1. Background
Liver cirrhosis is a pathological stage characterized by diffuse liver fibrosis, pseudolobular formation, and blood vessel proliferation in and outside the liver as the characteristics of various chronic liver diseases. About two million people die of liver disease, and one million people die of complications of liver cirrhosis in the world every year. Liver cirrhosis is currently the 11th most common cause of death in the world, bringing a serious health and economic burden to the world (1, 2). Liver cirrhosis can lead to gastrointestinal bleeding, ascites, hepatic encephalopathy, and even life-threatening. Thus far, there is still no clear consensus on the treatment of liver cirrhosis. At present, it is mainly etiological treatment, anti-inflammatory, anti-hepatic fibrosis, and active prevention and treatment of complications. We may need a liver transplant if the liver is severely damaged (3). Patients with early liver disease usually have no symptoms and may have reached the advanced stage of the disease when they are found. Cirrhosis can lead to irreversible damage, but the early stage of chronic liver disease can be reversed to a healthy state. Therefore, it is essential to find biomarkers to identify the early stage of chronic liver disease to prevent severe liver damage. The discovery of biomarkers for liver cancer and liver cirrhosis has promoted sequencing technology development, and high-throughput sequencing is one of the representative technological innovations in the biological field in recent decades (4). Recently, many studies have been conducted to clarify the pathogenesis of liver cirrhosis based on the experimental data of microarray and high-throughput sequencing (5-8).
The cost reduction of high-throughput technology and the development of bioinformatics have brought new opportunities to reveal the pathogenesis of liver cirrhosis. Although most studies only pay attention to the differences between cirrhotic cases and normal controls, they rarely pay attention to the differences between cirrhotic cases. To reveal the heterogeneity between tumors, cancer cases are usually divided into multiple subgroups based on gene expression patterns (9). Inspired by cancer research, we used the same method to divide cirrhosis cases into four subgroups, the differences between subgroups were found by annotating the corresponding coexpression functional modules with the path of Kyoto Encyclopedia of Genes and Genomes (KEGG), thereby deepening understanding of the differences between cases of cirrhosis.
2. Methods
2.1. Data Collection
2.1.1. Data Download and Annotation
We used R/Bioconductor package GEOquery (10) to extract "gene expression omnibus" (GEO) objects. Searched the GEO database (https://www.ncbi.nlm.nih.gov/geo/), the search term was "cirrhosis", the gene chip that meets the requirements was incorporated into our research. According to the annotation information of the platform file, converted the probe matrix into a gene matrix.
2.1.2. Inclusion and Exclusion Criteria
Inclusion criteria: (1) populations diagnosed with cirrhosis, regardless of the severity of the disease; (2) study type is expression profiling by array; (3) entry type is series; (4) research contains samples from the cirrhosis disease group and the normal group.
Exclusion criteria: (1) there is no comparison between the disease group and the normal group; (2) the sample size of each group is less than six.
2.2. Removal of Batch Effect
Firstly, the installation package in R/Bioconductor was used to merge the obtained gene matrix, and batch correction was carried out (11). When merging data, log2 was taken for the data with large value for conversion. When there is a difference between batch effects in the data, ComBat-seq can obtain better statistical ability and control the false positive rate compared with other available methods (12). Therefore, the combat method was used to eliminate the batch effect. Finally, principal component analysis (PCA) was performed to evaluate the results.
2.3. Consensus Clustering
First, we used the "consensus cluster plus" package in R/Bioconductor for consensus clustering (13). The included cases of liver cirrhosis were clustered using the K-means algorithm with Spearman distance, the clustering results were determined by referring to the consistency score (> 0.8).
2.4. Extraction of Specific Upregulated Genes and Hub Genes in Subtypes
The subtypes were compared with other subtypes to determine the subgroup-specific upregulated genes. Wilcoxon rank-sum test was used to test the differential expression. The corrected P-value was < 0.05, and the absolute difference of means > 0.2. The difference of means is the mean of the subgroup minus the mean of the other subgroups. The specific upregulated genes in the subgroup were imported into the STRING database, and the species condition was set to human, the confidence level was greater than 0.9, and the protein-protein interaction (PPI) network was constructed. The PPI was imported into Cytoscape 3.8.0, and the Degree algorithm in the cytoHubba plug-in was used to obtain the most significant top 10 hub genes.
