1. Background
Cells and tissues react when the skin is exposed to thermal injury. This damage includes cell death and tissue necrosis. Oxidative stress with the formation of free radicals, inflammation, infection, and edema can cause many problems in burns treatment (1). Wound healing is a physiologic process. The repairing phases include inflammation, proliferation, granular tissue formation, reepithelialization, matrix deposition, and remodeling. The essential treatment goals are healing skin lesions and regaining ideal performance (2). Wound healing is a complex process, which represents an attempt to restore a normal anatomical structure and function (3). The inflammatory process has essential effects on wound healing that includes coordination between immunological and biological responses. The healing process in an acute wound includes initial inflammation, deposition of collagen and fibroblast, angiogenesis, contraction of the wound, and scar remodeling (4).
The wound healing process can be improved by shortening the time needed for healing (5, 6). Chemical and herbal medicines are locally or systemically administrated to help wound healing. For this purpose, antibiotics, antiseptics, extracts, etc., have been used. Some chemical medicines are limited because of their side effects. Plants or herbal medicines derived from plants are needed to treat and manage wound healing. Herbal medicines have become more prevalent in recent years due to fewer side effects (7). Dwarf elder (Caprifoliaceous/Anoxaceae) is widely distributed in Iran. In Tradition Iranian Medicine (TIM), this plant is used to treat inflammation and arthritis (8-10). Pharmacologically, this plant has been reported to possess antibacterial and antioxidant activities and be effective against burns, wounds, eczema, and inflammation. An ointment made from the alcoholic extract of dwarf elder increased the wound tensile strength and collagen deposition (11).
2. Objectives
In this study, we evaluated the topical application of dwarf elder in full-thickness epidermal thermal wounds in Wistar rats. The rate of wound closure was measured in specific post-burn periods, and clinical and histological evaluations were performed to determine its efficacy.
3. Methods
3.1. Animal
Adult male Wistar rats (n = 30) weighing about 300 g were used in this study. All animals were obtained from the Animal Laboratory of AJA University of Medical Sciences. Rats were kept for one week in a 12 h light/dark cycle, 45 - 55% humidity, and 20 - 25°C temperature based on the AJA University Ethics Committee's approval (code IR.AJAUMS.REC.1398.124). After the adaptation period, the rats were randomly divided into three groups: (1) control group with no treatment; (2) vehicle groups treated with an ointment base; and (3) treatment group treated with dwarf elder ointment 10% (10).
3.2. Preparation and Extraction of the Plant
The plant was collected from Savadkuh heights. Then, it was approved in the Pharmaceutical Information Center of Tehran University with the Herbarium code PMP-370 Sambucus. L family Anoxaceae. The young leaves and twigs of the plant were dried and then pulverized. Extraction was performed by alcoholic variable maceration (12). For every 100 mL of concentrated extract, 8.2 g of dried extract was obtained. The dried extract was then collected from a container, and eucerin was used to make the ointment. Finally, 10 g of pure dwarf elder extract was dissolved in 100 g of eucerin and applied topically.
3.3. Thermal Injury Model
Animals were anesthetized by intraperitoneal injection of ketamine (80 mg/kg) and xylazine (10 mg/kg). Using the Meyer method (13) and the standard brass bar in the size of 1 × 1 centimeter, a second-degree burn with an area of 1 cm2 was created.
The treatment duration was 21 days. The wounds were photographed on 7, 14, and 21 days with a Canon camera. The wound area and healing degree were calculated by Image J software. Wounds were sampled on 7, 14, and 21 days. The wound areas were calculated as:
Where n is the number of days (14). After performing the tissue procedures, slides were stained with H&E. Then, they were examined by light microscopy and GraphPad Prism software to detect new blood vessels.
3.4. Statistical Analysis
Data were analyzed by SPSS software using ANOVA and Tukey’s test. The significance level was considered at P < 0.05.
