1. Background
It has been revealed that the property of immune responses can be influenced by various environmental factors [1, 2]. The active form of vitamin D (1α-25(OH)-vitamin D3, calcitriol) is one of the most prominent of the factors that influence the immune system [3, 4]. Multiple evidence over the past few decades has documented the existence of an association between the body level of the vitamin D3 and the prevalence of some autoimmune diseases [3-6]. The major sources of vitamin D3 include dietary intake and manufacture via the UV exposure to precursor 7-dehydrocholesterol in the skin [7]. Interestingly, some subset of dendritic cells and macrophages can produce the active form of vitamin D3 [6]. The receptor of vitamin D3 exists on the cells of the immune system especially in dendritic cells and macrophages and in a low level in resting T cells [8, 9].
Mesenchymal stem (or stromal) cells (MSCs) are fibroblast-like, multipotent progenitor cells that may be isolated and expanded from bone marrow, umbilical cord, fat, gingiva, and other tissues [10-12]. They have the capacity to self-renew and can differentiate into the mesenchymal tissues like bone, cartilage, and fat [12, 13]. MSCs also possess potent immunomodulatory properties and can be a valuable tool for cell-based immunotherapy [14-16]. MSCs in bone marrow and tissue produce a niche that has unavoidable interactions with hematopoietic cells including macrophages [17, 18]. Recent documents indicate that bone marrow-derived MSCs express vitamin D receptors and stimulation of this receptor may play an active role in proliferation and differentiation of MSCs [19-22]. Nevertheless, there is no information about the role of 1α-25(OH)-vitamin D3, as an environmental factor, on the crosstalk between MSCs and macrophages. The aim of the present study was to investigate the effects of the MSCs treated with 1α-25(OH)-vitamin D3 on the some of the macrophages functions.
2. Methods
This experimental study was designed in Urmia University and all the procedures were approved by the local Ethics Committee at veterinary faculty of Urmia University.
2.1. Reagents
Tetradecanoylphorbol acetate (TPA), 1α-25(OH)-vitamin D3, natural red (NR), nitro blue tetrazolium (NBT), dioxin, dimethyl sulfoxide (DMSO), 3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyl tetrazolium bromide (MTT) and phosphate-buffered saline (PBS) were provided from Sigma-Aldrich (St.Louis, MO). Fetal calf serum, Dulbecco’s Modified Eagle Medium (DEMEM) were bought from GIBCO/Life Technologies Inc. (Gaithersburg, MD). May-Grunwald-Giemsa stain was procured from Merck (Darmstadt, Germany).
2.2. Isolation and Culture of MSCs
Bone marrow derived MSCs were obtained on the basis of their ability to adhere to the culture plates as described elsewhere [23]. Briefly, bone marrow cells from deep anesthetized Wistar male rats (8 - 10 weeks old) were collected by flushing the femurs and tibias and were rinsed twice by centrifugation at 1,200 rpm for 5 minutes in Phosphate-buffered saline (PBS). Isolated cells were plated in 75-cm2 tissue-culture flasks with concentrations of 0.3 to 0.4 × 106 cells/cm2 in the DMEM medium supplemented with 15% fetal calf serum. Cells were incubated in a humidified 5% CO2 at 37°C. Four days following primary culture initiation, the culture medium was collected, centrifuged and the cell the pellets were re-suspended in a fresh medium in 75-cm2 flask. These cells were fed twice weekly and upon 70% confluence, the cells were removed by Trypsin/EDTA, counted and passed at 1:3 ratios (about 1.5 × 106 cell/75-cm2 flask). Cell passage was done up to subculture 3.
2.3. Treatment of MSCs with 1α-25(OH)-Vitamin D3 and Supernatants Collection
Subculture 3 of MSCs was incubated with different concentrations of1α-25(OH)-vitamin D3 (0, 50, 100 and 200 nM) for 48 hours. Afterwards, Medium was aspirated and cells were washed three times with PBS. Cells were cultured with serum-free DMEM for 24 hours. Supernatants were collected, centrifuged for 10 minutes at 300 g and filtered through a 0.2-μm filter to remove cellular debris.