2.5. Gene Set Enrichment Analysis
Gene set enrichment analysis (GSEA) was implemented in GseaPrerank mode in GSEA 4.1.0. It was used to judge whether the specific differential genes in each subtype are also different from normal samples. This operation requires two files, one is the gene set file (type-specific gene file) composed of subgroup-specific upregulated genes, and the other is the gene list file, which is obtained by comparing the specific type with the normal sample. We compared these two files to see how similar they are.
2.6. Weighted Gene Coexpression Network Analysis
Weighted gene coexpression network analysis (WGCNA) is often used to find modules of highly relevant genes (14, 15). We used WGCNA to type the gene and got gene’s module. Firstly, a distance matrix can be obtained by finding the optimal power value and calculating the distance between genes. It was convenient for gene clustering and dynamic identification module. After merging similar modules, we finally determined six functional modules. At the same time, we used the labeled heat map function option in the WGCNA package to draw the heat map, visualized the data, set the low expression of the gene as blue, the middle expression as white, and the high expression as red.
2.7. GO Enrichment Analysis and KEGG Enrichment Analysis
GO enrichment analysis, and KEGG enrichment analysis were performed for each functional module of WGCNA, in which GO was divided into three modules: (1) biological process (BP); (2) cellular component (CC); and (3) molecular function (MF) (16). KEGG is a database that integrates genomic, chemical, and system function information (17). We compared the pathways between modules through KEGG enrichment analysis. During KEGG enrichment analysis, set p-value < 0.05 for filtration. The enrichment results were visualized, and the bubble diagram was drawn.
3. Results
3.1. Microarray Data Characteristics
This study included four independent microarray information, involving four independent clinical trials, all from the GEO database, GSE14323, GSE77627, GSE123932, GSE128726, a total of 127 samples (including 79 patients with liver cirrhosis and 48 healthy subjects). GSE123932 provided clinical information of gender and age.
3.2. Removal of Batch Effect
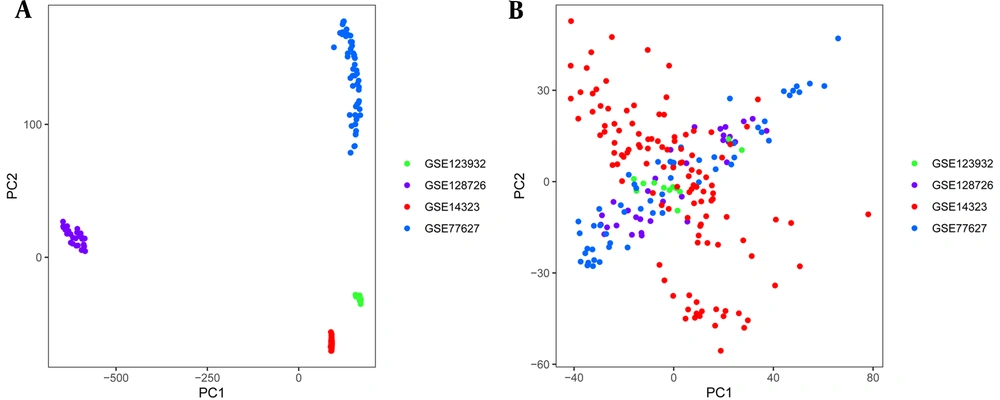
Four gene chips detected a total of 11,152 genes. In order to eliminate the batch effect, we used the combat method. The PCA chart before correction showed that there were large sample differences between experiments before the batch effect was eliminated (Figure 1A). In contrast, the samples in the corrected PCA chart were randomly distributed, and the results showed that the cross-platform normalization successfully eliminated the batch effect (Figure 1B).
