1. Background
Sewage sludge is a byproduct of wastewater treatment process, which is composed of organic compounds, micronutrients, trace elements, microorganisms, etc. In fact, sludge includes large amounts of organic materials and nutrients, such as N, P, and K, making sludge useful as a fertilizer in agriculture or as an appropriate soil enhancer. However, physical and chemical processes involved in wastewater treatment cause heavy metals that are present in raw sewage to accumulate in sludge, which limits utilization of sludge as a fertilizer (1). Up to now, several techniques have been investigated for removal and recovery of heavy metals from wastewater sludge, including chemical precipitation, electrochemical methods using organic and inorganic acids, and bioleaching (2, 3). Each of these methods have advantages as well as disadvantages, such as using large amounts of reagents and chemicals, toxic sludge production, high costs, long process, and problems related to disposal of their remainders. (1, 4, 5). Bioleaching is a natural process that involves interaction between some ionic species, such as iron, sulfates, and microorganisms, such as A. ferrooxidans and A. thiooxidans (6). The unique characteristics of these microorganisms survive in acidic environments and insoluble oxidation of iron and sulfur compounds (7). Bioleaching, as a low-cost process for treating sewage sludge, solid waste, and other contaminated waste, has gained great importance. However, long reaction times, as well as the inability of the system to adapt to natural conditions are the main obstacles to its application.
Fenton advanced oxidation process is also one of the most common techniques that have been studied to remove heavy metals from sewage sludge (8). Fenton’s reagent is a mixture of Fe2+ and H2O2. Fe2+ is an initiator and catalyzes decomposition of H2O2, resulting in the production of hydroxyl radicals (OH) that are very reactive (9). Hydroxyl radicals are produced from the reaction of hydrogen peroxide with ferric ions under acidic conditions, which is known as Fenton-like reaction (10). Fenton process is a proper method due to low response time, enjoying the process of coagulation and flocculation, non-toxic compounds, and the possibility of being used in different scales (1, 8). Fu et al. found that the Fenton process was effective in removal of nickel from wastewater (98%) (8). In another study by Dewil et al., thermal acid hydrolysis decreased the content of heavy metals, except for Cu, Hg, and Pb, in the sludge layer (11). Mohammadi et al. studied dewatering of sludge using bio/Fenton-like oxidation process. Results showed that the combined method of bioleaching/Fenton-like oxidation using a lower dose of H2O2 and Fe significantly improved sludge dewatering (4).
Nevertheless, very few studies have been conducted using bioleaching combined with Fenton or Fenton-like methods to remove heavy metals from sludge. Chemical methods, such as Fenton and Fenton-like processes, combined with bioleaching method reduce the response time and the possibility of removal of more metal bioleaching compared to bioleaching alone. Besides, combined methods using a bioleaching process can reduce the need for chemicals in chemical processes.
2. Objectives
Therefore, the objectives of the present study are (1) to investigate the leaching efficiency of heavy metals (Cu, Zn, Cd, and Pb) during sludge bioleaching with inoculation of A. ferrooxidans and the addition of Fe2+ at different concentrations, and (2) to examine the removal efficiency of sludge-borne metal at optimal conditions in (a) bioleaching, (b) Fenton/bioleaching, and (c) bioleaching/Fenton-like processes.
3. Methods
3.1. Materials
Equal proportions of primary and secondary sludge from Shiraz, Fars wastewater treatment were collected in 4-liter polyethylene containers, transported to laboratory, and refrigerated (4°C) until the test. Each experiment was repeated twice and sometimes more on a factor at a time basis. The number of experiments was 140 and the number of sludge sampling was 10 times in autumn. Table 1 shows characteristics of raw sludge. All the chemicals were obtained from Merck Company, Germany.
| Parameter | Value |
|---|---|
| Hydration, % | 96 ± 0.6 |
| pH | 6 ± 0.3 |
| TSS, g/L | 35.1 ± 1 |
| VSS, g/L | 24.4 ± 0.4 |
| SCOD, g/L | 3.1 ± 0.2 |
| Zn, mg/kg | 4795 ± 150 |
| Pb, mg/kg | 264.1 ± 10 |
| Kd, mg/kg | 48.9 ± 5 |
| Cu, mg/kg | 821.6 ± 15 |
3.2. Microorganisms and Culture Medium
A. ferrooxidans (PTCC 1647) species of Iranian Research Organization for Science and Technology (IROST) were prepared as native species. The microorganisms were cultured in a 9 K culture medium that consisted of 3.0 g/L (NH4)2SO4, 0.1 g/L KCl, 0.5 g/L K2HPO4, 0.5 g/L MgSO4.7H2O, 0.01 g/L Ca(NO3)2, and 44.22 g/L FeSO4.7H2O (12). The pH of the culture medium was adjusted to 2.5 after adding FeSO4.7H2O. Then, 100 mL of the 9 K culture medium was poured into a 250 mL flask. After that, 10 mL of bacteria were added and the bacteria were cultured on a shaker at a speed of 180 rpm.
