HBG1 Gene Primers
2.3. Statistical Analysis
Quantitative data were expressed as a mean ± standard deviation (SD). Differences categorical variables were presented using frequency counts and compared with a logistic regression test. Deviation from Hardy-Weinberg equilibrium was analyzed by a χ2 test. Odds ratio (OR) and 95% confidence interval (CI) were calculated as an estimate of risk. P < 0.05 was considered statistically significant. All statistical analyses were performed with SPSS 16.0 software.
3. Results
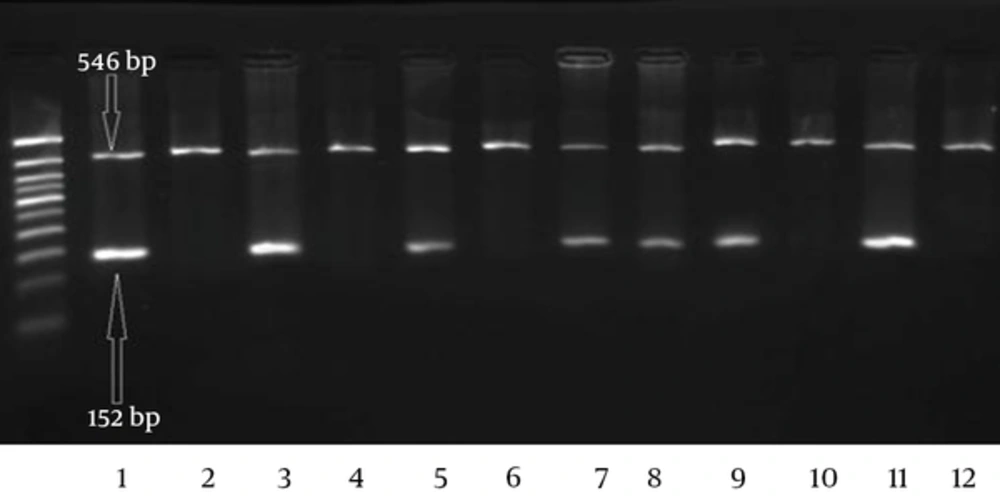
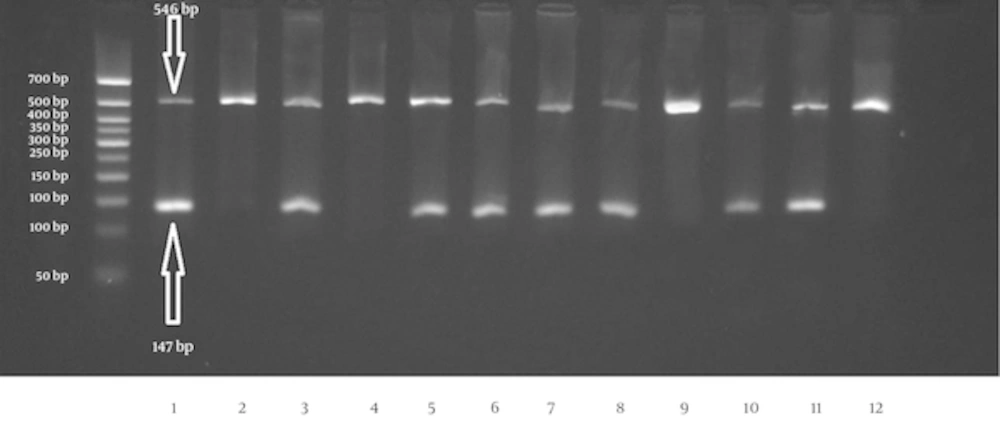
We investigated two mutations, rs5985, and rs6025 in 73 patients with myoma and 73 healthy individuals. The schematic representations of Agarose gel electrophoresis for mutation analysis are shown in Figures 1 and 2. The internal control band was 546 bp in length, but the allele specific products for the FV Leiden and FXIII V34L mutations were 152 bp and 147 bp, respectively.
Agarose gel electrophoresis of the amplification products of FV Leiden mutation in F5 gene by ARMS-PCR. The first of each two well belongs to the normal primer for Arg allele (G allele), and the second is for the mutant allele Gln (A allele). Wells 1/2, 3/4, 5/6, 9/10 and 11/12 show the GG (Arg/Arg) genotype. Wells 7/8 related to heterozygote genotype. The 50 bp size marker is loaded in the first well.
Schematic representation of the agarose gel electrophoresis for the rs5985 polymorphism in F13A1 gene. The first wells of each two well are related to the normal primer for allele Val (G) and the second one is for the mutant primer for allele Leu (T). Wells 1/2, 3/4, 11/12 show the wild type homozygote genotype (GG; Val/Val). The heterozygote genotype is seen in 5/6 and 7/8 wells and the wells 9/10 are related to mutant homozygote individual (TT; Leu/Leu). The first well from the left is the 50 bp size marker.
