1. Background
Liposuction is one of the most commonly performed surgical procedures worldwide. According to the American Society of Plastic Surgeons, this surgery is ranked second, following breast augmentation surgery (1, 2). With the introduction of wetting solutions, liposuction has become much safer. Before liposuction, a wetting solution is injected into the subcutaneous tissue, decreasing the bleeding caused by the early “dry” liposuction procedure (3). Despite its increased safety, the increased rates of the operations have enhanced relevant complications, some of which are rare but serious (2).
Epinephrine is an essential component of liposuction-wetting solutions. It is often used in 1/1000 mL because of its vasoconstrictor effect, which decreases intraoperative bleeding. Moreover, it prolongs the duration of local anesthesia if used with the injectate (4, 5). However, over time, large volumes of wetting solution can be installed with procedure improvement, exposing patients to doses > 5 - 10 mg during large-volume cases (4, 6).
Several studies have attributed the mortality of liposuction procedures to cardiac dysrhythmias, myocardial infarction, fluid overload, pulmonary edema, and fatal asystole. However, no clear etiology has been determined (7-9).
Although epinephrine toxicity could be incriminated in most of such cardiac side effects, this is difficult to prove on the clinical aspect for many reasons. First, epinephrine has a very short half-life (2 minutes). Second, post-mortem metabolism can also occur in humans. Moreover, the patient usually receives exogenous epinephrine during resuscitation attempts (9).
Magnesium has a documented beneficial effect on the cardiovascular system and can stabilize membrane potential, activate antioxidant pathways, and enhance mitochondrial function (10). Moreover, its deficiency has been incriminated in multiple cardiovascular pathologies, including cardiomyopathy, atherosclerosis, hypertension, and dyslipidemia (11, 12).
2. Objectives
We hypothesized that adding MgSO4 during the liposuction procedure could counteract the cardiac epinephrine-related side effects. Few studies have addressed this issue; hence, we conducted this study to evaluate the effectiveness of intravenous (IV) versus wetting solution MgSO4 in counteracting epinephrine-induced cardiac adverse events in patients undergoing abdominal liposuction procedures.
3. Methods
This randomized controlled single-blind study included 129 patients aged 20 - 60 years, with body mass index (BMI) of 25 - 35 kg/m2, and American Society of Anesthesiologists (ASA) class I or II, who underwent abdominal liposuction. The study was conducted in the Plastic Surgery Department, Tanta University Hospitals, Egypt, from March 2021 to March 2022 after gaining approval from the Ethics Committee of the Tanta University (code: 35029/11/21) and then registration in the Pan African Clinical Trial Registry (code: PACTR202105734020705). Informed written consent was obtained from all participants’ guardians.
Exclusion criteria were hepatic or renal insufficiency, known allergy to the study medications, and non-abdominal liposuction.
3.1. Randomization and Blindness
The participants were randomly assigned into 3 equal groups. Randomization was done using the sealed envelope method by a nurse not participating in the study. The research groups were as follows: Group I (control group) was only subjected to the wetting solution along with wetting fluid with 1 mL 1/1000 epinephrine added to 1000 mL of normal saline with no further adjuvants; group II was subjected to the IV administration of MgSO4 (40 mg/kg) for 1 minute at the same time of wetting solution injection, and group III was subjected to MgSO4 (40 mg/kg) added to the wetting solution (the total dose was divided equally according to the expected number of saline bottles injected).
Before surgery, all cases were subjected to full history recording, clinical examination, and routine laboratory investigations. All operations were performed under general anesthesia (GA) using a wide bone IV cannula after establishing routine hemodynamic monitoring (pulse oximetry, blood pressure, ECG, capnogram, and temperature probe). Moreover, GA was induced by fentanyl 1 μg/kg, propofol 2 mg/kg, and atracurium 0.5 mg/kg. The maintenance of anesthesia was achieved by isoflurane (1 ‑ 1.5%) in an oxygen and air mixture, along with cisatracurium (0.03 mg/kg) (MV > ETCO2 35 - 90).
Following anesthesia and skin sterilization, wetting fluid was injected. For all the included cases, 1 mL 1/1000 epinephrine was added to 1000 mL of normal saline. The tumescent technique was used for all cases, as the solution was administered until the targeted tissues (fatty deposits) were sufficiently filled or engorged. The dose of MgSO4 injected was set based on the previous study (13).
