1. Background
About seven million people in the United States suffer from full-thickness skin wounds each year for various reasons including burns, surgery, and trauma (1). The treatment of these wounds imposes a huge cost on the health system and causes death and disability of patients (2). Prompt and proper treatment of these wounds reduces the complications, but in some complications, such as infection and hematoma of the surgical site or tension on the suture line, primary wound healing fails and secondary intention wound healing is required. This occurs especially in patients with cancer who received chemoradiation before surgery. In these cases, the wound healing process is very slow and the infection rate is high (3, 4). Therefore, different methods and tissues have been introduced to accelerate the wound healing process such as stem cells and vacuum sealing techniques (5), but the high cost of these materials prevented them from becoming widespread. On the other hand, the use of the human amniotic membrane (AM) is considered by researchers due to its unique properties such as accelerating the process of epithelialization and reducing bacterial colonization (6, 7) and it has been proven to have very promising results in burn wounds (8, 9). Amniotic membrane has been used to treat such wounds for almost 100 years. The amniotic membrane is full of collagen and also full of growth factors, which accelerate the healing process and reduce scar formation (10). Previous studies have mostly used wet AM which has been difficult to maintain and prepare. Therefore, the production of dehydrated (dry) AM was considered. Limited studies have shown the effect of dry AM on the healing process of burn wounds and chronic diabetic wounds without any significant complications. Patients who were treated with dehydrated human amniotic membrane allografts had better outcome versus standard treatment (10-12). Few studies have been carried out on surgical wounds and trauma but none of these studies used dry AM. (13) The cost of this tissue is lower than other pre-prepared tissues and is always available and easy to prepare, so it seems that dry AM is a good choice for dressing such wounds (14).
2. Objectives
The aim of this study was to investigate the effect of dry AM on accelerating the healing process of full-thickness skin wounds in patients who underwent urological cancer surgery.
3. Methods
3.1. Patients
After receiving IRCT id (IRCT20180218038788N2) for this study, a total of 40 patients from January 2018 to July 2020 who underwent urological cancer surgery in Imam Reza and Shohada-Tajrish Hospital, Tehran, Iran, enrolled in this study. Secondary intention wound healing was considered for the patients. Then, the patients were randomly assigned into 2 groups (20 patients in each group) using a random number table and allocation was concealed by sealed envelopes.
3.2. Inclusion and Exclusion Criteria
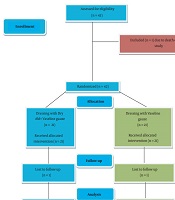
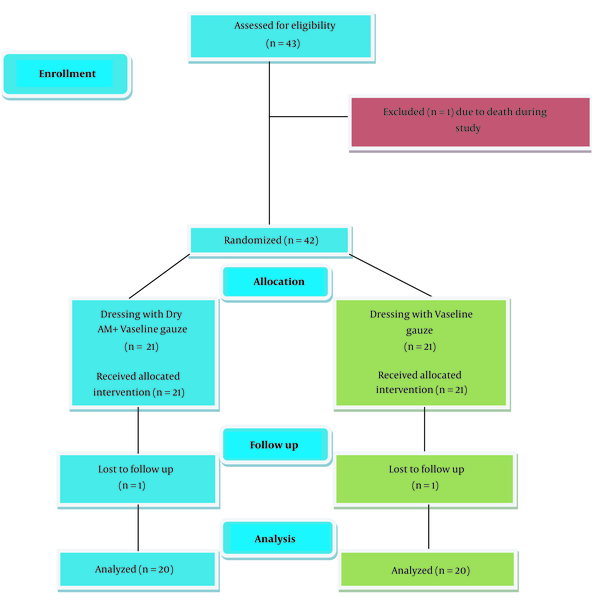
Patients older than 40 years old who underwent urological cancer surgery and their primary wound closure failed due to infection and hematoma of the surgical site or tension on the suture line, and secondary intention wound healing chosen for them, included in the study (43 patients). Patients with active wound infection received proper antibiotics based on the wound culture and after eradication of infection, which proved by negative wound culture, enrolled in the study, and patients who were expired during hospitalization (1 patient) or lost their follow-up (2 patients) were excluded from the study. Figure 1 shows the case selection process.
3.3. Procedure and Follow-up
After observing the absence of active wound infection in the case group, according to the size of the wound, a dry AM (Regen Membrane) prepared by the Iranian Tissue Product Company, was selected (15). According to the instruction of the company, bedside preparation was done in a sterile manner by soaking AM in sterile water for less than 5 minutes and applying it to completely cover the bed and the wound margins. Then dressing in vaseline gauze was done. In the control group, the dressing was done in the same manner without AM. During hospitalization and after discharge, the dressing was changed daily till wound healing occurred. It should be noted that in the case group, AM was used for the first dressing, and after that dressing was done without it. During hospitalization, if there was any evidence of wound infection such as a foul odor, abnormal discharge, and the like a wound culture was sent, and if more than 100,000 of the same type of microorganism grew, patients were given antibiotic therapy based on the antibiogram. In the end, any wound infection, sepsis, reoperation, and duration of hospitalization were recorded. The degree of patients’ pain was recorded based on a visual analog scale (VAS) after each change of dressing. After discharge, patients were visited in 2 weeks, 1 month, 3 three months after the first dressing. The wounds' condition was examined and finally, the duration of complete was wound healing recorded.
