1. Context
Sulfur Mustard (SM) has been an important subject of research for Iranian scientists since 1983, when Iraq army used this chemical weapon against Iranians, causing injury in more than 30 000 people, still suffering from its late effects (1, 2).
The first development of SM was documented in 1822 by Cesar-Mansuete Despretz (3). Years later, many other scientists, including Frederick Guthrie, Albert Niemann, Viktor Meyer, Hans Thacher Clarke, and Emil Fischer played major roles in developing synthetic methods for this hazardous viscount (4). The first use of SM, as a chemical weapon, refers to world war I in 1917, when the German army used SM against British and Canadian soldiers and also later against the French second army. It is worthy to mention that the British army developed its own formulation of mustard gas and used it during the breaking of the Hindenburg Line in 1918 (4). Unfortunately, since WWI, the dark history of using SM, as a chemical weapon, began (5). In 1919, the United Kingdom used this weapon against the red army (6), in 1921 to 1927, Spain and France used SM against the Rifian resistance in Morocco (5, 7), in 1930, Italy used it in Libya (5), in 1935 to 1940, Italy used SM against Abyssinia (now Ethiopia) (5), in 1935 to 1947 the Japanese Empire used it against China (6), in 1963 to 1967 Egypt used SM against North Yemen (5), and finally Iraq used SM against Iranians during 1983 to 1988 in the town of Halabja (6, 8).
One of the darkest memories of SM history belongs to the US army in 1943 in the harbor of Bari, Italy, where an American shipment of mustard gas exploded and killed 83 of 628 injured victims (9).
2. Evidence Acquisition
Data by searching online databases, including MEDLINE/PubMed, Google Scholar, and Science Direct, using related keywords, including mustard, gas, and veterans. While many researchers have tried to explain the underlying mechanisms of sulfur mustard-induced symptoms, yet the outcomes have been inconclusive thus the current review was performed to give an overview of what has been determined so far.
3. Results
3.1. Chemical Properties
Sulfur mustard as an organic compound has a formula of (Cl-CH2CH2)2S, which is synthesized by different methods (Table 1).
At normal temperatures SM is a viscous liquid. The pure compound has a melting point at 14°C (57°F) and before boiling, it decomposes at 218°C. If pure, it is colorless, and normally ranges from pale yellow to dark brown and has a slight garlic or horseradish type odor (13).
During the history of its development, sulfur mustard has been employed with various types and mixtures. These include (10, 11, 14):
- H - known as Levinstein mustard or HS (“Hun Stuff”).
- HD - Codenamed Pyro by the British and distilled mustard by the US.
- HT - Codenamed Runcol by the British, and Mustard T- mixture by the US.
- HL - A blend of distilled mustard (HD) and Lewisite (L),
- HQ - A blend of distilled mustard (HD) and sesquimustard (Q).
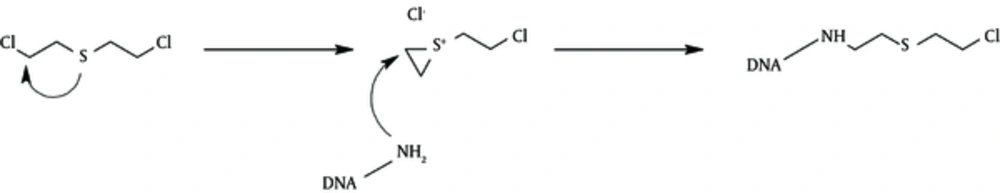
Sulphur Mustard alkyls has a variety of functional groups, such as amino and sulfide groups (15). Therefore, SM will affect the majority of proteins, making an alteration or cross-linkage, which results in a change in their function by varying degrees (Figure 1).
3.2. Attempts for Understanding the Underlying Mechanisms
The field of drawing pathways for SM mechanism is currently a land of challenge in scientific researches. While many researchers have tried to explain the underlying mechanisms of SM-induced symptoms, yet the outcomes have been inconclusive. In this review, some of the attempts are discussed:
A recent study claimed that sulfur mustard vesicants target cytochrome P450 reductase and this effect may be an important mechanism mediating oxidative stress and lung injury (16). Another study suggests a death receptor pathway of apoptosis using a feedback amplification mechanism involving an activated death receptor complex resulting in activation of caspase-9 via a caspase-3 pathway. Designing inhibitors of these pathways for therapeutic intervention is considered to attenuate SM injury in respiratory tract lesions (17). One study showed that it is possible to distinguish necrosis from apoptosis, two modes of cell death, and to quantify their extent in the pathophysiology of SM exposure (18).
Sulphur Mustard binding to specific proteins could contribute to the complex pathology seen after SM exposure (19). An Iranian study revealed that Smad3 and 4 may be involved in the airway remodeling process in SM-induced patients by activation of TGF-b (20). A study attempted to explain the pathophysiology of long-term consequences of SM toxicity. The results of this study suggested that skin exposure to SM leads to immune activation, infiltration of T cells into the SM exposed skin, delayed-type hypersensitivity response, and molecular imprints of inflammation in the tissues distal to the site of SM exposure (21).
Another study tried to draw the map of oxidative stress and SM exposure by presenting a protein called thioredoxin reductase (TrxR), a selenocysteine-containing flavoprotein, that catalyzes the NADPH-dependent reduction of oxidized thioredoxin and plays a key role in regulating cellular redox homeostasis. This study suggested that SM targets TrxR and this is an important mechanism mediating oxidative stress and tissue injury (22). A recent study investigated the pathogenesis of SM-induced lung injury. They claimed that IL-17+ secreting cells are likely to play an important role in the pathogenesis of SM-induced lung injury (23). Nanotechnology has currently introduced many opportunities to the research field of this challenging problem, and may lead to finding safe and effective ways to detoxify chemical warfare agents. This condition does not endanger human life or the environment. One of the important ways to achieve protection against these agents is to use appropriate materials able to perform the function of both physisorption followed by chemisorption (24).
