1. Background
According to the World Health Organization, osteoporosis (OP) is a systemic disease of musculoskeletal system, which is characterized by low bone mass, decreased bone density and bone microstructural destruction that predisposes the individual to osteoporotic fractures. Osteoporosis is the most common metabolic bone disease in the world. Women constitute 80% of patients with osteoporosis (1). Fracture is one of major complications of osteoporosis in the elderly, so that one out of every three women and one out of every five men over 50 years will experience osteoporotic fractures (2). The primary goal in treating people with osteoporosis is to reduce fracture risk (3). One of the major pharmaceutical categories used in this situation is bisphosphonates that have been marketed in oral and injectable forms (4). This class of drugs inhibits bone resorption by blocking osteoclastic activity and can reduce the risk of vertebral and non-vertebral fractures. One of the disadvantages of oral bisphosphonates is the lack of compliance, acceptance and adherence of patients to regular consumption (daily or weekly) as well as gastrointestinal complications (5-7). This decrease in medication compliance reduces treatment success in reducing fractures and increases the cost of healthcare (8, 9), while intravenous infusion of 5 mg zoledronic acid once a year in addition to 100% guaranteed acceptance and lack of gastrointestinal side effects has been associated with improved bone density (10).
2. Objectives
Therefore, this study was conducted to evaluate and compare the therapeutic effect on Iranian patients with postmenopausal osteoporosis using weekly oral alendronate for a year and intravenous infusion of zoledronic acid once a year.
3. Methods
In this retrospective cohort study, the samples were collected by census sampling method. Thus, all postmenopausal women with osteoporosis whom referred to Physical Medicine Clinic of Tehran Imam Reza Hospital and two private rheumatology clinics in the cities of Gorgan and Shiraz over three years (2010 to 2012) who were under one year of treatment with 70 mg oral alendronate (OsteoFos, Cipla company, India) once a week or intravenous infusion of 5 mg zoledronic acid once a year (Aclasta, Novartis company) were included in this study. They had a history of bone mineral density (BMD) test before treatment and one year after treatment. Exclusion criteria included thyroid and parathyroid disorders, concomitant use or history of taking some drugs in the past six months including estrogen, calcitonin, strontium or sodium fluoride, anti-epileptic drugs, systemic corticosteroids, heparin, insulin and chemotherapy drugs.
In this study, 66 women with postmenopausal osteoporosis (N = 33 in each treatment group) were studied. Both alendronate and zoledronic acid treatment groups, in addition to taking mentioned bisphosphonate, had received simultaneously daily oral supplements containing 1,000 milligrams of calcium and 400 IU of vitamin D. Both of the BMD tests before and after treatment had been carried out at the same center for each person.
The T-score improvement (recovery rate) is defined as amount of increase in T-score number compared to before treatment. T-score is defined as the number of standard deviations above or below the mean for a healthy 30-year-old adult of the same sex and ethnicity as the patient (11).
Information of patients including age and T-score of femur and spine were collected before and after treatment, and then statistically analyzed by SPSS for windows version 19. One-sample Kolmogrov-Smirnov test was used to assess the normality of data distribution, unpaired sample t-test and for mean comparison before and after treatment, independent sample t-test and Mann-Whitney u-test for mean comparison between the two. Pearson’s test was used to assess the correlation of data. The values were considered statistically significant at P value < 0.05.
4. Results
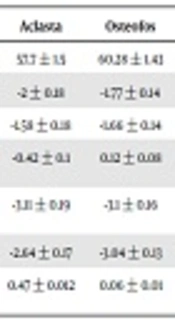
In total, 33 patients in the alendronate group and 33 patients in the zoledronic acid treatment group were studied. The mean age of the two groups was, respectively, 60.28 ± 1.43 and 57.7 ± 1.5 years, and there was no statistically significant difference (P = 0.223). the results are summarized in Table 1.