3.3. Consensus Clustering of Cirrhosis Cases
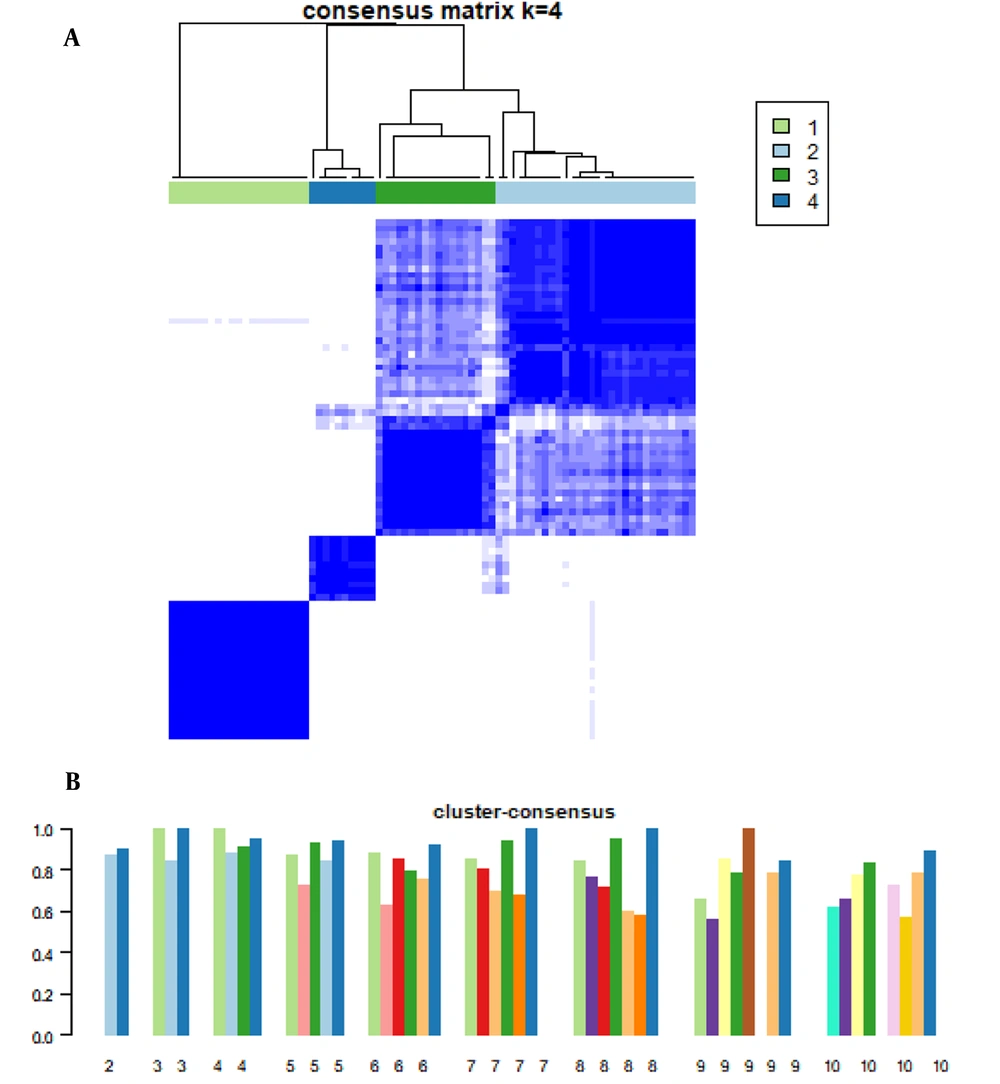
After eliminating the batch effect, 79 patients with liver cirrhosis were divided into subgroups. According to the consistency scoring situation in the data statistics, the consistency cluster analysis of gene expression profiles was divided into four subgroups, among which the number of cases in subgroups I, II, III and IV were 21, 30, 18, and 10, respectively. The internal relationship of subgroups is close, while the correlation between subgroups is weak (Figure 2A). Generally speaking, the stability of the type is positively correlated with the consistency score. According to the results of this study, when divided into 2, 3, or 4 groups, the cluster agreement score of each subgroup was higher than 0.8 (Figure 2B), which indicated that these classifications were more robust than other clusters. When considering more groups better, 79 patients with liver cirrhosis were divided into four subgroups.
Consensus clustering of gene expression profiles in patients with liver cirrhosis. A, the consistency matrix, when the number of clusters, is 4 that is determined by the minimum consistency score (> 0.8) of the subgroup; B, consistency scores for subgroups with cluster numbers between 2 and 10.