Mean arterial blood pressure (MAP) and heart rate (HR) were recorded at the baseline, induction, installation of wetting solution, and then every other 15 minutes by the end of the procedure. Intraoperative cardiac adverse effects, including sinus tachycardia, premature ventricular contractions, and cardiac arrest, were recorded. Total isoflurane consumption was measured using MVCGE after surgery, and all patients received IV paracetamol (1 mg/8 hours) in addition to ketorolac (30 mg per 12 hours). Postoperative pain was assessed by the visual analogue scale (VAS) at PACU and then 1, 3, 6, 12, and 24 hours after surgery. If the patient reported VAS > 3, IV morphine (2 mg) was administered.
Our primary outcome was the mean HR following the installation of the wetting solution during liposuction. In contrast, the secondary outcomes were intraoperative isoflurane consumption, postoperative pain, morphine consumption, and the incidence of cardiac side effects.
Sinus tachyarrhythmia is a heart rhythm with a ventricular rate of > 100 beats/min. Tachyarrhythmias are broadly categorized as narrow complex tachycardia (NCT; < 120 ms) or wide complex tachycardia (≥ 120 ms) (14). PVCS is a premature ventricular contraction, expected when the heartbeat is initiated by Purkinje fibers in the ventricles rather than by the sinoatrial node (15).
3.2. Sample Size Estimation
The sample size was calculated using G*Power 3.1.9.2 (Universitat Kiel, Germany). A pilot study (10 cases per group) was conducted. It was revealed that the mean ± standard deviation (SD) of HR following the installation of the wetting solution during liposuction (i.e., primary outcome) was 92.02 ± 12.73 in the epinephrine group, 80.88 ± 15.20 in the IV MG group, and 86.71 ± 18.76 in the SC MG group. The sample size was determined considering the following points: Effect size = 0.29, 95% confidence limit, and 80% study power. Four cases were added to each group to eliminate the dropout effect; hence, 43 patients were assigned to each group.
3.3. Statistical Analysis