3.3. Adaptation of Bacteria in the Sludge
Adaptation of bacteria in the sludge consisted of two consecutive phases. In the first phase, 100 mL of sludge were poured in a 250 mL Erlenmeyer flask. After adding Fe2+ (4 g/L), 15 mL of the cultured bacteria were added and the flask was set on the shaker at the temperature of 28°C and speed of 180 rpm. When the pH reached about two, 15 mL of the sludge that had covered the first adaptation phase were added to 100 mL fresh sludge to which Fe2+ was added (2).
3.4. Metal Bioleaching Tests
Bioleaching tests were carried out in 250 mL flasks containing 100 mL fresh sludge, 15 mL of the adapted bacteria, and Fe+2 at concentrations of 1 - 4 g/L at the temperature of 28°C and speed of 180 rpm for nine days. The parameter levels were selected based on similar studies (2, 13).
3.5. Combined Bio-Acidification/Fenton-Like Method
In the combined Bio-acidification/Fenton-like method, first bioleaching process was conducted by addition of 15 mL of bacteria and Fe2+ at the concentrations of 0.5, 1, and 2 g/L to the sludge samples for 24 and 48 hours. For the Fenton-like process, H2O2 at the concentrations of 0.25, 0.5, and 1 g/L was added to the samples. In all the methods, the samples were dewatered using a Buchner pump at the end of the test and the remaining layer on the filter paper was dried in the kiln at the temperature of 105°C (4).
3.6. Analysis of the Samples
The dried samples were first turned into powder. Next, 250 mg of the sample (dry weight) were weighed and added to the furnace. Afterwards, 4 mL of 96% sulfuric acid was added to the sample and the mixture was heated to 200°C. When all the water was evaporated and the color became brown, 1 mL of 30% hydrogen peroxide was added to the mixture. After cooling of the crucible, the volume of the remaining mixture was increased to 100 mL by adding distilled water and some of the digested sample was taken for determining the amount of heavy metals. Polarography (Metrohm) and analysis method No. 113/2e were used to measure the amount of heavy metals (14).
3.7. Statistical Analysis
In the combined tests, the data were analyzed using the SPSS statistical software, version 21. At first, Shapiro-Wilk test was used to check the normality of dependent quantitative variables. Then, t-test and ANOVA were used to determine the significant variables.
4. Results
4.1. Bioleaching Tests
4.1.1. pH Changes with Time During the Bioleaching Process