3.4. GSEA Enrichment Analysis and WGCNA Analysis Results
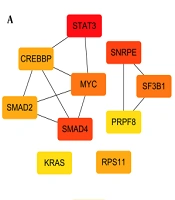
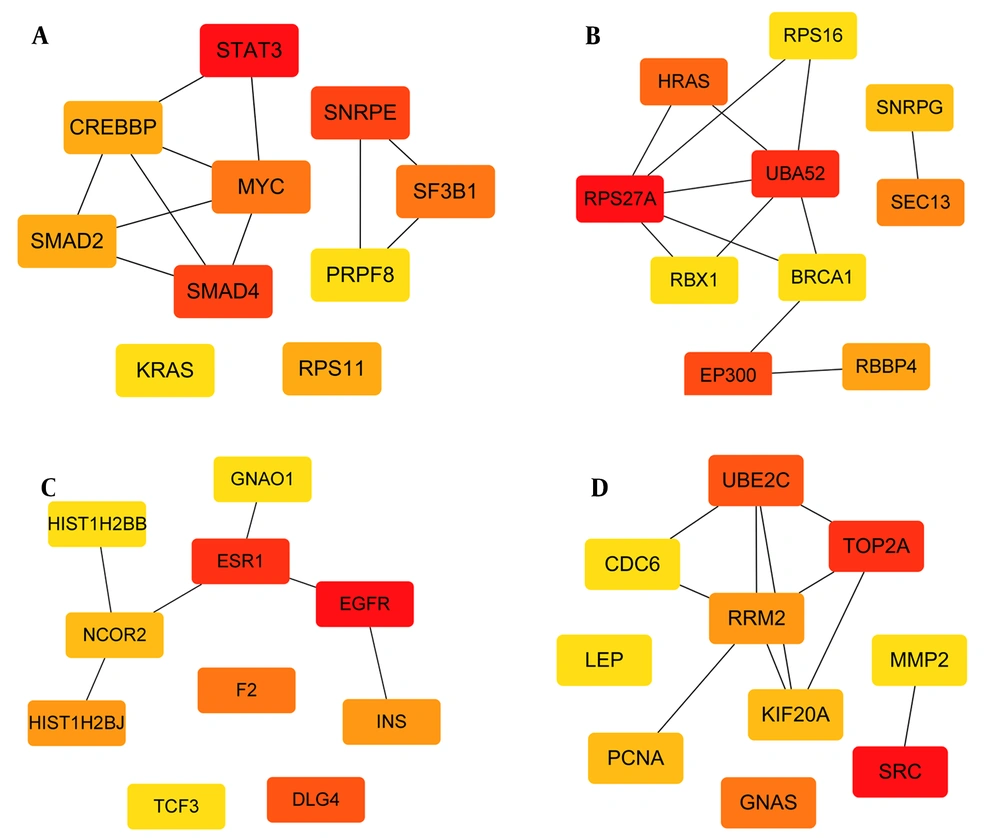
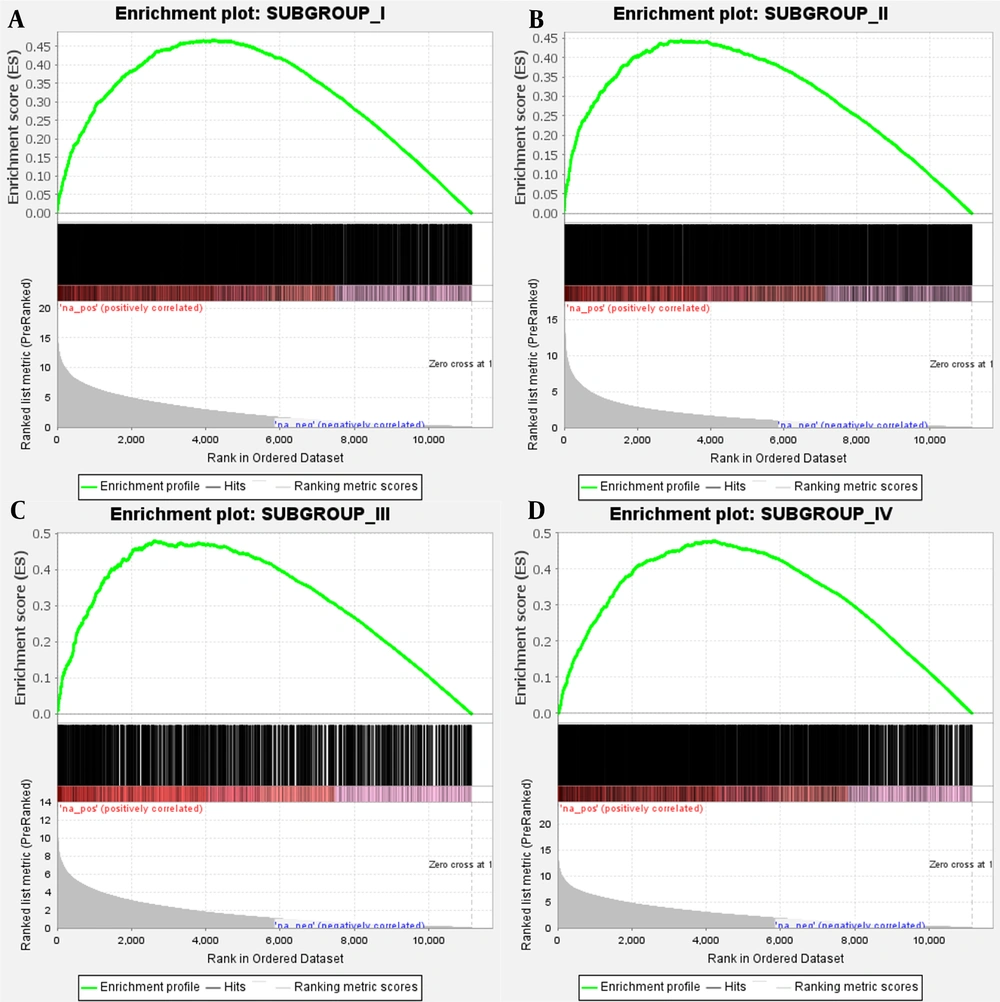
The 2033, 2134, 804, and 1892 genes that were specifically upregulated in subgroups I, II, III, and IV were determined by differential analysis. The subgroup differential genes were imported into the STRING database, and a PPI network was constructed. Imported it into Cytoscape 3.8.0 software and arranged the nodes in an orderly manner according to the degree value. The top 10 hub genes of each subgroup are shown (Figure 3A - D). The results of GSEA enrichment analysis showed that the specific upregulated genes in the subtypes were also significantly different when compared with normal samples (Figure 4A - D, FDR < 0.05). It still needs to be emphasized that subgroup III had the least subgroup-specific upregulated genes, and compared with the normal group, there were less specific genes (Table 1).
| Subtype | Specific Genes were Compared with The Normal Group | Specific Genes Compared with Other Subgroups | Specific Upregulated Genes in Subgroup | Modular |
|---|---|---|---|---|
| I | 6,021 | 4,848 | 2,033 | Black |
| II | 3,387 | 3,169 | 2,134 | Brown, purple |
| III | 3,929 | 2,274 | 804 | |
| IV | 6,404 | 6,407 | 1,892 | Turquoise |
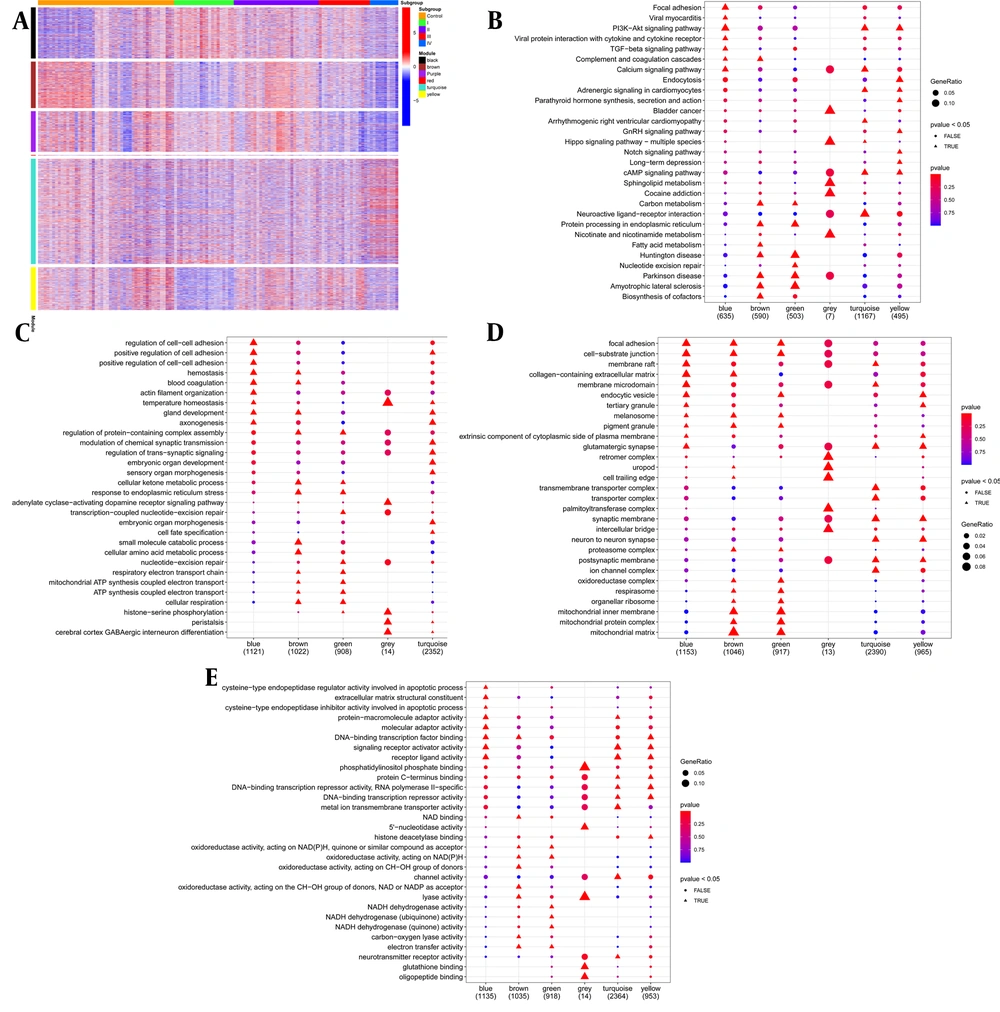
We typed 6,863 specific genes upregulated in four subgroups by WGCNA, and identified six WGCNA modules. The subgroups corresponding to WGCNA modules are summarized in Table 1. The genes of six modules were analyzed by KEGG. The results showed that TGF-β signaling pathway, viral protein interaction with cytokines, and cytokine receptors, including a variety of chemokines and inflammatory factors, were only enriched in the black module