Statistical analysis was performed with SPSS software version 26 (IBM Inc., Chicago, IL, USA). Shapiro-Wilks normality test and histograms were used to test the distribution of quantitative variables. Parametric variables were expressed as mean and SD and compared using ANOVA. Categorial variables were expressed as frequency and percentage and then statistically analyzed using chi-square tests. A two-tailed P-value ≤ 0.05 was considered the significance level in this study.
4. Results
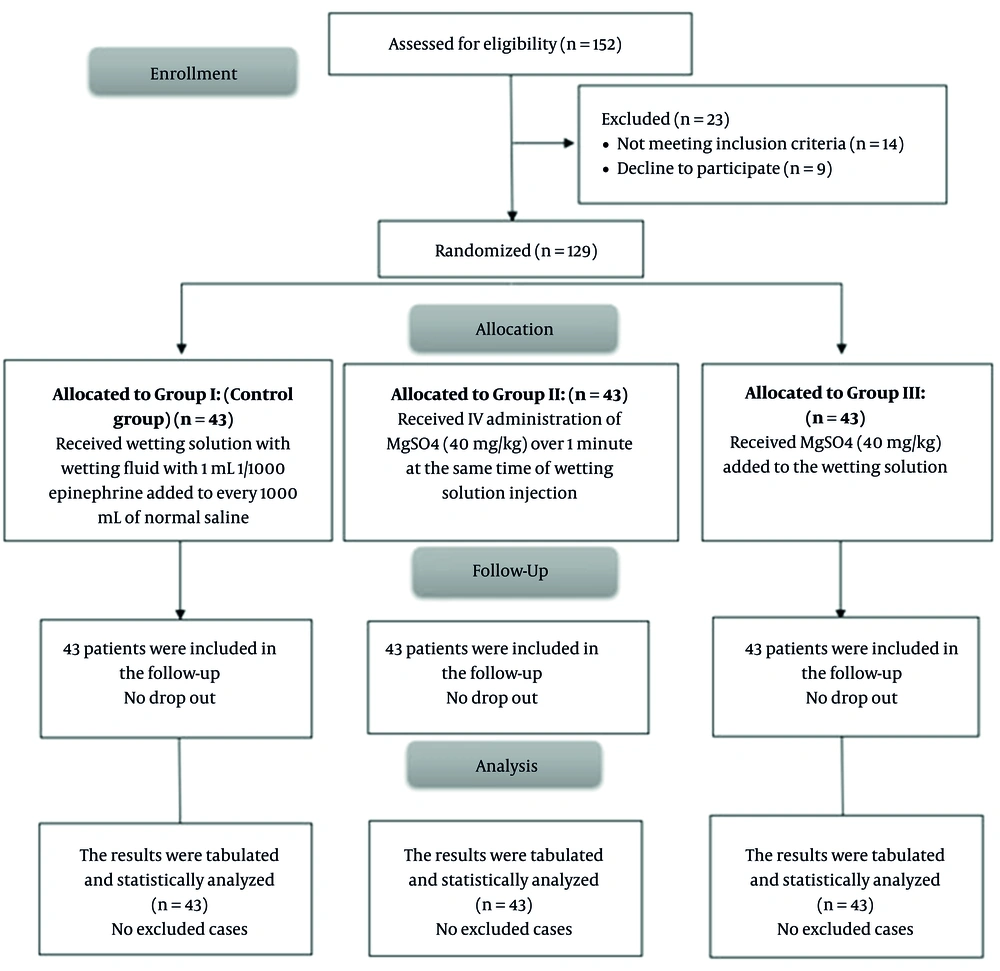
In this study, 152 patients were assessed for eligibility, among whom 14 did not meet the inclusion criteria, and 9 refused to participate. Accordingly, 129 patients were randomly assigned into 3 equal groups (n = 43 per group) and then followed up and analyzed statistically (Figure 1).
The patients’ demographics and duration of surgery showed no significant difference among the three groups (P > 0.05) (Table 1).
| Variables | Group I (n = 43) | Group II (n = 43) | Group III (n = 43) | P |
|---|---|---|---|---|
| Age (y) | 35.16 ± 8.748 | 37.33 ± 10.200 | 36.84 ± 8.174 | 0.513 |
| Gender | 0.215 | |||
| Male | 18 (41.9) | 11% (25.6) | 12% (27.9) | |
| Female | 25 (58.1) | 32% (74.4) | 31% (72.1) | |
| BMI (kg/m2) | 31.59 ± 2.013 | 31.85 ± 1.798 | 31.91 ± 1.983 | 0.724 |
| ASA physical status | 0.376 | |||
| I | 20 (46.5) | 15% (34.9) | 21% (48.8) | |
| II | 23 (53.5) | 28% (65.1) | 22% (51.2) | |
| Duration of surgery (h) | 146.70 ± 18.55 | 150.62 ± 16.97 | 153.37 ± 19.68 | 0.247 |
Abbreviations: BMI, body mass index; ASA, American Society of Anesthesiologists.
a Data presented as mean ± SD or No. (%).
Basal and induction HRs were not significantly different among the three studied groups, and intraoperative HR was not significantly different regarding all measurements between the MgSO4 groups.
Intraoperative HR was significantly lower in group II compared to group I from wetting to 120 min, and it was significantly lower in group III compared to group I at wetting, 30, 45, and 105 min; however, there was no significant difference between group III and group I regarding these measurements (Table 2).
