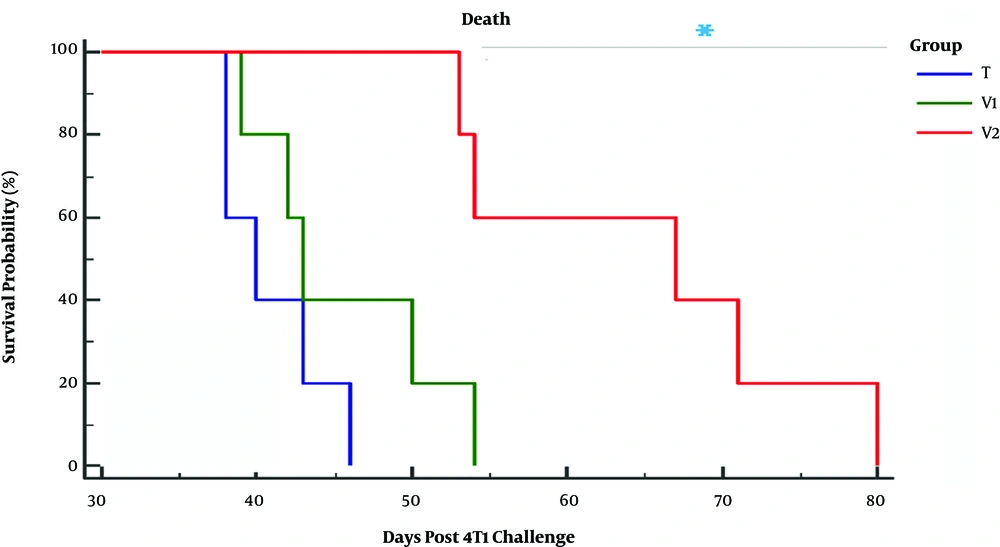
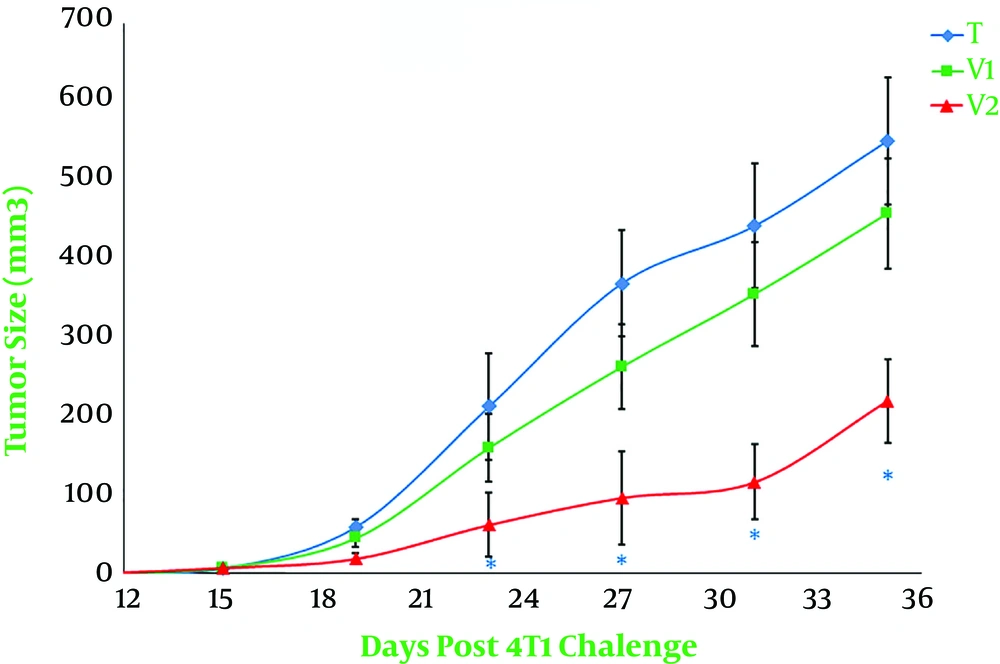
The purpose of the current investigation was to evaluate the efficacy of a combination of the extract of heated 4T1 cells and propranolol for the improvement of murine 4T1 mammary carcinoma in BALB/c mice. It is worth noting that the protocol of immunotherapy should have a good safety margin. Therefore, in the current study, we used propranolol that is frequently prescribed in humans. Despite the useful results, no notable adverse event was observed in this study. Moreover, attained data indicated that the combined immunotherapy with propranolol and the extract of heated 4T1 cells had synergistic effects, causing a more desirable survival curve and slower malignancy growth when compared to other tumor-bearing mice that received only heated 4T1 or PBS. Concurrent with these results, low cost and appropriate safety margin (
19) introduced propranolol as an ideal candidate for combination with tumor antigens. Similar to our results, it has been shown that the tumor lysate of fibrosarcoma and propranolol could reduce tumor growth (
20).