| Aclasta | Osteofos | P Value | |
|---|---|---|---|
| Age | 57.7 ± 1.5 | 60.28 ± 1.43 | 0.223 |
| Hip T-score before treatment | -2 ± 0.18 | -1.77 ± 0.14 | 0.321 |
| Hip T-score after treatment | -1.58 ± 0.18 | -1.66 ± 0.14 | 0.757 |
| Hip T-score changes after treatment | -0.42 ± 0.1 | 0.12 ± 0.08 | 0.019b |
| Spine T-score before treatment | -3.11 ± 0.19 | -3.1 ± 0.16 | 0.953 |
| Spine T-score after treatment | -2.64 ± 0.17 | -3.04 ± 0.13 | 0.07 |
| Spine T-score changes after treatment | 0.47 ± 0.012 | 0.06 ± 0.01 | 0.011b |
Patients Data Before and After Treatmenta
The mean T-score of femur in the patients before treatment was, respectively -1.77 ± 0.14 and -2.00 ± 0.18 in alendronate and zoledronic acid treatment groups, with no statistically significant difference (P = 0.321).
The mean T-score of spine in the patients before treatment was, respectively -3.1 ± 0.16 and -3.11 ± 0.19 in alendronate and zoledronic acid treatment groups, and the difference was not statistically significant (P = 0.953).
The recovery rate (T-score improvement) of femur in patients after one year of treatment in alendronate and zoledronic acid treatment groups was respectively 0.12 ± 0.08 and 0.42 ± 0.1, which was statistically significant (P = 0.019).
The recovery rate (T-score improvement) of spine in patients after one year of treatment in alendronate and zoledronic acid treatment group was respectively 0.06 ± 0.1 and 0.47 ± 0.012, and there was a statistically significant difference (P = 0.011).
The patient’s age did not have correlation with any of the variables (Table 2).
| Age | Hip T-Score Before Treatment | Hip T-Score After Treatment | Hip T-Score Changes After Treatment | Spine T-Score Before Treatment | Spine T-Score After Treatment | Spine T-Score Changes After Treatment |
|---|---|---|---|---|---|---|
| r | -0.212 | -0.208 | -0.019 | -0.016 | -0.103 | -0.112 |
| P | -0.076 | 0.082 | 0.873 | 0.892 | 0.390 | 0.347 |
Correlation of Age with Hip and Spine T-Scores Before and After Treatmenta
5. Discussion
According to our study, both alendronate and zoledronic acid groups experienced improvements in the BMD T-score. This increase in T-score was statistically significant in both groups. In a retrospective cohort study conducted by Craig et al. in 2011 on 169 patients with minor traumatic fracture, both oral and injection therapy improved bone density to the same value and even the risk of re-fracture was lower in the zoledronic acid group (12). However, we did not evaluate the incidence of fractures in the current study.
In another study by Shane et al. in 2012 on the prophylactic effect of alendronate and zoledronic acid on bone density loss in 84 liver and heart transplant patients, both drugs were effective in maintaining bone density in liver transplant patients but vertebral bone density has dropped in heart transplant patients in the alendronate group in spite of treatment (13). The study population in this study was different and simultaneously consumed corticosteroids, but its findings confirmed the strong effect of zoledronic acid in maintaining bone density.
In another study by Lyn Ferguson and colleagues in 2013 for three years on 65 osteoporotic patients, the effect of oral alendronate was greater than zoledronic acid although this difference was not statistically significant (14). Findings of this study were contrary to our results.
Orwoll et al. in 2010 examined the effects of alendronate and zoledronic acid on 302 men with osteoporosis. The effects of both medications on maintaining bone density at two-year follow-up were similar (15). Although the study population exclusively was men, their findings, similar to our study, showed beneficial effect of both drugs in maintaining bone density.
Lv et al. in 2018 studied effect of zoledronic acid vs alendronate in the treatment of 136 children with osteogenesis imperfecta. They concluded that yearly 5 mg zoledronic acid and weekly alendronate had similar effects in increasing BMD and reducing bone resorption in children and adolescents with osteogenesis imperfecta. However zoledronic was superior to alendronate in reducing the clinical fracture rate (16). Results of our study were similar to this study, but in a different population.
5.1. Conclusions
The injectable form of bisphosphonates (zoledronic acid) compared to oral form (alendronate) has more effect on improving osteoporosis and BMD T-score of postmenopausal women. It is worth examining patients in terms of reducing or increasing the risk of axial and pelvic bone fractures in prospective studies, particularly with a longer follow-up period.