| Heart rate (bpm) | Group I (n = 43) | Group II (n = 43) | Group III (n = 43) | P | P1 b | P2 c | P3 d |
|---|---|---|---|---|---|---|---|
| Basal | 80.49 ± 9.468 | 80.81 ± 11.076 | 79.00 ± 13.765 | 0.741 | --- | --- | --- |
| Induction | 85.95 ± 9.388 | 86.21 ± 12.007 | 83.93 ± 15.363 | 0.652 | --- | --- | --- |
| Wetting | 98.02 ± 12.805 | 87.49 ± 13.754 | 89.86 ± 15.824 | 0.002 | 0.002 | 0.026 | 1 |
| 15 min | 100.12 ± 12.004 | 88.91 ± 13.151 | 95.02 ± 18.089 | 0.002 | 0.002 | 0.329 | 0.165 |
| 30 min | 97.60 ± 12.600 | 85.95 ± 12.928 | 89.09 ± 20.892 | 0.003 | 0.003 | 0.044 | 1 |
| 45 min | 92.67 ± 11.704 | 83.00 ± 12.786 | 85.51 ± 15.182 | 0.003 | 0.003 | 0.041 | 1 |
| 60 min | 88.70 ± 11.281 | 79.21 ± 12.779 | 83.37 ± 16.311 | 0.007 | 0.005 | 0.217 | 0.482 |
| 75 min | 88.65 ± 11.080 | 78.98 ± 12.540 | 82.60 ± 20.096 | 0.013 | 0.011 | 0.197 | 0.814 |
| 90 min | 88.70 ± 12.474 | 79.28 ± 12.374 | 81.12 ± 18.931 | 0.010 | 0.012 | 0.060 | 1 |
| 105 min | 88.70 ± 12.260 | 79.37 ± 12.458 | 80.98 ± 17.805 | 0.007 | 0.010 | 0.043 | 1 |
| 120 min | 88.81 ± 11.576 | 79.56 ± 12.655 | 83.40 ± 18.564 | 0.015 | 0.012 | 0.263 | 0.677 |
a Data presented as mean ± SD.
b Group I vs. group II
c Group I vs. group III
d Group II vs. group III
At baseline and induction, MAPs were insignificant among the three studied groups. However, MAP was significantly lower in the MgSO4 groups (groups II and III) compared to group I, and it was significantly lower in group II compared to group III (Table 3).
| MAP (mmHg) | Group I (n = 43) | Group II (n = 43) | Group III (n = 43) | P | P1 b | P2 c | P3 d |
|---|---|---|---|---|---|---|---|
| Basal | 97.33 ± 9.286 | 96.63 ± 6.786 | 94.88 ± 9.027 | 0.388 | --- | --- | --- |
| Induction | 103.53 ± 9.876 | 100.30 ± 7.418 | 99.60 ± 11.681 | 0.145 | --- | --- | --- |
| Wetting | 112.70 ± 13.534 | 94.02 ± 9.595 | 100.58 ± 13.548 | < 0.001 | < 0.001 | < 0.001 | 0.035 |
| 15 min | 114.23 ± 13.325 | 88.60 ± 9.725 | 100.49 ± 13.188 | < 0.001 | < 0.001 | < 0.001 | < 0.001 |
| 30 min | 107.23 ± 14.410 | 80.93 ± 10.366 | 97.93 ± 12.747 | < 0.001 | < 0.001 | 0.003 | < 0.001 |
| 45 min | 99.72 ± 14.413 | 75.40 ± 10.347 | 90.40 ± 13.968 | < 0.001 | < 0.001 | 0.004 | < 0.001 |
| 60 min | 94.40 ± 14.721 | 71.95 ± 10.021 | 86.30 ± 13.562 | < 0.001 | < 0.001 | 0.013 | < 0.001 |
| 75 min | 95.26 ± 14.206 | 71.86 ± 10.218 | 85.21 ± 13.731 | < 0.001 | < 0.001 | 0.001 | < 0.001 |
| 90 min | 95.93 ± 15.890 | 72.09 ± 10.768 | 85.95 ± 13.856 | < 0.001 | < 0.001 | 0.003 | < 0.001 |
| 105 min | 95.72 ± 15.443 | 72.14 ± 10.255 | 86.07 ± 14.757 | < 0.001 | < 0.001 | 0.004 | < 0.001 |
| 120 min | 95.05 ± 14.705 | 72.42 ± 10.658 | 87.77 ± 13.082 | < 0.001 | < 0.001 | 0.030 | < 0.001 |
Abbreviation: MAP, mean arterial blood pressure.
a Data presented as mean ± SD.
b Group I vs. group II
c Group I vs. group III
d Group II vs. group III
Intraoperative isoflurane consumption was significantly lower in the MgSO4 groups (groups II and III) compared to group I, with no significant difference between the MgSO4 groups (P < 0.001). Postoperative morphine consumption revealed no significant difference among the research groups.
