1. Background
2. Objectives
3. Methods
3.1. Study Population
3.2. Sample Preparation
3.3. cDNA Synthesis and qRT-PCR Assay
3.4. Statistical Analysis
4. Results
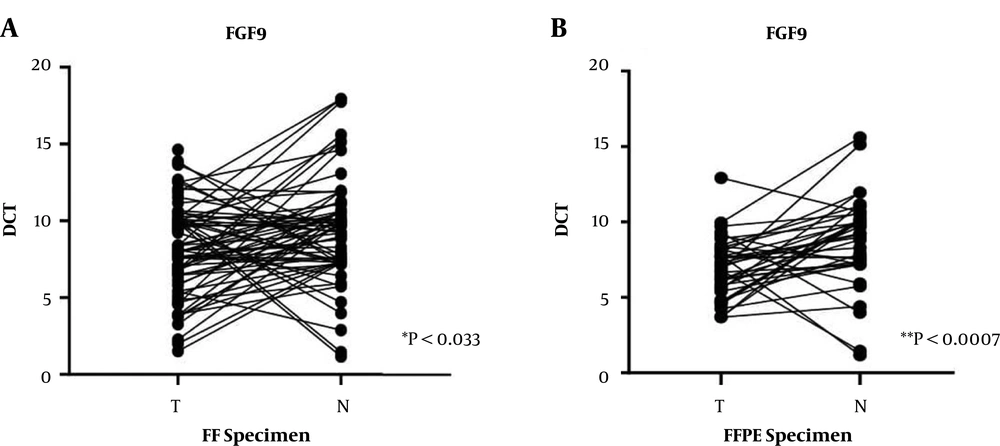
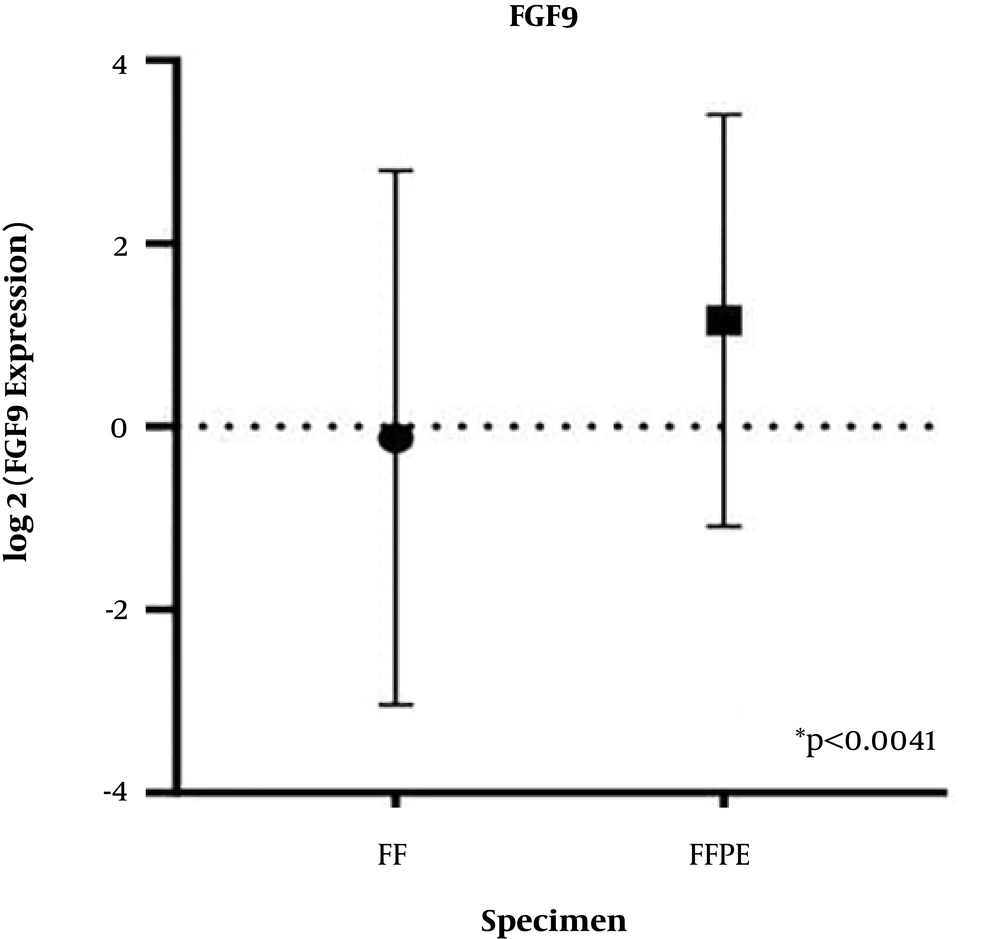
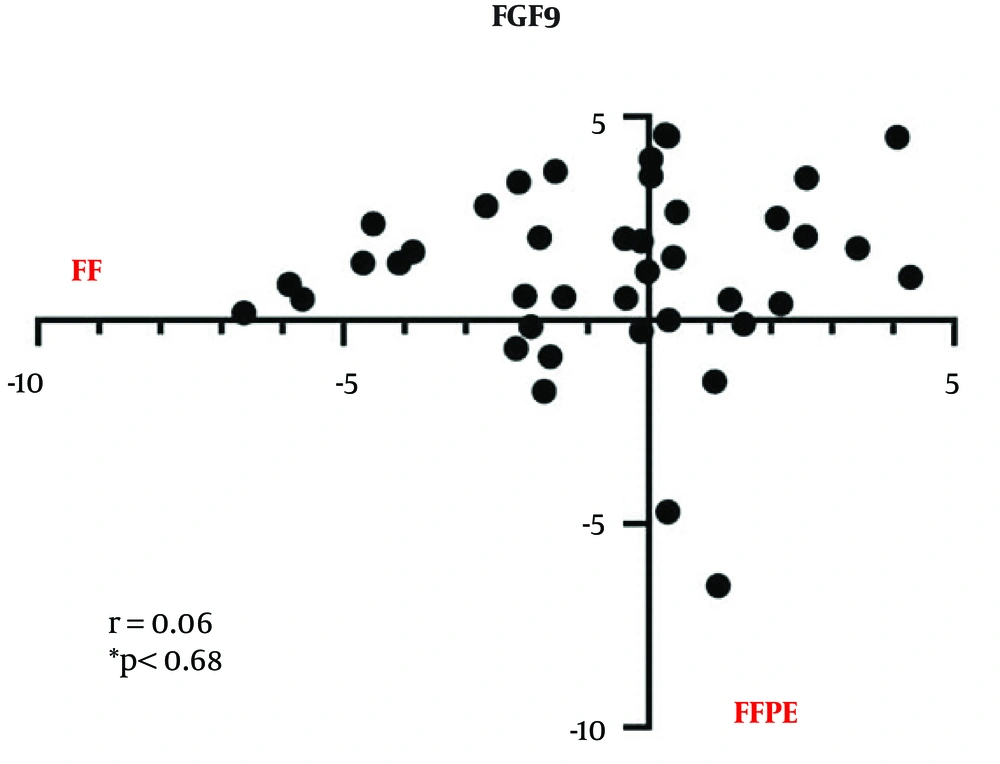
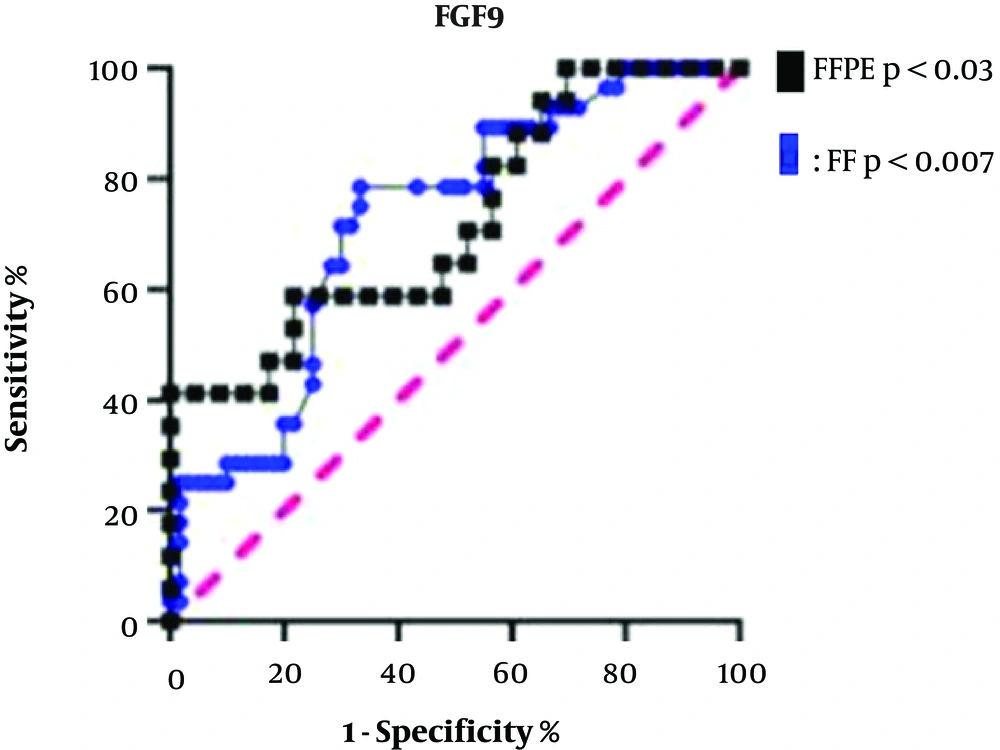
4.1. FGF9 Upregulation in CRC Patients
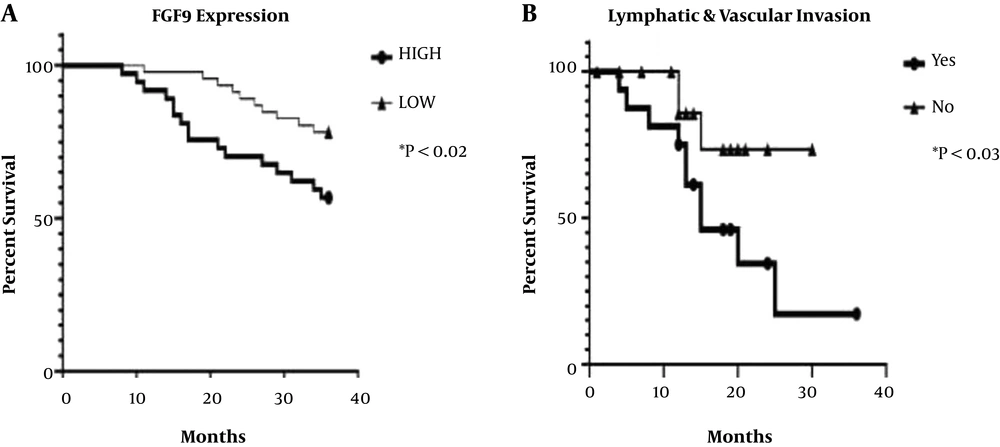
4.2. Association of FGF9 Expression with the Clinicopathological Data
| Parameters | FFPE Samples, Mean ± SD of RQ | P-Value | FF Samples, Mean ± SD of RQ | P-Value |
|---|---|---|---|---|