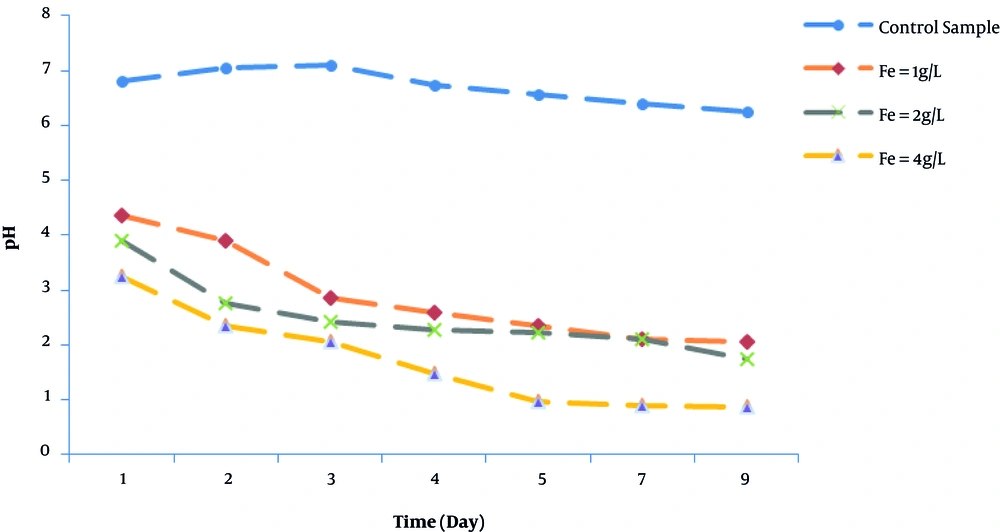
pH is one of the most important parameters affecting the solubility of heavy metals in sludge during bioleaching. Moreover, reduction of pH in bioleaching is indicative of activity of iron oxidizing bacteria (13, 15). pH changes with time during the bioleaching process in the samples containing A. ferrooxidans and iron sulfate (in different concentrations) and the control sample (without substrate and bacteria) have been shown in Figure 1. Accordingly, increase of iron concentration in the samples led to faster reduction of pH. In the control sample, pH of 6.82 decreased to 6.25 during nine days. This indicates less acid production since no antimicrobial activity and no power are supplied to support the growth of bacteria in the sludge. Reduction of pH during bioleaching indicates bacterial activity and growth. On the other hand, reduction of pH can be attributed to sulfuric acid production and hydrolysis of ferrous sulfate. Therefore, dissolution of metals depends on the iron hydrolysis and production of acid during bioleaching (6). One reason for sludge acidification is biological oxidation of S0 to SO42- compounds. Fe3+ precipitations in form of ferric hydroxide and jarosite also produce sulfuric acid (16).
4.1.2. The Effect of Different Concentrations of Fe2+ and Times on the Efficiency of Bioleaching
Iron sulfate, which is used as the energy source for the bacteria in the biological stage, affects heavy metals dissolution and removal efficiency (17). A. ferrooxidans needs Ferro ions as an energy source. H+ is produced by bio oxidation of Fe2+ that reduces pH and releases metals from the sludge (6).
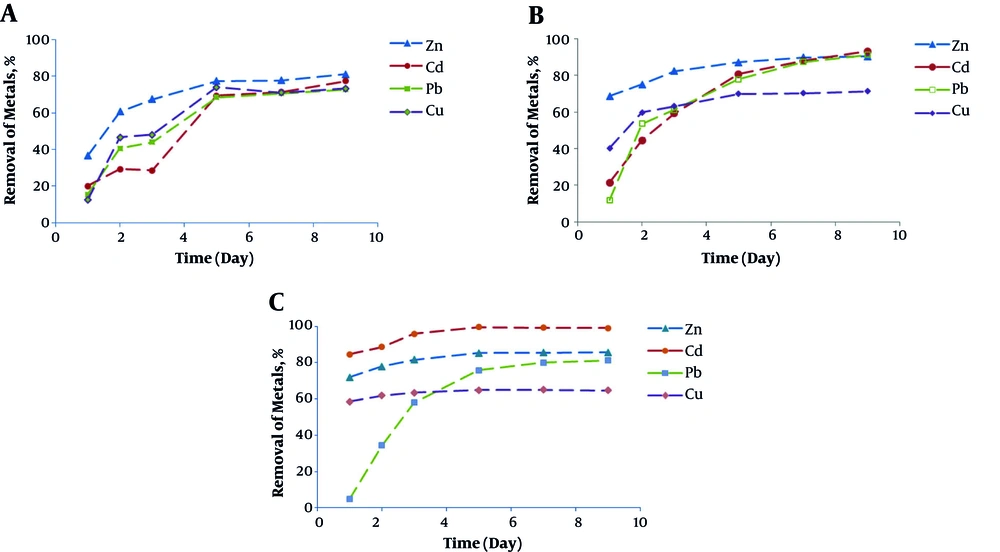
The effects of different concentrations of Fe2+ (1, 2, and 4 g/L) on the removal of heavy metals from the sludge have been shown in Figure 2 (a, b, c). As the figures depict at all the three concentrations of Fe2+, the metal removal rate significantly increased in 5 days but slightly decreased from day 5 to 9. At the Fe2+ concentrations of 1, 2, and 4 g/L in 24 hours, the removal rates were respectively 36.5%, 68%, and 72% for Zn, 20%, 21.5%, and 84.7% for Cd, 15.4%, 12%, and 4.8% for Pb, and 12.5%, 40.3%, and 58.6% for Cu.
4.2. The Effect of Combined Fenton/Bioleaching Method
In the next step, the combination of Fenton and bioleaching processes was employed to examine the possibility of leaching heavy metals from sewage sludge.