significantly. At the same time, the PI3K/Akt signaling pathway was also significantly enriched in the black module, and there were many upregulated genes. The genes in the black module were upregulated in subgroup I, indicating that subjects in subgroup I may exhibit inflammatory characteristics. Fatty acid metabolism and biosynthesis of cofactors were only significantly enriched in the brown module, and carbon metabolism and protein processing pathways in the endoplasmic reticulum were only significantly enriched in the brown and purple module. The genes in the brown and purple module were upregulated in subgroup II, indicating that subjects in subgroup II may show metabolic activity; arrhythmogenic right ventricular cardiomyopathy and neuroactive ligand-receptor interaction pathways were only significantly enriched in the turquoise module, and the genes in the turquoise module were upregulated in subgroup IV. We did not find a significant pathway of upregulation in the third subgroup. Based on the above results, different subgroups regulate the occurrence or progression of liver cirrhosis through specific pathways (Figure 5A - B).
After GO enrichment analysis (Figure 5C - E), the biological process (BP): the black module mainly included the regulation of cell-cell adhesion, positive regulation of cell adhesion, positive regulation of cell-cell adhesion, blood coagulation, and hemostasis; the brown module mainly included small molecule catabolic process, cellular amino acid metabolic process, cellular ketone metabolic process, and response to endoplasmic reticulum stress; the purple module mainly included nucleotide-excision repair, respiratory electron transport chain, ATP synthesis coupled electron transport, mitochondrial ATP synthesis coupled electron transport, cellular