4. Results
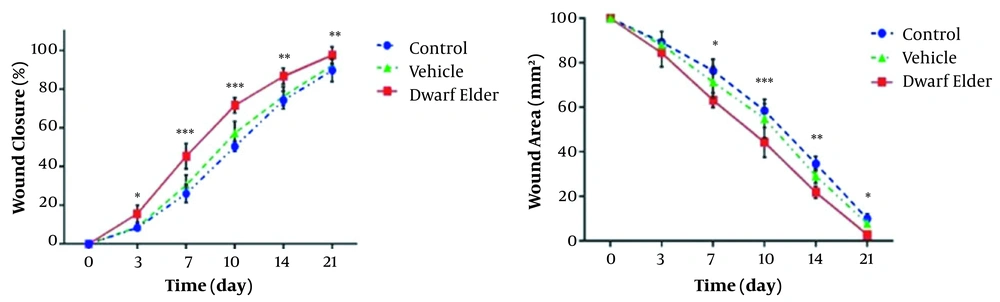
4.1. Wound Area
The wound areas were 34.69 ± 0.95 in the control group, 29.19 ± 0.96 in the vehicle group, and 21.81 ± 0.82 in the treatment group on the 14th day. The results showed that the wound area was significantly lower in the treatment group than in the control and vehicle groups (P < 0.01). The wound healing rates were 74.29 ± 1.35 in the control group, 76.37 ± 1.41 in the vehicle group, and 86.69 ± 1.23 in the treatment group on the 14th day, which showed a significant difference between the groups (P < 0.01).
The wound area on the 21st day was significantly lower in the treatment group than in the control and vehicle groups (P < 0.01). The highest wound healing rate was seen in the treatment group (97%). The wound healing rates on the 21st day were 89.69 ± 1.73 and 92.37 ± 1.21 in the control and vehicle groups, respectively, which showed a significant difference (P < 0.01) (Figure 1).
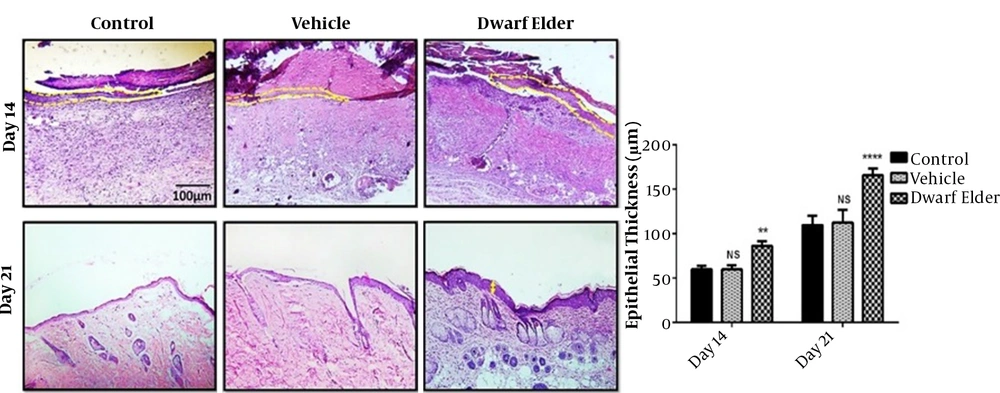
4.2. Epithelial Thickness
The epithelium thickness (μm) was significantly higher on the 14the day in the treatment group than in the control and vehicle groups (P < 0.001). The epithelium was present in all groups with distinct layers on the 21st day, but the thickness differed between the groups. The epithelium thickness (μm) was significantly higher in the treatment group than in the control and vehicle groups (P < 0.001) (Figure 2).
Left, histological sections of the wound site of different groups after creating a burn wound model (H&E staining, 200x magnification); right, epithelial thickness of the studied groups compared to the control group (data are reported as mean ± SEM; NS, not significant; ** P < 0. 01, **** P < 0.0001).