The results of these tests have been summarized in Table 2. In the Fenton process, using iron as a catalyst in acidic conditions led to decomposition of H2O2 and production of hydroxyl radicals. Hydroxyl radicals have the property of breaking up and decomposition of organic materials and release metals from sludge by degradation of metals attached to organic materials.
| Time (Day) | pH | Fe2+ (g/L) | H2O2 (g/L) | Cu Removal (%) | Pb Removal (%) | Cd Removal (%) | Zn Removal (%) |
|---|---|---|---|---|---|---|---|
| 2 | 6.06 | 0.5 | 0.25 | 50.1 | 44.6 | 73.7 | 76.1 |
| 2 | 5.64 | 0.5 | 0.5 | 60.5 | 45.6 | 75.8 | 78.9 |
| 2 | 5.48 | 0.5 | 1 | 71 | 58.7 | 76.6 | 81.3 |
| 2 | 5.03 | 1 | 0.25 | 37.5 | 42.7 | 74.5 | 74.7 |
| 2 | 4.46 | 1 | 0.5 | 69.2 | 60.2 | 81.2 | 78.5 |
| 2 | 3.98 | 1 | 1 | 77 | 68.9 | 82.5 | 83.3 |
| 2 | 4.18 | 2 | 0.25 | 66.2 | 45.6 | 72 | 77.1 |
| 2 | 3.95 | 2 | 0.5 | 70.2 | 59.2 | 77 | 80.17 |
| 2 | 3.08 | 2 | 1 | 86 | 82.5 | 87.5 | 83.9 |
Based on Table 2, at Fe2+ 0.5 g/L and H2O2 0.25 g/L, the removal rates of zinc, cadmium, lead, and copper were 59%, 45.8%, 13.3%, and 21.2%, respectively. By increasing Fe2+ to 2 g/L and H2O2 to 1 g/L, these measures reached 83.9%, 87.5%, 82.5%, and 86%, respectively. In fact, by increasing the concentrations of H2O2 and FeSO4, more reactions happen between the two substances and more hydroxyl radicals are produced, eventually increasing decomposition (18).
The study findings showed that in the combined Fenton/bioleaching method, Fe had a significant effect on the removal of Cu and Pb (P < 0.05). Cu has a great affinity for organic materials and when Fenton decomposes organic materials, Cu will be released from the sludge (11). In this method, the effect of H2O2 alone on the removal of metals was not significant. pH alone also had no significant impacts on the removal of Zn, Pb, and Cd, however, it had a great effect on Cu removal. This might be due to the fact that Cu is a stable metal with low mobility in which pH threshold is 2 - 3; thus, reducing pH has a great impact on its dissolution (19).
4.3. The Effect of Combined Bio-Acidification/Fenton-Like Method
In this method, Bio-acidification process was first conducted as pre-treatment followed by Fenton-like process. In this method, ferrous sulfate added to the sludge at the bioleaching stage was affected by biological oxidation and created sulfate, ferric ions, and other ferric products (10). After 48 hours of bioleaching, pH reduced from 5.84 to 2.1 in the presence of Fe3+ and favorable conditions were created for the Fenton-like reaction. Under acidic conditions from the reaction between ferric ions and H2O2, known as Fenton-like reaction, hydroxyl radicals are generated (20). These radicals oxidize organic compounds in the sludge and consequently destroy extracellular polymeric materials, decompose bacteria cells, and release heavy metals from the sludge (11).
Table 3 depicts the effect of the combined Bio-acidification/Fenton like process on the removal of metals depended on the concentration of Fe2+. The results also showed that in this method, H2O2 and Fe2+ concentrations alone were not effective in the removal of any of the metals, however, simultaneous presence of these two materials in the samples increased the removal of heavy metals from the sludge. In this process, pH was effective in the removal of Cd and Pb and to some extent effective in that of Cu. Overall; the best removal efficiency was obtained at 2 g/L concentration of Fe2+ and 1 g/L concentration of H2O2.