2.4. Collection of Macrophages
Resident macrophages were isolated from the peritoneal cavity of Wistar rats by injection of 20 ml of ice cold PBS. The peritoneal fluid was removed and centrifuged at 600 g for 10 min at 4°C. The pellet was washed twice in PBS and suspended in RPMI-1640 medium containing 10% heat inactivated FCS. Cells were counted in Neuber’s chamber and the vitality of the cells was assumed by Trypan blue dye exclusion. Then, 1 mL of live cell suspension (2 × 106 cell/mL) was pre-incubated in 24-well flat-bottomed plates for 40 minutes in a humidified 5% CO2 at 37°C. This process promoted macrophage adherence to the plate. Non-adherent cells were discarded by washing the plate twice with PBS.
2.5. Incubation of Macrophages with Supernatants of MSCs
The bottom chambers of 24-well flat-bottomed plates were filled with supernatants of MSCs. Then, adherent macrophages were isolated by incubation plates on ice for 5 minutes and 2 × 106 viable macrophages were added to each well of plates loaded with supernatants of MSCs and incubated for overnight.
2.6. Evaluation of Macrophage Vitality
Macrophage vitality was evaluated by MTT assay similar to procedure as described earlier [24, 25]. Briefly, after treatment of macrophage with supernatant of MSC, cultures were pulsed with 20 μL of the MTT solution (5 mg/mL) for 4 hours at 37°C. Then 150 mLl DMSO was added and shaken vigorously to dissolve formazan crystal. The optical density (OD) at 550 nm at reference wavelength length 620 nm was measured using microplate reader (Dynatech, Denkendorf, Germany). The experiments were done in triplicate sets.
2.7. Phagocytic Assay by Natural Red
The basic phagocytic ability of macrophage was measured by NR uptake. After macrophages were incubated for overnight with supernatants of MSCs, 100 µL of 0.33% NR solution was added to the wells and plates incubated another 2 hours at 37°C. At the end of the incubation period, the medium was removed and the cells were washed twice with PBS. The internalized NR was solubilized for 30-min incubation by mixing 1 mL 10% acetic acid plus 40% ethanol solution. The optical density was measured at 550 nm.
2.8. Phagocytosis Assay by opsonized Heat-Killed Baker’s Yeast
This test was done as previously described with some modifications [26]. In brief, fallowing stationary incubation of the macrophages with the heat-killed opsonized yeast at 37°C for 1 hour, macrophages were isolated form bottom of microplate by incubation microplate on ice for 5 minutes. The cells were washed, cytocentrifuged onto glass slides, and fixed in methanol. Next, the slides were stained with May-Grunwald-Giemsa staining. Yeast ingestion was assayed by light microscopy under oil immersion. Phagocytosis activities of neutrophils were expressed as percentage of neutrophils which internalized at least one yeast.
2.9. Respiratory Burst of Activated Macrophages
The respiratory test of activated macrophages was evaluated as previously described with some modifications [25, 27]. Macrophages were incubated for 20 minutes with 100 ng/mL TPA and 0.1% NBT. The unused NBT was washed and the reduced dye was extracted in dioxin and measured at 520 nm.
2.10. Yeast Killing Activity
In brief, following 1 hour incubation of the macrophages with the opsonized yeast at ratio 1:10, the medium was discarded and the cells were washed twice. Macrophage was lysed by saponin (20 mg/ mL) and medium was serially diluted and cultured on sabouraud dextrose agar plates. Yeast killing activity was calculated by the formula: 100 × [(colony forming unit (CFU) of control- CFU of test)/CFU of control]. Yeast cultured in the absence of macrophage in 24-well flat-bottomed plates was used as control.
2.11. Statistical Analysis
Data were analyzed using the one-way ANOVA plus Dunnett’s post-hoc test utilizing the SPSS version 20. Data were presented as means ± SEM. P values of less than 0.05 were considered statistically significant.