Our data showed that the risk of myoma tumor and age are in a direct relationship (P < 0.05). Genotyping of the FV rs6025 mutation results in a surprising data in our studied population. Nearly all subjects (except one myoma patient) showed wild-type homozygote genotype (patient: controls; 98.6% vs. 100%, GG genotype). Regarding this homozygosity, no more statistical analysis was done for any association between this mutation and uterine myoma. As the data in Table 3 shows, the frequency of the wild-type Val allele (G allele) of the FXIII V34L mutation is 84% and 92% in cases and controls, respectively. This difference was statistically significant and showed the protective role on the risk of myoma development (P = 0.05).
The possible association between FXIII V34L and myoma risk was estimated by calculating OR and 95% CI (Table 3). The results showed the protective roles for the genotypes with at least one wild-type allele (Val/Val: GG or Val/Leu: GT genotype) compared with homozygous mutant genotype (Leu/Leu: TT) (OR: 0.1, 95% CI: 0.006 - 1.98). After adjusting the genotype for age, no significant association was found between this mutation and myoma risks (OR: 1.2, 95% CI: 0.41 - 3.5, P = 0.7). The FXIII V34L genotype frequencies were in Hardy-Weinberg equilibrium (P > 0.05).
4. Discussion
The results of our study showed that only F13A1 V34L mutation was associated with myoma. The Val allele showed the protective role on myoma among the studied population. Myomas are the most common benign tumors of woman’s uterus with severe symptoms such as bleeding, pain and miscarriage as a consequent, thus, myoma is a significant health issue among women [1, 20]. Age is considered as one of the risk factors for uterine myomas so that most diagnoses were recorded for women in their forties and above. The risk of myoma increases to more than 30% for women in this age range [21]. The results of the present study showed that 46.5 % of the affected women were at the age higher than 40 years of old.
Myomas are the heterogeneous tumors with multifactorial inherent, so more than one specified class of gene expected to contribute to their development [22]. Dehraisi et al. (2015) published the data of their comprehensive investigation on the effect of Glutathione S-transferase (GST) gene polymorphisms (GSTM1, GSTT1 and GSTP1 genes) on the susceptibility to uterine myoma. All members of the GSTs gene family were reported as a risk factor for uterine myomas [23]. The result of the other study by Taghizade Mortezaee et al. (2015) introduced Interleukin-1β gene polymorphisms (IL-1β-511 and IL-1β3954) as a probable risk factor for uterine myoma in Iranian patient women [24]. Salimi et al. (2014) also investigated the association between IL-4 VNTR polymorphism and the risk of developing uterine fibroid in about 100 Iranian women with uterine fibroid. They reported IL-4 PR2/PR2 genotype as an age- related risk factor for fibroid [25]. The results of the two recent meta-analyses confirmed the involvement of CYP1A1 Ile462Val and HSD17B1 rs605059 polymorphisms in the pathobiology of uterine fibroids [26, 27].
Among all, the role of coagulation abnormalities, disturbance of fibrin formation and fibrinolysis, increased in clotting reaction by-products, protein C resistance and high levels of microparticles are the most questionable etiologies in tumor cells [28]. The correlation between thrombosis and tumorigenesis suggesting thrombosis factors as a good candidate for diagnosis and prognosis of tumor formation. Although coincidence between myoma and thrombosis have been reported in many cases [10, 29, 30], to our knowledge, this is the first investigation of the relationship between myoma and thrombosis-related mutations. There is some evidence of the involvement of thrombosis-related mutations in the establishment and progression of other tumors. For instance, the frequency of oral cancer increased in relation to the F13A1 V34L mutation [20]. FV Leiden is known as a causative genetic variation in gastrointestinal tumor and lymphoblastic leukemia [31, 32]. In the present study, no association was found between FV Leiden mutation and the risk of uterine myoma, as our studied population showed homozygosity for this mutation.
The results of our study regarding V34L mutation are consistent with the previous one. Franco et al. (1999) showed the protective effect of the homozygote state for V34L (V/V; GG) mutation against venous thrombosis and myocardial infarction, respectively [33]. the co-existence of V34L and MTHFR is reported in non-Hodgkin lymphoma [34] on the other hand, a study in Netherland showed a 15% reduced the risk of colorectal cancer in the present of G allele in relation to V34L mutation [35].
In conclusion, the result of our study did not show any relationship between the FV Leiden mutation and uterine myoma. In contrast, the results suggest the protective role for the wild-type Val allele of the F13A1 V34L mutation, but it seems the results influenced by age. Further investigation may require to clear the importance of this polymorphism in the diagnosis and prognosis of myoma.