| Time (Day) | pH | Fe2+ (g/L) | H2O2 (g/L) | Cu Removal (%) | Pb Removal (%) | Cd Removal (%) | Zn Removal (%) |
|---|---|---|---|---|---|---|---|
| 2 | 4.10 | 0.5 | 0.25 | 34.4 | 20.9 | 9.8 | 34.3 |
| 2 | 3.48 | 0.5 | 0.5 | 66.5 | 55.3 | 30.4 | 48.3 |
| 2 | 3.20 | 0.5 | 1 | 69.1 | 62.4 | 51.9 | 58.8 |
| 2 | 3.45 | 1 | 0.25 | 52.4 | 45.4 | 23.5 | 51.08 |
| 2 | 3.08 | 1 | 0.5 | 62.3 | 67.1 | 57.8 | 65.3 |
| 2 | 2.79 | 1 | 1 | 70.9 | 81.4 | 74.5 | 71.2 |
| 2 | 2.5 | 2 | 0.25 | 83.7 | 62.9 | 91.1 | 68.9 |
| 2 | 2.36 | 2 | 0.5 | 90.6 | 78.1 | 94.1 | 81.9 |
| 2 | 2.11 | 2 | 1 | 94.5 | 87.06 | 97.06 | 93.02 |
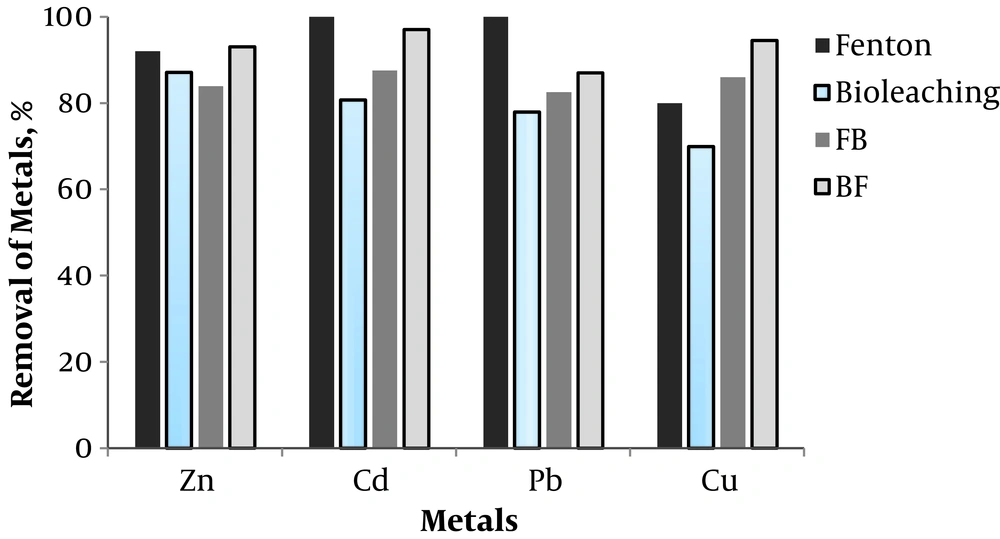
The removal efficiency of metals by bioleaching, Fenton, Fenton/bioleaching, and Bio-acidification/Fenton-like methods has been presented in Figure 3. The results of a previous study showed that in this method, the optimal conditions for the removal of heavy metals were pH of 2 - 3, hydrogen peroxide concentration of 3 g/L, Fe2+ concentration of 2 g/L, and leaching time of 15 min. Under these conditions, 92% of Zinc, 100% of cadmium, 100% of lead, and 80% of copper were removed from the sludge (8).
After using Bio-acidification/Fenton-like method, the remaining amounts of zinc, cadmium, lead, and copper were 434, 0.12, 7.4, and 76 mg/kg, respectively, which is suitable for use in the farm range. The maximum allowable concentrations of Cd, Cu, Pb, Zn, and Fe in sludge for agricultural purposes recommended by EPA are 39, 1500, 300, 2800, and 11000 mg/kg, respectively (21).
5. Discussion
This study aimed to assess the removal of heavy metals from sewage sludge by bioleaching and combined Bio-acidification/Fenton-like methods. In Bio-acidification/Fenton-like method, iron oxidizing microorganisms can oxide Fe2+ to Fe3+, produce H+, and decrease pH of sludge to the required value in order to respond to the Fenton-like process. Subsequently, an addition of small amounts of H2O2 significantly improved the removal of heavy metals. In this process, the optimal condition included Fe2+ concentration of 2 g/L, bioleaching time of 2 days, and H2O2 concentration of 1 g/L. Under these conditions, the removal efficiency of zinc, cadmium, lead, and copper was 93%, 97%, 87%, and 94.5%, respectively, which corresponds to the sludge disposal standards for use in agriculture.