1. Introduction
Choriocarcinoma (CC) is the most aggressive type of gestational trophoblastic disease. Histologically, it is composed of mononuclear cytotrophoblasts, extravillous trophoblasts, and syncytiotrophoblasts. Hemorrhage and necrosis may be predominant in CC. Due to extensive bleeding, extensive tissue sampling is required to demonstrate the biphasic histological characteristics of these neoplasms (1). Approximately one-half of CC cases are reported following a hydatidiform mole, while the other half is reported after non-molar pregnancies (2, 3).
CC often presents with abnormal uterine bleeding and can have a variety of symptoms, depending on the site of metastasis. However, uterine rupture, which presents as acute hemoperitoneum, is rarely documented. Currently, we are in an ongoing global pandemic of COVID-19 infection, which has caused many challenges for patients with cancer in terms of the required equipment, facilities, and treatment. Here, we present a rare case of CC, complicated by uterine rupture, and concurrent COVID-19 infection.
2. Case Presentation
The patient was a 34-year-old woman with a history of irregular menstrual cycle and consumption of oral contraceptives and Accutane tablets without a history of pregnancy. After heavy vaginal bleeding with positive human chorionic gonadotropin (hCG) and retained products of conception on sonography, the patient had undergone emergency curettage in another country in December 2019. We did not have access to pathology tests. However, after 2 months, she underwent a second curettage in Iran in February 2020 because of vaginal rebleeding and retained products of conception on sonography, and the pathology report confirmed CC.
Within 2 months, due to the persistence of bleeding and new sonographic report of myoma, she was administered gonadotropin-releasing hormone agonists (Decapeptyl) regardless of pathology tests. Finally, the patient was referred to our hospital in Iran on April 3, 2020, for further management of CC. On admission, she showed non-productive cough without fever, intermittent lower abdominal pain, and irregular vaginal bleeding. Also, the bimanual examination revealed a uterus as large as 16 weeks of gestation.
The hCG level was 1,975,255 mIU/mL, and pelvic and abdominal ultrasounds revealed a mixed hypervascular mass in the uterine fundus, measuring 10 × 5 × 5 cm. Chest radiography showed more than 9 - 10 bilateral pulmonary nodules without evidence of brain metastasis in magnetic resonance imaging. Based on the mentioned findings, the patient was diagnosed with metastatic CC stage III, according to the International Federation of Gynecology and Obstetrics staging system and as a high-risk patient, according to the World Health Organization scoring system. Consequently, multi-agent chemotherapy with etoposide, methotrexate, and actinomycin (EMA) was initiated.
After the first day of administering EMA, the patient presented with increasing abdominal pain, associated with diarrhea, fatigue, dyspnea, and persistent coughs, without fever or chill. The vital signs included the pulse rate of 104 bpm, respiratory rate of 24 bpm, blood pressure of 100/70 mmHg, the body temperature of 37ºC, and oxygen saturation of 96% (4 L of nasal oxygen). Her abdomen was distended with generalized tenderness to palpation without vaginal bleeding. The results of laboratory tests, including complete blood count, were as follow: hemoglobin level of 8 g/dL, leukocyte count of 16 900, 90% neutrophils and 10% lymphocytes, lactate dehydrogenase of 1085, creatinine of 0.8 mg/dL, and C-reactive protein of 3+.