3.3. Symptoms and Clinical Manifestations
Sulphur Mustard or mustard gas is an actual potent vesicant, the exposure of which could cause severe, irreversible damage to the skin, eyes, and the respiratory tract. Based on the level of exposure and the target organ, symptoms and signs vary differently. In the eye, it has a wide spectrum where it begins with an asymptomatic period following irritation and redness and ends with corneal damage with photophobia, blepharospasm, and temporary blindness (15, 25, 26).
Respiratory exposure primarily targets the nasal, laryngeal, and tracheobronchial mucosa, with early mild irritation leading to epistaxis, laryngeal injury, and bronchial edema and damage (27). Respiratory barrier function is compromised, and at higher concentrations, the lower respiratory epithelium becomes involved, resulting in impairment of gas exchange (15).
Sulphur Mustard exposure could cause laryngitis, tracheobronchitis, bronchiolitis, obliterans, bronchopneumonia, chronic obstructive pulmonary disease (COPD), bronchiectasis, asthma, and large airway narrowing (28-30). The mechanisms that underlie these SM-induced respiratory lesions, however, are unknown.
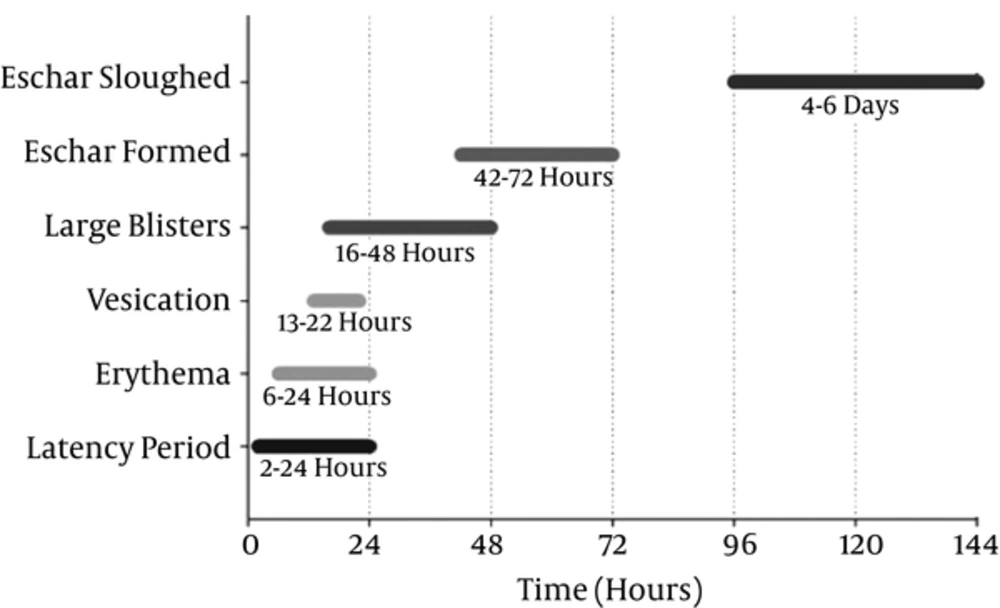
In skin exposure, erythema, the first sign, develops after a latency of 2 to 24 hours and is probably associated with intense itching. Several hours later, vesicles filled with a pale yellow fluid appear and with time, they may coalesce to form pendulous blisters. Blisters subsequently burst, leading to the formation of an eschar or a necrotic layer on the surface. Naturally, the wounds, resolve over a course of 10 to 50 days, causing pigmentation changes, which may persist for months or even years (1). show show the time course of pathophysiological responses after initial cutaneous SM exposure (Figure 2).
4. Discussion
The current study evaluated the effects of mustard gas on veterans. In many other studies, this gas and other related poisonous gases have been considered and examined. Based on this, these studies were evaluated in the current review to find poisonous and other effects of mustard on veterans. Annenbaum et al., in their study, reported that individuals, who had contact with this gas had lower education level and were more concerned about their health (31). Schnurr et al. identified factors, which protect or predispose veterans, who were exposed to mustard gas. It was reported that being a volunteer, poorer physical health prohibited disclosure, a higher likelihood of several chronic illnesses and health-related disability, greater functional impairment, and higher likelihood of healthcare were predisposing factors for partial and full posttraumatic stress disorder in these patients, although the role of education level was not evaluated (32). During the Iraq and Iran war, Iraq used chemical weapons almost 260 times, and more than 5000 Iranian soldiers were exposed to poisoning chemicals, especially the mustard gas (33, 34). Many studies mentioned that symptoms and signs of soldiers imply immunologic and inflammatory mechanisms (5, 30, 34-36), yet others mentioned that molecular patterns should be concerned in these patients. Life style, psychiatric parameters, and socioeconomic status are some factors affected by these clinical complications. Many previously performed studies have evaluated the lung complications of these patients (33, 36, 37), and showed that the frequency of lung complications in mustard gas-intoxicated patients was about 100%.
4.1. Conclusion
Nanoparticles of metal oxides have received enormous interest in the recent years because of their unique physical and chemical properties, such as Al2O3. A recent study showed that hydrolysis and dehydro-halogenation reactions could be the main route of degradation of 2-CEES (a less toxic simulant of sulfur mustard). However, we are still in the first steps of a very long road in understanding the underlying mechanisms of SM pathophysiology and applying detoxification methods.