respiration, etc.; and the turquoise module mainly included axonogenesis, gland development, regulation of trans-synaptic signaling, modulation of chemical synaptic transmission, embryonic organ development, sensory organ morphogenesis, embryonic organ morphogenesis, etc. Cell components (CC): the black module concentrated on focal adhesion, cell-substrate junctions, membrane rafts, collagen-containing extracellular matrix, membrane microdomain, and endocytosis vesicles; the brown module concentrated on mitochondrial matrix, mitochondrial protein complex, mitochondrial inner membrane, focal adhesion, cell-substrate junction, collagen-containing extracellular matrix; the purple module concentrated on mitochondrial matrix, mitochondrial protein complex, mitochondrial inner membrane, organellar ribosome, respirasome, and oxidoreductase complex; the turquoise module mainly concentrated on transmembrane transporter complex, transporter complex, synaptic membrane, neuron to neuron synapse, postsynaptic membrane, and ion channel complex. Molecular function (MF): the black module mainly included receptor ligand activity, signaling, receptor activator activity, DNA-binding, transcription factor binding, etc.; the brown module mainly included DNA-binding, transcription factor binding, oxidoreductase activity, acting on the CH−OH group of donors, NAD or NADP as acceptor, lyase activity, carbon-oxygen lyase activity, electron transfer activity; the purple module mainly included NADH dehydrogenase activity, NADH dehydrogenase (quinone) activity, NADH dehydrogenase (ubiquinone) activity; the turquoise module mainly included signaling receptor activator activity, receptor ligand activity, metal ion transmembrane transporter activity, channel activity, neurotransmitter receptor activity, etc.