Sympathomimetic agents released from adrenal glands or sympathetic nerves, and interestingly from antigen presenting cells (APCs), could modulate immune responses (
13). It is clear that epinephrine and norepinephrine, especially via the stimulation of β-adrenergic receptors, can limit Th1 immune responses and direct immunity toward the Th2 profile (
21,
22). Therefore, beta-blockers like propranolol may promote immunity towards Th1, an essential anti-tumor arm of immunity. Similar to the current work, several reports also indicated that beta-blockers can potentiate immune responses (
12,
13,
21). Nadolol, a beta-receptor antagonist, can reportedly augment cytotoxic T cells against influenza A virus infection (
23). Antigen-presenting Cells (APCs) have β-adrenergic receptors (
24). Propranolol can block these receptors and promote local inflammation by reducing the local effects of antigen-presenting cells (APC)-, adrenal gland, or sympathetic nerve-secreted catecholamines (
24,
25). The suppression of regulatory T lymphocytes (Tregs) is another possible mechanism to accelerate cell-mediated immunity by propranolol (
20,
26). Treg cells are major sources of IL-10 and TGF-β, two potent immunosuppressive cytokines (
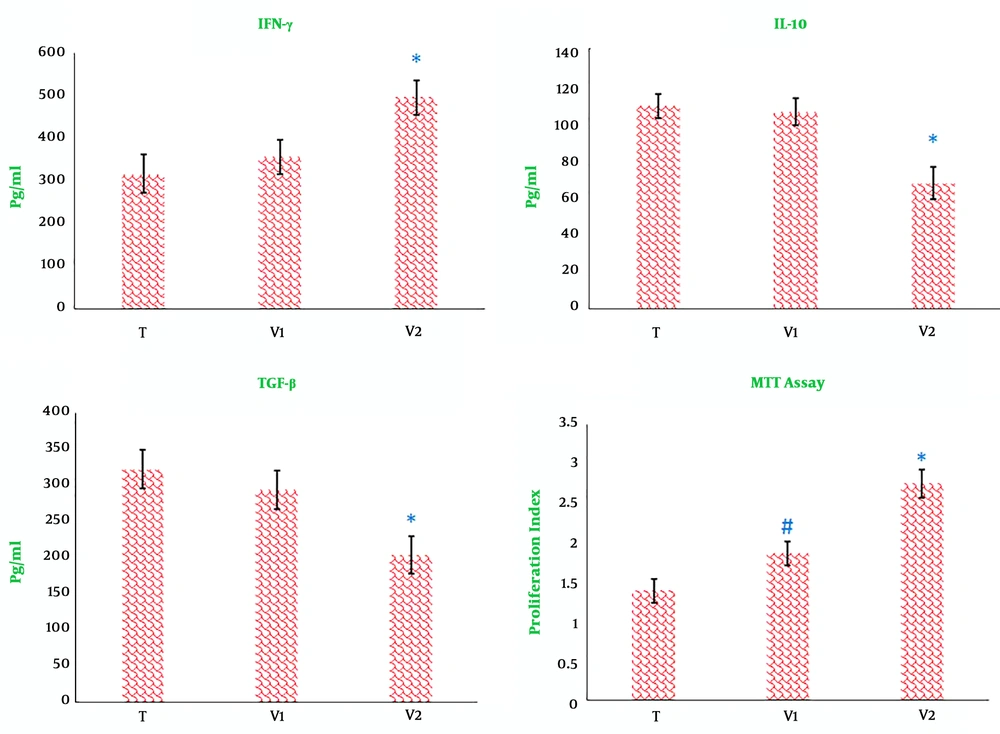
26). Interestingly, our results demonstrated that the combined immunotherapy significantly decreased the production of IL-10 and TGF-β in splenocytes compared to the production of cytokines from splenocytes in other groups. Tumor cells also escape immunity by the secretion of TGF-β and IL-10 (
27). As known, IL-10 and TGF-β can regress the proliferation of lymphocytes and activation of M1 anti-tumor macrophages (
28). The data also revealed that the use of propranolol, as an adjuvant in combination with the extract of heated 4T1 cells, significantly augmented the production of IFN-γ and nitric oxide, as well as respiratory burst. As is clear, IFN-γ possesses potent anti-tumor activity (
14). Nowadays, it is accepted that macrophages have remarkable plastic nature. They may show a reprogramming based on their milieus (
17). Besides, M1 anti-tumor macrophages can produce many cytotoxic factors for tumors like reactive nitrogen and oxygen species (
17). However, cancer can induce anti-tumor macrophages with potency to promote tumor growth via the production of trophic and growth factors with very low production of reactive nitrogen and oxygen species (
27,
28). Considering the published documents, the adrenergic system can impress not only the malignancy progress directly but also the functions of macrophages (