3. Results
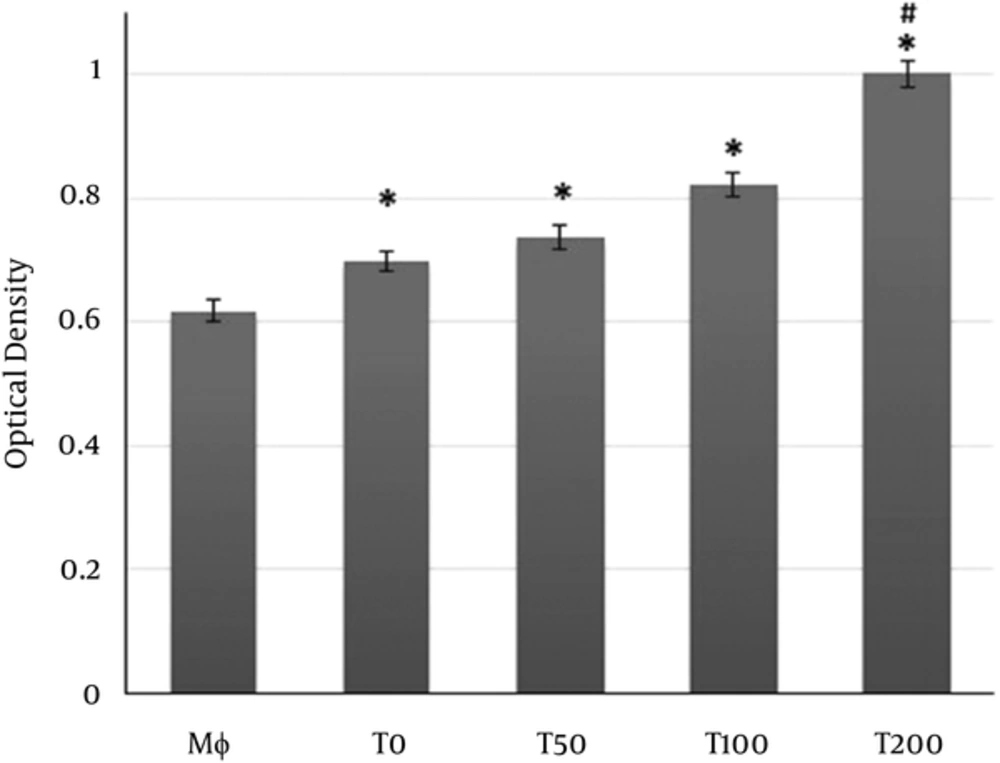
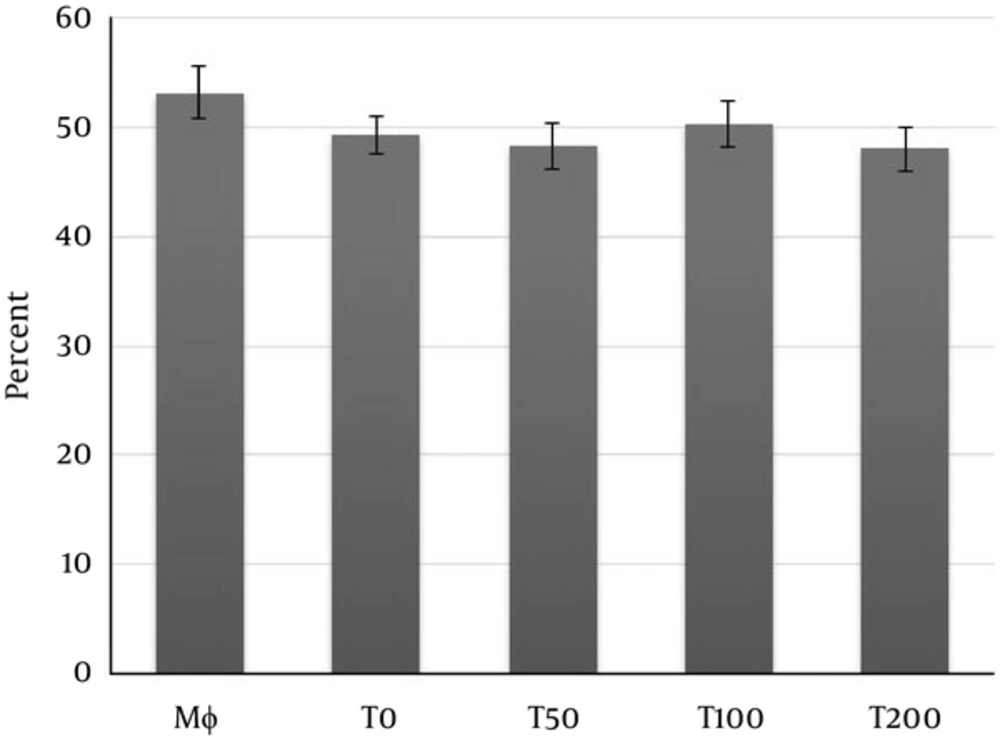
As shown in Figure 1, NR uptake by macrophages was significantly increased in macrophage treated with the supernatants of MSCs treated with vitamin D3 or supernatants of MSCs alone compared to macrophages without treatment. The NR uptake by macrophages treated with supernatants collected from MSCs treated with 100 nM or higher was significantly more pronounced than NR uptake induced by supernatants of MSCs alone (Figure 1).