4. Discussion
Cancer molecular subtypes inspired this research. The current molecular subtypes guide the diagnosis and clinical treatment of many cancer types (18-20). In addition to cancer research, non-cancerous diseases can also identify new and clinically relevant molecular subtypes of diseases based on gene expression profiles (21, 22). Although these molecular subtypes are not perfect, they help for the diagnosis and treatment of diseases to a certain extent.
Liver cirrhosis is relatively complex and shows clinical heterogeneity. Previous studies mostly compared the gene expression profiles of liver cirrhosis cases with normal controls. In this study, we further divided liver cirrhosis cases into subgroups. Unlike previous studies, we linked specific pathways with specific subgroups of liver cirrhosis.
The hub genes in the first subgroup are mainly STAT3, SMAD2, SMAD4, MYC. STAT3 regulates basic cell processes, including inflammation, cell growth, proliferation, differentiation, etc. Studies have shown that STAT3 integrates a variety of profibrotic signals and may be the core mediator of fibrosis (23). Moreover, TGF-β is one of the key drivers of fibrosis. The production of TGF-β is related to the progression of liver fibrosis. In many experimental models, inhibiting the TGF-β1 signaling pathway has been shown to reduce the development of fibrosis (24). In TGF-β1 signaling, the Smads protein is the main mediator of receptor-induced phosphorylation and nuclear translocation (25, 26). In chronic liver disease, inflammatory mediators interact with c-Myc, leading to fibrosis, liver cirrhosis, and liver tumors (27). In our study, the TGF-β signaling pathway was most significantly enriched in the first subcomponent type. TGF-β signaling is involved in the entire process from liver injury to inflammation and fibrosis, cirrhosis, and cancer, TGF-β plays a key role in hepatocyte cell growth and apoptosis and promotes liver differentiation during embryogenesis and physiological liver regeneration. However, due to chronic liver injury, high levels of TGF-β cause stellate cells to activate myofibroblasts and a large number of hepatocytes to die, promoting liver fibrosis and cirrhosis (28).Viral protein interactions with cytokine and cytokine receptors were also most significantly enriched in the first subcomponent type, including a variety of chemokines and inflammatory factors, such as chemokines CCL2, CX3CL1, etc., biliary epithelial cells (BECs) in damaged small bile ducts in primary biliary cirrhosis (PBC) show senescent features. Senescent BECs exhibit upregulation of a variety of chemokines and chemotactic activities. The expression of CCL2 and CX3CL1 increases in senescent BECs in PBC, which may promote the infiltration of corresponding cells expressing CCR2 and CX3CR1, and further aggravate the PBC bile duct inflammation of the lesion (29, 30).
At the same time, PI3K/Akt signaling pathway was also significantly enriched in subgroup I, and there were many upregulated genes. PI3K/Akt signaling pathway regulates the activation of downstream signaling molecules. It plays a key role in regulating the immune response and the release of inflammatory factors in vivo and in vitro (31). Combined with the results of GO functional enrichment analysis, the first subcomponent mainly included the regulation of cell-cell adhesion, positive regulation of cell adhesion, positive regulation of cell-cell adhesion, blood coagulation, and hemostasis, and other biological processes. The combination of cell to extracellular matrix interaction and cell-cell adhesion depends on intercellular adhesion molecules. Cell adhesion molecules participate in various interactions. They can broadly support fibrosis formation by releasing TGF-β, promoting fibrosis, or mediating leukocyte migration. Some candidate cell adhesion molecules have been evaluated as targets for preventing and/or reversing liver inflammation and fibrosis (32). Inflammation triggers clotting, reduces the activity of natural anticoagulant mechanisms, and impairs the fibrinolytic system. Inflammatory mediators can increase platelet count, platelet reactivity, downregulate the natural anticoagulation mechanism, activate the coagulation system, promote the spread of coagulation response, and damage fibrinolysis. Similarly, coagulation can increase the inflammatory response by releasing mediators from platelets and activated cells (33). In summary, it was indicated that subjects in subgroup I may exhibit inflammatory features.