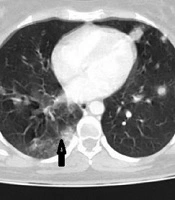
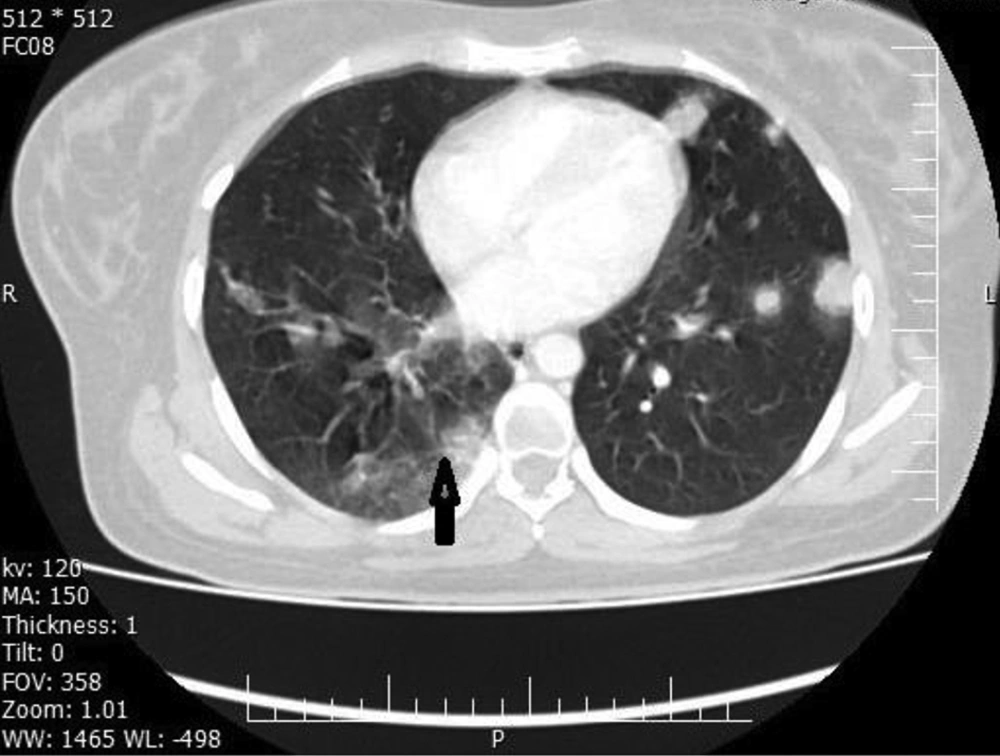
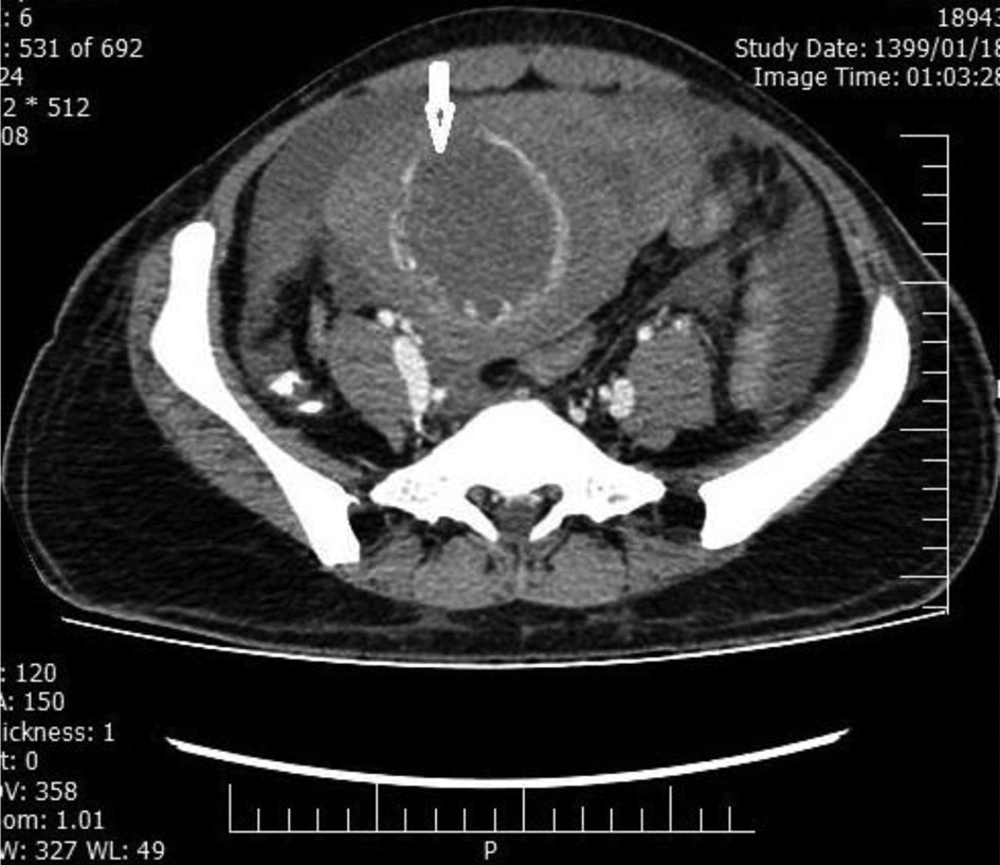
Considering the ongoing global pandemic of COVID-19 and the patient’s symptoms, to protect our team against COVID-19 and manage this emergency condition in the operating room, we resuscitated the patient with 2 L of IV normal saline and 2 units of packed red cells. A throat swab test for COVID-19 was also performed. Moreover, computed tomography (CT) scans of the chest, abdomen, and pelvis with double contrast were acquired by a radiologist. The results showed multiple well-defined hypodense pulmonary nodules due to metastasis, ground-glass opacity at the apical segment of the right lower lobe in favor of COVID-19-related pneumonia, a large hypodense, heterogeneously ring-enhancing, expansile mass, distending the endometrial cavity; and necrosis with uterine rupture and free fluid (Figures 1 and 2).