Macrophages co-cultured with supernatants of MSCs pulsed with vitamin D3 and/or supernatants of MSC alone for overnight. Mφ: Macrophages only; T0: Macrophages + supernatants of MSC alone; T50, T100, T200: Macrophages + supernatants of MSC pulsed with 50, 100 and 200 nM of vitamin D3, respectively. (*P < 0.05 versus Mφ; # P < 0.05 versus Mφ+ MSC (T0)).
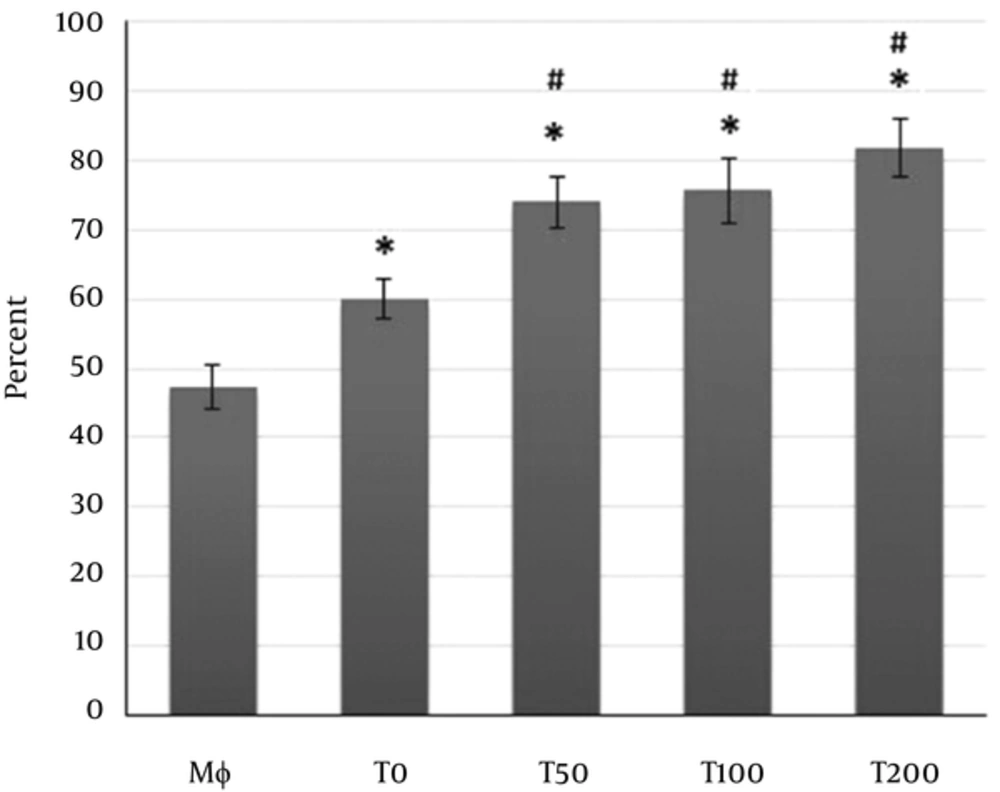
The exposure of macrophages with the supernatants of MSCs pulsed with vitamin D3 or supernatants of MSCs alone caused a significant increase on the phagocytosis of macrophages (Figure 2). Albeit, the increases in phagocytosis induced by supernatants of MSCs pulsed with vitamin D3 was more pounced than the increases in phagocytosis induced by supernatants of MSCs alone (Figure 2).