In the second subgroup, the hub genes mainly include RPS27A, HRAS, SEC1, UBA52, etc. Among them, RPS27a performs extra-ribosomal functions in addition to playing a role in ribosome biogenesis and post-translational modifications of proteins. RPS27a plays a role in mouse liver cancer and some overexpressed in human tumors. Studies have reported that STAT3 is the key activator of the RPS27a promoter, and upregulation of RPS27a depends on STAT3 phosphorylation (34). Studies have shown that increased HRAS protein level can directly stimulate transduction of the Smad2/3 signal and the accumulation of type I collagen, which is related to the appearance fibrosis (35, 36). Upregulation of metabolism was observed in subgroup II, including fatty acid metabolism, cofactor biosynthesis, carbon metabolism, and protein processing pathways in the endoplasmic reticulum. Combined with the results of the GO functional enrichment analysis, subgroup II mainly included the small molecule catabolic process, the metabolic process of cellular amino acids, the metabolic process of cellular ketones and the response to endoplasmic reticulum stress, repair of nucleotides, respiratory electron transport chain and other biological processes, indicating that subjects in subgroup II may exhibit metabolic activity characteristic. Systems biology studies have shown that carbohydrates, amino acids, and lipid-related pathways have changed in liver fibrosis and cirrhosis, and more studies are needed to verify metabolic characteristics and determine therapeutic targets of liver fibrosis (37).
The hub genes in the fourth subgroup mainly include SRC, TOP2A, UBE2C, and so on. Studies have shown that Src is related to liver fibrosis. The expression of Src mRNA in liver tissues of mice with liver fibrosis is significantly increased, the levels of phosphorylated Src and total Src are significantly increased; Src in liver tissues of patients with liver cirrhosis is significantly higher than in liver tissue from the normal control group. Src inhibition can prevent TAA-induced liver fibrosis, inhibit HSC activation, and is a potential therapeutic target for liver fibrosis (38). Differential gene expression analysis and weighted gene correlation network analysis were performed in tissue samples from normal liver, cirrhosis, and HCC to determine the pathological progress of 20 hub genes from cirrhosis to HCC. TOP2A is one of the pivotal genes in the pathological progression from liver cirrhosis to HCC (39). Research results indicate that UBE2C may be a key gene in the progression of HCC and a promising therapeutic target for the treatment of HCC (40, 41). Arrhythmogenic right ventricular cardiomyopathy and the neuroactive ligand-receptor interaction pathway were only significantly enriched in subgroup IV. Studies have shown that the neuroactive ligand-receptor interaction pathways are closely related to the appearance and development, invasion, and metastasis of liver cancer. Various traditional Chinese medicine and Chinese patent medicine exert their curative effects by influencing this signaling pathway (42-44). However, the relationship between this pathway and its key targets and liver cirrhosis needs further study. In the third subgroup, the hub genes mainly include B4GALT2, FUT9, FUT3, EGFR, and so on. Relevant studies have shown that the expression of EGF in the liver increases during cirrhosis, and inhibition of EGFR can reverse liver fibrosis. The use of FDA-approved inhibitors to inhibit EGFR provides a promising treatment for reducing liver fibrosis and preventing HCC (45). We did not find a significantly upregulated pathway in subgroup III, which may be related to the small sample size.
The above results show that different subgroups of liver cirrhosis have different gene expression patterns and obvious heterogeneity, which may represent different stages of liver cirrhosis, and we seek for large-sample prospective trials to verify it in the future. This study has certain limitations. Because only GSE123932 provided clinical information of gender and age, and the sample size was small, the correlation between clinical characteristics and WGCNA module could not be carried out.
In conclusion, inspired by the study of cancer subgroups, we adopted a similar strategy to reveal the molecular subgroups of liver cirrhosis. The ultimate goal of the study of these different phenotypes is to find patient groups with unique treatment characteristics and formulate targeted treatment plans. Our study suggests that patients in different subgroups may have their unique molecular characteristics. This new classification method is helpful for researchers to explore new treatment strategies for liver cirrhosis according to clinical phenotypic characteristics, to improve the prognosis of the disease and improve the quality of life of patients.