Regarding epinephrine-related side effects, sinus tachycardia occurred in 51.2% of the patients in group I, 25.6% in group II, and 27.9% in group III which was significantly lower in group II and in group III compared to group I (P = 0.014 and 0.027 respectively) and insignificantly different between group II and group III. PVCs occurred in 11.6% of the patients in group I, 0.0% of the patients in group II, and no patients in group III. PVCs were significantly lower in the MgSO4 groups compared to group I (Table 4).
| Variables | Group I (n = 43) | Group II (n = 43) | Group III (n = 43) | P | P1 b | P2 c | P3 d |
|---|---|---|---|---|---|---|---|
| Intraoperative isoflurane consumption (mL) | 100.23 ± 24.832 | 83.49 ± 19.009 | 76.05 ± 15.452 | < 0.001 | 0.001 | < 0.001 | 0.240 |
| Postoperative morphine consumption (mg) | 2.23 ± 1.324 | 1.79 ± 0.965 | 1.77 ± 1.130 | 0.111 | |||
| Arrhythmia | |||||||
| Sinus tachycardia | 22 (51.2) | 11 (25.6) | 12 (27.9) | 0.023 | 0.014 | 0.027 | 0.807 |
| PVCs | 5 (11.6) | 0 (0.0) | 0 (0.0) | 0.005 | 0.039 | 0.021 | --- |
Abbreviation: PVCs, premature ventricular contractions.
a Data presented as mean ± SD or No. (%).
b Group I vs. group II
c Group I vs. group III
d Group II vs. group III
The VAS scores were significantly lower in the MgSO4 groups than in group I. Nevertheless, the three groups showed statistically significant VAS scores after 12 and 24 hours (Table 5).
| VAS | Group I (n = 43) | Group II (n = 43) | Group III (n = 43) | P | P1 b | P2 c | P3 d |
|---|---|---|---|---|---|---|---|
| PACU | 4.60 ± 1.198 | 2.84 ± 1.132 | 3.35 ± 1.412 | < 0.001 | < 0.001 | < 0.001 | 0.182 |
| 1 hr | 4.12 ± 1.401 | 2.95 ± 1.253 | 3.02 ± 1.389 | < 0.001 | < 0.001 | 0.001 | 1 |
| 3 hr | 2.91 ± 0.895 | 2.60 ± 0.877 | 2.26 ± 0.875 | 0.004 | 0.344 | 0.003 | 0.207 |
| 6 hr | 2.88 ± 1.418 | 2.21 ± 0.965 | 2.44 ± 1.098 | 0.029 | 0.026 | 0.251 | 1 |
| 12 hr | 2.58 ± 1.006 | 2.16 ± 1.022 | 2.35 ± 0.948 | 0.151 | |||
| 24 hr | 2.16 ± 1.045 | 2.12 ± 0.851 | 1.91 ± 0.996 | 0.428 |
Abbreviations: VAS, visual analogue scale; PACU, post anesthesia care unit.
a Data presented as mean ± SD.
b Group I vs. group II
c Group I vs. group III
d Group II vs. group III
5. Discussion
The wetting solution injected during the liposuction procedure should inform anesthesiologists of some important physiological considerations (3, 16). Most injected fluids are transferred into the general circulation; however, only 22 - 29% of the fluids are recovered during the liposuction procedure (17, 18). This implies that about 2/3 of the injectate with its added drugs (including epinephrine) transfers to the intravascular compartment (19). A previous study confirmed that 70% of injected epinephrine is reabsorbed into the systemic circulation, with a maximum peak after 5 hours (20).
Some studies have reported epinephrine toxicity following the accidental subcutaneous infiltration of 3 mg 1: 1000 (21-23). Toxicity may be associated with hypertension, tachycardia, arrhythmia, chest pain, diaphoresis, and limb paresthesia, leading to pulmonary edema, asystole, and mortality (9).
Previous studies dealing with liposuction-induced complications have stated that cardiac side effects requiring therapy were observed in < 1% of the cases undergoing surgery (24, 25). Although their prevalence is not high, they may be fatal. Moreover, previous studies are not conducted recently and used wetting solutions with lower doses of epinephrine.
Few studies have discussed preventing such cardiac complications, and there is no rich literature on this issue.