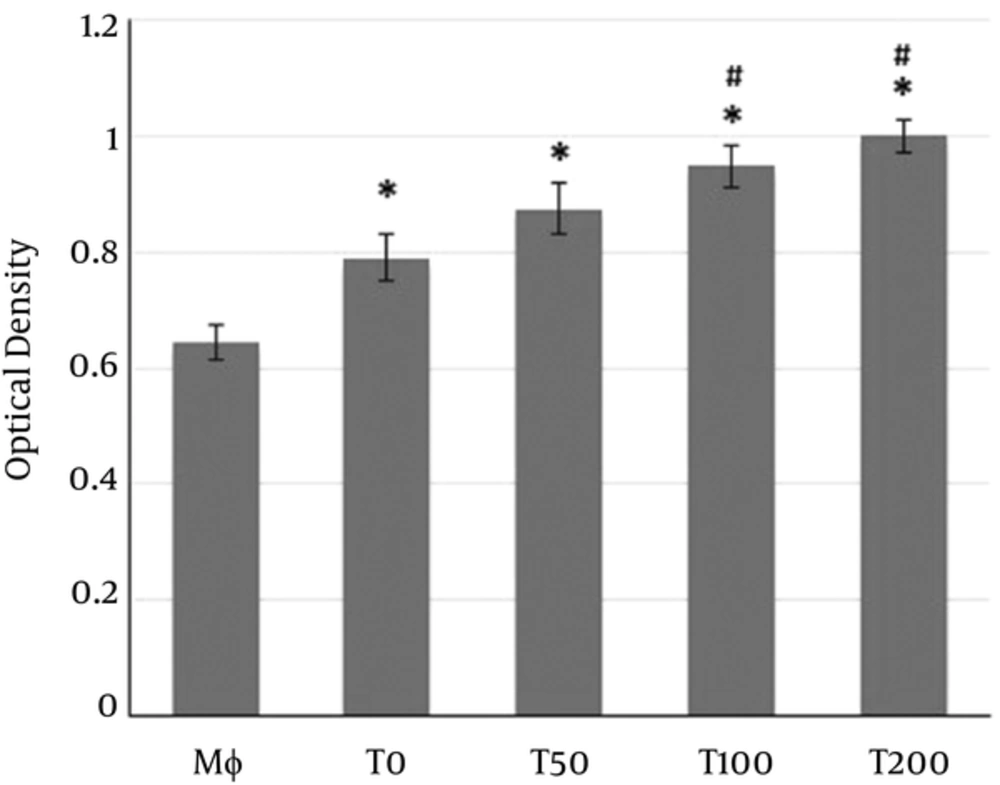
MTT test showed that supernatants of MSCs could significantly increase the vitality of macrophages (Figure 3). Moreover, the supernatants of MSCs pulsed with vitamin D3 significantly enhanced the vitality of co-cultured macrophage more than observed in macrophages co-cultured with supernatants of MSCs alone (Figure 3).