Despite the patient’s instability and chest CT evidence of COVID-19 infection, she was transferred directly to the operating room for emergency laparotomy, using the personal protective equipment for COVID-19. The abdomen was opened and a hemoperitoneum volume of 2300 mL was evacuated. The uterus was enlarged to an 18 weeks’ gestational size and there was a ruptured area with tumor tissue on the uterine fundus; there was no evidence of liver or other visceral metastases. A total abdominal hysterectomy was carried out under emergency conditions as a lifesaving procedure.
The isolated intensive care unit room was booked for the patient’s recovery, and conservative treatment of COVID-19 included oxygen therapy, hydroxychloroquine, azithromycin, and Kaletra (200 mg of lopinavir co-formulated with 50 mg of ritonavir). The patient was discharged from the hospital after 8 days without respiratory complications due to COVID-19; quarantine and self-isolation were advised for the patient. After 2 to 3 weeks of recovery, based on the suggestion of the infectious diseases specialists, chemotherapy was initiated. She has received 1 course of chemotherapy after surgery without any complications, which will continue for 3 consecutive courses until hCG reaches the normal range.
Written informed consent was obtained from the patient.
3. Discussion
CC should be suspected when there is persistent uterine bleeding following pregnancy. It is a rare neoplasm characterized by rapid growth, invasion into the myometrium, and distant metastasis (4, 5). A theory of intratumoral bleeding suggests the invasion of trophoblastic cells into vessels leading to thrombosis, infarction, and formation of vascular aneurysms (5, 6). Another possible etiology is a myometrial invasion and rapid necrosis of tumor cells due to chemotherapy leading to uterine perforation (7). Even after successful chemotherapy and a negative hCG titer, if the necrotic area and healing defect are significant, they may cause a spontaneous uterine rupture in the future (7). Nonetheless, hemoperitoneum secondary to CC is rare (8).
Emergency cases of active intraabdominal bleeding usually require hysterectomy as a lifesaving procedure (7-9). Fertility-sparing uterine lesion resection and uterine reconstruction for young women, who desire pregnancy, are preferred when the lesion is confined to the uterus without metastasis (4, 7, 10, 11). In hemodynamically stable patients, selective embolization of the main pelvic blood supply to the tumor can be performed by interventional radiology. Successful pregnancy after conservative surgery has also been reported (4, 12, 13).
In this case report, the patient presented with CC in an emergency setting due to COVID-19 infection (positive RT-PCR and CT results). Although the majority of COVID-19 cases are mild (14), our patient was at high risk of infection because her immune system was compromised due to cancer and previous chemotherapy, and surgery could not be postponed. Overall, these patients are at high risk of complications associated with COVID-19; therefore, it was difficult to choose between conservative management and surgery. In this case report, the main causes of hysterectomy included the heavy burden of tumor in the ruptured area, metastatic disease, and unpredictable conditions due to COVID-19.
3.1. Conclusions
Since CC is a chemosensitive tumor, conservative surgery should be considered in the setting of low-risk tumors. Due to the outbreak of the new coronavirus disease with serious impacts on the treatment of patients with cancer, it is suggested to perform optimal surgery in a high-risk emergency setting to prevent further complications, especially when hospital facilities are inadequate due to the COVID-19 pandemic.