| Sex | 0.62 | 0.0052 | ||
| Male | 5.37 ± 4.22 | 0.73 ± 1.002 | ||
| Female | 3.87 ± 3.89 | 0.68 ± 0.66 | ||
| Age | 0.81 | 0.01b | ||
| ≤ 50 | 4.38 ± 4.20 | 0.47 ± 0.52 | ||
| > 50 | 6.26 ± 4.51 | 0.83 ± 0.98 | ||
| Tumor size | 0.71 | 0.56 | ||
| ≤ 5 | 2.84 ± 2.91 | 0.69 ± 0.89 | ||
| > 5 | 6.51 ± 5.00 | 0.96 ± 1.10 | ||
| TNM stage | 0.003b | 0.013b | ||
| I and II | 2.61 ± 2.71 | 0.66 ± 0.72 | ||
| III and IV | 8.25 ± 8.02 | 0.79 ± 1.15 | ||
| Lymphnode metastasis | 0.047b | < 0.0001b | ||
| No (N0) | 2.57 ± 2.76 | 0.10 ± 0.24 | ||
| Yes (N1, N2, N3) | 8.45 ± 8.26 | 0.78 ± 1.17 | ||
| Vascular invasion | 0.047b | 0.004b | ||
| No | 2.85 ± 2.91 | 0.61 ± 0.73 | ||
| Yes | 9.01 ± 9.62 | 0.85 ± 1.25 | ||
| Differentiation | 0.63 | 0.36 | ||
| Well + moderate | 4.75 ± 3.87 | 0.67 ± 0.78 | ||
| Poor | 7.60 ± 6.50 | 0.87 ± 1.03 |
Abbreviations: AUC: area under curve; cDNA, complementary DNA; CRC, colorectal cancer; FF, fresh frozen; FGF: fibroblast growth factor; FGF9, fibroblast growth factor 9; FFPE, formalin-fixed, paraffin-embedded; LncRNA, long non-coding RNA; ROC, receiver operating characteristic; TNM, tumor, node, metastasis.
aMann-Whitney U-test.
bSignificant at P < 0.05.