14,
29). As is clear, β2 adrenergic receptor promotes breast cancer progression through the induction of M2 macrophages (
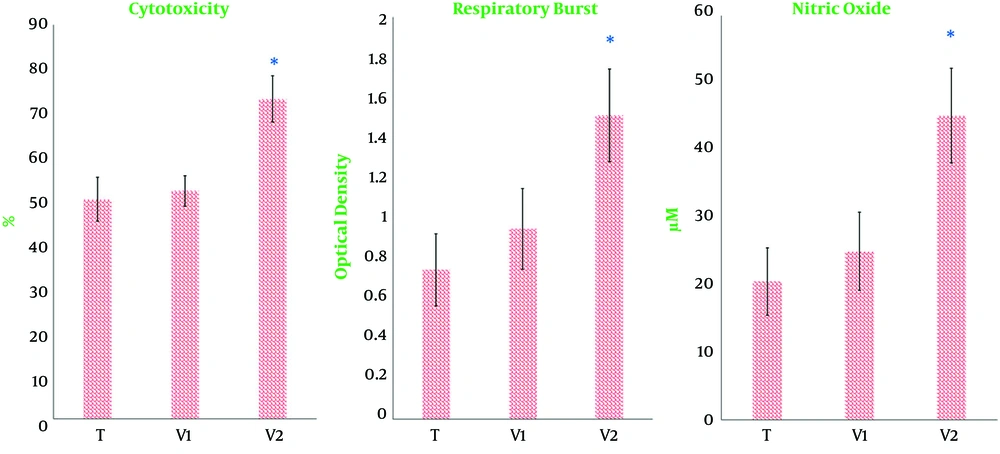
14). The data in this study exhibited that the production of nitric oxide and reactive oxygen species were up-regulated in cancer-bearing mice receiving the combined immunotherapy more than in tumor-bearing mice receiving the extract of heated 4T1. Previously, the regression of nitric oxide production by macrophages via the activation of β adrenergic receptors has been documented (
30).
In addition to macrophages, natural killer (NK) cells are also essential innate cells involved in the elimination of malignant cells. These cells eliminate tumor cells directly by the initiation of apoptosis in tumor cells and the secretion of cytokines like IFN-γ (
31). Lactate Dehydrogenase (LDH) cytotoxicity is a simple and rapid experiment to quantify cytotoxicity by determining LDH activity released from injured cells (
27). Our data suggested that cytotoxicity of NK cells was significantly elevated only in cancer-bearing mice treated with the extract of heated 4T1 cells and propranolol.
In this study, we used the extract of heated 4T1 cells as the source of tumor antigens. The induction of heat shock proteins (HSPs) by heat shock is a logical approach to promote cell-mediated immunity against tumors (
32). The HSPs, especially Hsp70, are molecular chaperone families binding to tumor antigens that potentiate their phagocytosis into antigen APCs (
33). The HSP-antigen complexes can initiate the cross-presentation of antigens by APCs and therefore, promote the generation of cytotoxic T cells against tumor antigens (
33-
35). In this survey, a sublethal heated dose was applied before 4T1 cells underwent the freeze-and-thaw procedure. In this regard, it has been previously reported that a nonlethal heat shock can reduce the growth rate and metastatic potential of 4T1 cells in vivo via Hsp70 induction (
36).
In conclusion, ameliorating the tumor growth rate and better survivability in tumor-bearing mice receiving the extract of heated tumor cells combined with propranolol indicated that this approach is a favored strategy. It can be noted that breast cancer induced by 4T1 cells has a low-immunogenic property (
27,
37). This is only a preliminary study, and further in vivo investigations should be designed to draw a clear conclusion, especially with a larger group of animals.