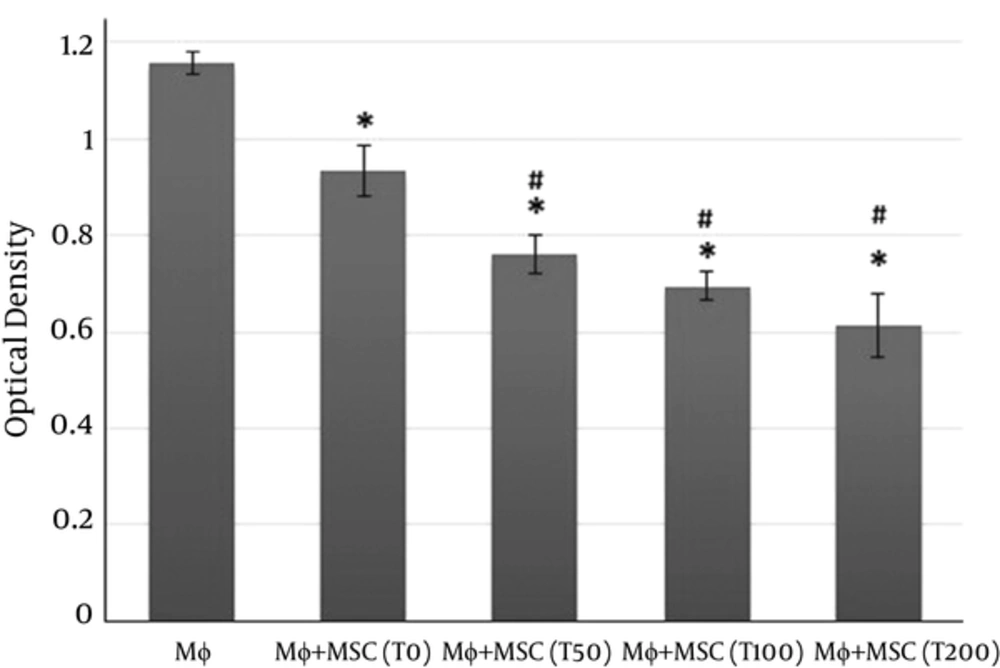
Intracellular generation of reactive oxygen species (ROS) was measured by NBT reduction assay. In addition to microbes, many diverse cytokines and mediators can also activate macrophages. TPA was used in this study to activate macrophages. NBT reduction test showed that the respiratory burst of macrophages was significantly decreased in macrophages co-cultured with the supernatants of MSCs treated with vitamin D3 compared to macrophages without treatment (Figure 4). However, the respiratory burst was markedly reduced in the macrophages co-cultured with the supernatants of MSCs, macrophages without treatment, but this reduction wasn’t as profound as that observed in macrophages co-cultured with the supernatants of MSCs treated with vitamin D3 (Figure 4).
Macrophages co-cultured with supernatants of MSCs pulsed with vitamin D3 and/or supernatants of MSC alone for overnight and stimulated by tetradecanoylphorbol acetate Mφ: Macrophages only; T0: Macrophages + supernatants of MSC alone; T50, T100, T200: Macrophages + supernatants of MSC pulsed with 50, 100 and 200 nM of vitamin D3, respectively. (*P < 0.01versus Mφ; #P < 0.01versus Mφ + MSC(T0)).
As shown in Figure 5 the killing activity of yeast by macrophages could not show any significant difference between groups.
4. Discussion
The results of study showed that the killing activity of macrophages triggered by opsonized yeast didn’t show any significant effect by supernatants of MSCs pulsed with vitamin D3 compared to the supernatants of MSCs alone.
Macrophages exist in almost all tissues and possess an inevitable role in the maintenance of tissue homeostasis and repair, such as removal of cellular debris and clearance of apoptotic cells [28]. Moreover, macrophages are key effectors and modulator cells in the orchestration and expression of innate immunity and instruction of the adaptive immune responses [29, 30]. Macrophages are remarkably diverse and plastic. Based on the environmental factors, they can undergo a reprogramming which leads to the emergence of a spectrum of distinct functional phenotypes [28-30]. Classically-activated macrophages or M1 macrophages have potent anti-microbial and inflammatory properties and may contribute to the pathogenesis of inflammatory disease. Alternatively-activated macrophages or M2 macrophages product less pro-inflammatory cytokines and possess a role in resolution of inflammation via trophic factor secretion and high phagocytic activates [28-30].
It is well known that MSCs modulate the immune responses via multiple mechanisms. Induction of M2 macrophage with anti-inflammatory capacity is one the recent approved mechanisms about the immunomodulatory effects of MSCs [28, 31]. On the other hand, recent works have revealed that vitamin D3 can alter the biology of MSCs. Accordingly, vitamin D3 diminished MSCs proliferation, promoted cell cycle arrest and induced accumulation of MSCs in the G0/G1 phase without inducing apoptosis [21, 22]. These results were associated with a decrease in the GTPase Rho and the atypical Rho family GTPase Rhou/Wrch-1 expression without inducing the Wnt-1 expression and an increase in the expression of surviving [21].