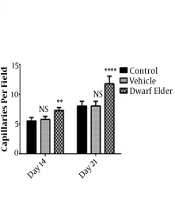
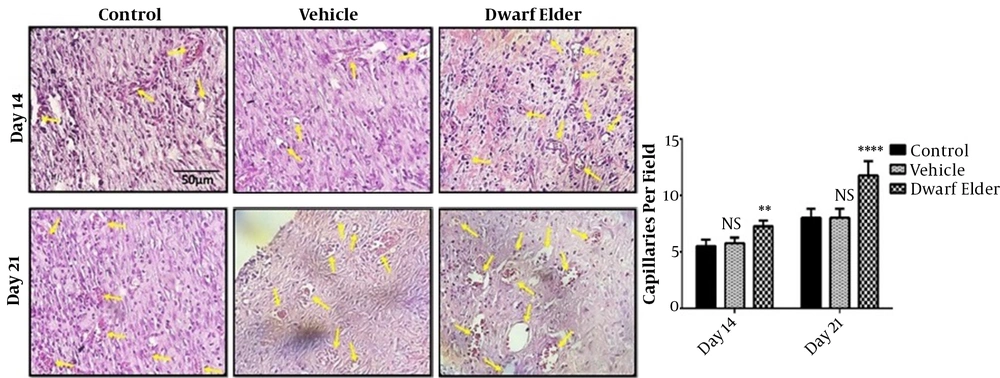
4.3. Blood Vessels
The results of counting blood vessels (number per field) on the 14th day were 5.8 ± 0.18 in the control group, 5.75 ± 0.16 in the vehicle group, and 7.25 ± 0.16 in the treatment group which was significantly higher in the treatment group (P < 0.001). The number of blood vessels on the 21st day was 8 ± 0.26 in the control group, 7.62 ± 0.49 in the vehicle group, and 11.75 ± 0.41 in the treatment group, which was significantly higher in the treatment group (P < 0.001) (Figure 3).
5. Discussion
Wound healing is a process that involves the interaction of cell types and matrix components. This process is characterized by three steps: Inflammation, proliferation, and regeneration. The proliferative phase usually included angiogenesis, collagen deposition, granulation tissue formation, epithelialization, and wound contraction (15). Recent studies have shown that medicinal plants with antioxidant properties can play a role in wound healing (16-22). Many researchers have reported dwarf elder to have potential antioxidant properties (23, 24). Elder contains flavonoids and several commonly active and diverse chemical metabolites (21, 25). Rutin and quercetin are the two main flavonoids in elder (26). The amounts of antioxidants rutin (1.77 μg/mL) and quercetin (0.376 μg/mL) (11) confirm the therapeutic value of elder. Mazandarani et al., in their research on the secondary active ingredients of elder, reported that the number of flavonoids was higher in the leaves than in other organs. In the highlands plants, the amounts of flavonoids increase (27). In our research, we used the elder leaves in the mountainous region of Savadkuh, which is located at an altitude of 900 m above sea level, showing more flavonoid properties.
Singh et al. reported in 2002 that due to the high levels of flavonoids such as rutin and quercetin in elder, the plant has anti-inflammatory properties and prevents the inflammatory activities of agents such as prostaglandins (28). Bobek et al. (2001) stated that flavonoids are the sources of antioxidants in elder (29). It is written in medical books that burn injuries, by creating and spreading free radicals and other inflammatory mediators, mainly contribute to lipid peroxidation. The increase in peroxidation is due to the thiobarbituric acid reaction. Quercetin has antioxidant and anti-inflammatory properties that reduce the epithelialization period in wound healing (30). The increase in epithelial thickness and wound healing in this study may be due to the presence of quercetin in elder (31). Bhatia et al. reported in 2016 that quercetin in burn wounds significantly reduced the wound area. They also reported that the effect of quercetin was low on day 11 but increased on day 21 with high doses of quercetin (30). Cvetanovic et al. reported in 2016 that only the elder leaves contain chlorogenic acid, a natural antioxidant that increases collagen synthesis during various phases of wound healing (31). In this study, the epithelium thickness in the treatment group increased on days 14 and 21, which was significant compared to the vehicle and control groups. Bagdas et al., in a series of studies in 2014 and 2015 on wound healing with chlorogenic acid, reported that this antioxidant increased epithelialization, angiogenesis, and proliferation of fibroblasts, and collagen formation, and decreased infiltration of multinucleated leukocytes. Their studies indicated a decrease in the process of inflammation due to the effect of this substance (32, 33). The results of our study are consistent with their reports.
5.1. Conclusion
This study showed that elder extract could be an effective remedy to repair and heal thermal injury and burn wounds.