Honarmand et al. (13) reported that MgSO4 in doses < 50 mg/kg could effectively reduce the cardiovascular instability associated with intubation. They tested 3 different doses of this drug (30, 40, and 50 mg/kg), according to which we decided to use the intermediate dose proposed by these researchers in the present study.
In this study, although basal and induction HR were not significantly different among the research groups, intraoperative HR was significantly lower in the MgSO4 groups compared to group I in most intraoperative readings. In the same context, Elsharnouby and Elsharnouby reported a significant decline in intraoperative HR readings following the administration of IV MgSO4 (26). Stanbury reported that MgSO4 prolongs nodal recovery time using both direct and indirect mechanisms (27). It also slows HR at rest by blocking the nictitating membrane of the sympathetic ganglia (13). Furthermore, it has a negative chronotropic effect following the administration of atropine (28).
According to the findings, intraoperative MAP was significantly lower in MgSO4 readings compared to group I. As mentioned in previous reports, MgSO4 does not affect cardiac output; hence, its effect on blood pressure is mediated by decreasing the peripheral arterial resistance using its vasodilator action (29, 30). This finding has been documented in vitro (31, 32), animal (33), and human studies (34, 35).
Our findings showed a significant decrease in isoflurane consumption following the MgSO4 administration compared to the controls. Likewise, another study reported lower doses of volatile anesthetic requirement following the administration of MgSO4 (26). Moreover, James reported that MgSO4 was used to produce general anesthesia and enhance the action of anesthetic agents (36). This effect could be explained by the antagonistic effect of MgSO4 on N-methyl-d-aspartate (NMDA) receptors, decreased catecholamine production by decreasing sympathetic stimulation, and decreased nociceptor sensitization (37). Moreover, it decreases acetylcholine release at the neuromuscular junction (38, 39).
In the present study, the incidence of cardiac arrhythmia showed a significant decrease in MgSO4 compared to group I. Sinus tachycardia was detected in 51.2, 25.6, and 27.9% of the cases. At the same time, PVCs were noticed in 11.6, 0, and 0 of the participants in groups I, II, and III, respectively. Magnesium sulfate is often used as an anti-arrhythmic agent by many anesthesiologists in critical intraoperative care settings (40). Moreover, it is usually administered for prophylaxis and the management of cardiac arrhythmia following cardiac operations (41). It decreases the sinoatrial node (SA node) impulse rate; however, it induces refractory period prolongation in the atrioventricular node (AV node) (42, 43). In their study, Saran et al. confirmed the decreased incidence of cardiac arrhythmia following the MgSO4 administration for patients undergoing thoracotomy (43).
Our findings showed that group I significantly expressed higher VAS scores than the other two groups (P < 0.05). Several studies have confirmed the analgesic effects of MgSO4 (44), which can induce the suppression of neuropathic pain, potentiate morphine-induced analgesia, and decrease the level of tolerance to opioids (45, 46). The mechanism of this analgesic effect has yet to be completely understood. However, it is assumed to be mediated by regulating calcium influx in the cells and antagonizing NMDA receptors in the central nervous system (47, 48).
The previously reported pain outcomes exhibited their effects on postoperative morphine consumption, which was increased in Group I; however, it was not significant (P = 0.111). Another study confirmed the slight but non-significant decrease in morphine need during the postoperative period after the MgSO4 administration. The insignificance of the statistical analysis results was probably due to the included small sample size (49).
To sum up, one should notice the significant positive effects of the IV or subcutaneous administration of MgSO4. The effects of its subcutaneous administration could be explained by its systemic absorption, as reported earlier in this discussion. Its administration should be encouraged by surgeons, especially for patients at high risk of epinephrine-related cardiac events during the liposuction procedure.
This study had some limitations; first, it was a single-center study, including relatively small sample size. Moreover, we should have evaluated serum magnesium levels in the receiving groups to detect existing correlations between its levels and hemodynamic variation. This would have also helped to estimate the level of MgSO4 absorbed into the systemic circulation in group III. Accordingly, further studies on more patients from different plastic centers are recommended.
5.1. Conclusions
The IV or subcutaneous administration of MgSO4 is associated with lower intraoperative HR, MAP, postoperative pain score, and a significant decrease in epinephrine-induced cardiac adverse effects during liposuction. Further positive effects were observed for the IV administration.