It has been found that MSCs co-cultured with macrophages caused an increase in phagocytic activity of macrophage against E. coli [28]. Higher phagocytic activity is a property of M2 macrophages [28, 31]. The current study similarly showed that the phagocytosis activity of macrophage markedly increased in group cultured with supernatants of bone marrow-derived MSCs compared to supernatants of bone marrow-derived MSCs alone. We have previously shown that the supernatants of MSCs treated with vitamin D3 could significantly up-regulate the phagocytosis of yeast by neutrophils compared to the supernatants of MSCs alone [17]. Moreover, the present results showed that the supernatants of bone marrow-derived MSCs pulsed with vitamin D3 increased the uptake of neutral red and phagocytic activity of opsonized heat inactivated baker’s yeast more pronounced than these effects induced by supernatants of bone marrow-derived MSCs alone. On the other hand, the neutral red uptake by macrophages depends on different factors connected with cell viability, activity, and cell membrane integrity [26]. The gained findings indicated that NR uptake of macrophages was significantly increased in macrophage exposed to the supernatants of MSCs treated with vitamin D3 or supernatants of MSCs alone compared to macrophages without treatment. However, the NR uptake by macrophages or activity of macrophages exposed to supernatants collected from MSCs treated with 100 nM or higher was significantly more pronounced than NR uptake induced by supernatants of MSCs alone.
The MTT assay is a rapid test for assessing cell vitality [17]. MTT assay also showed that treatment of mesenchymal stem cells with vitamin D3 enhanced M2 educated macrophage vitality compared to the control group. Similarly, supernatant of vitamin D3 treated MSCs caused a significant decrease in the percentage of neutrophils apoptosis [17].
Reactive oxygen species (ROS) play an essential role for the elimination of invading microorganisms by phagocytes [32]. Nevertheless, ROS may participate in host tissue injury and immunopathological conditions especially when its production is excessive or inappropriate [33]. It seems that ROS formation can modulated by several stimuli. For example, M2 macrophages educated by MSCs are remarkably anti-inflammatory property and produce a diminished level of ROS [29, 30]. The gained data showed that supernatants of MSCs and/or supernatants of vitamin D3 treated MSCs significantly decreased the respiratory burst of activated macrophages by TPA. Albeit, this reduction was significantly greater in macrophages treated with supernatants of vitamin D3 compared to supernatants of MSCs. Nevertheless, the yeast killing activity macrophages didn’t show any significant change in macrophage challenged with supernatants of MSCs or supernatants of MSCs pulsed with vitamin D3. These results may indicate that MSCs pulsed with vitamin D3, while exerting a protective role from an inappropriate and potentially harmful reaction may potentiate the essential macrophage effector functions after challenge with invader pathogens.
Besides the hygiene hypothesis, decreased outdoor activity, increased pollution and diets containing inadequate vitamin D3 may explain the increased incidence of some immune-mediated diseases over the last 50 years, especially in some geographical area [1]. In fact, former evidence revealed that vitamin D3, possesses direct immunosuppressive effects on immune cells and ameliorates the pathogenesis of T helper 1-mediated autoimmune diseases [6, 34, 35]. Based on the current work, we propose that some of the immunomodulatory and anti-inflammatory effects of vitamin D3 may be due to change in the effects of MSC on macrophages. Of note, supernatant of caffeine exposed MSCs at 100 nM or higher could increase the NR-uptake by macrophages. Therefore, despite decrease in un-specific ROS production, the macrophages are active and forwarded into the alternative activation.
Autologous cell therapy by M2 macrophages may modulate immune responses and could decrease inflammation [29, 30]. In this regard, in vitro education of macrophages towards anti-inflammatory phenotype by conditioned medium of MSCs may possess therapeutic interest because this method produces rapid and sufficient amounts of autologous cells with anti-inflammatory potentials. Interestingly, the observations in this research suggested that vitamin D3 can augments the instruction of anti-inflammatory macrophage by conditioned medium of MSCs.
4.1. Conclusions
Overall, our findings suggest that the supernatants of bone marrow derived MSCs pulsed with vitamin D3 can potentiate anti-inflammatory educated macrophage by MSCs via increasing the vitality and the phagocytosis ability of macrophage and decreasing the potentially harmful production of ROS. Nevertheless, the killing potential of macrophages after challenge with invading pathogen didn’t show any significant change.