3.4. Study Endpoints
The primary endpoints of this study were the duration of hospitalization and the time required for wound epithelialization, and the secondary endpoint was VAS, wound infection, sepsis, and reoperation of patients during hospitalization.
3.5. Statistics
In this study, Statistical Package for the Social Sciences (SPSS) 21 software was used to analyze the data. Quantitative data were reported based on the mean ± standard deviation and qualitative data were descriptively expressed. In cases where we have compared the 2 groups, we used the independent t-test and chi-square method. P-value < 0.05 was considered as a statistically significant level.
3.6. Ethics
The ethics committee of AJA University of Medical Science, Tehran, Iran, approved this pilot study in February 2019 (code: IR.AJAUMS.REC.1399.053) and all the patients entered the study with informed consent.
4. Results
Each group consisted of 18 males and 2 females. The mean age of patients in the case group was 57.9 ± 9.3 years and the mean age of the control group was 54.7 ± 9.5 years, so there was no significant difference between the 2 groups (P-value = 0.448). We matched the patients in the terms of the wound site and type of the surgery (Table 1).
Based on the underlying disease, the patients of the 2 groups had slight differences that were not significant (P-value > 0.05 ) and the most common underlying disease in both groups was diabetes mellitus (10 patients in the case group and 12 patients in the control group). Table 2 shows the underlying diseases of both groups.
During hospitalization, wound infection was more common in the control group (4 vs. 1) but was not significantly different (P = 0.34) and all of them received proper antibiotics based on the antibiogram and were treated.
The same results apply to the rate of sepsis and reoperation between the 2 groups. The number of patients with sepsis or in need of reoperation was higher in the control group compared to the case group, but there was no significant difference between the 2 groups. Table 3 shows the complications during hospitalization in both groups.
| Comlication | Groups | P-Value | |
|---|---|---|---|
| Case | Control | ||
| Sepsis | 0 (0) | 2 (10) | 0.48 |
| Reoperation | 2 (10) | 5 (25) | 0.4 |
The Number of Patients Who Developed with Sepsis or Needed Reoperation During Hospitalization in Both Groups Separately a
The mean hospital stay in the case group was 4.8 ± 1.1 days (3 - 7 days) and in the control group was 8.6 ± 2.1days (3 - 16 days) which was almost double and this difference was significant (P-value = 0.0) and as we expected, patients who developed with wound infection, sepsis, or need reoperation hospitalized for a longer time in both groups.
The average of VAS in the case group was significantly lower than the control group (P < 0.05) in the first 4 days. After that, the average pain score was not significantly different between the 2 groups. Table 4 shows the average of VAS in the first 5 days of dressing between the 2 groups.
| Day | VAS in Case Group | VAS in Control Group | P-Value |
|---|---|---|---|
| 1 | 4.85 ± 1.2 | 7.2 ± 1 | 0.00 |
| 2 | 4.5 ± 1.1 | 6.35 ± 0.9 | 0.00 |
| 3 | 4.5 ± 0.94 | 5.2 ± 0.89 | 0.02 |
| 4 | 2.9 ± 0.86 | 4.2 ± 0.89 | 0.00 |
| 5 | 2.5 ± 0.65 | 2.3 ± 0.8 | 0.9 |
Comparing the Mean VAS in the First 5 Days of Dressing Between the 2 Groups a
The time required for wound epithelization was 9.1 ± 2.2 days (12 - 46 days) in the case group and in the control group was 37.8 ± 12.8 days (15 - 68 days) and the P-value between the 2 groups was 0.0 and like hospitalization, epithelialization in patients who developed with wound infection or sepsis took longer.
5. Discussion
In most cases, surgical wounds are repaired primarily, but in some cases wound repairing is challenging for surgeons. In some patients especially in patients with cancer who received neoadjuvant chemoradiation (4) due to infection or hematoma of the surgical wound or excessive tension on the suture line primary intention wound healing was failed (16) so the secondary intention is selected. In these circumstances, the granulation tissue is allowed to fill the skin defect from the base of the wound (16).
This method increases the complications and duration of wound healing and morbidity of patients that all of these increased medical expenses (17) so new methods or materials are needed to accelerate the healing of these wounds.
Fresh human AM is one of these tissues that has been proven to be effective in wound healing, especially in burning and chronic wounds (18) but it has some drawbacks like unavailability and special storage conditions so tissue engineers produced the dehydrated (dry) type of AM that includes cellular scaffold without cell that has all advantages of fresh AM without its drawbacks (19).
Nowadays, dry human AM is used for burning and chronic wound healing (20, 21), but no study have shown the effect of dry human AM on post-cancer surgery full-thickness wounds, so we conducted this study to evaluate this.
The prevalence of surgical site infection can be up to 20% (21) and this prevalence is much higher in secondary intention wound healing, especially in patients with cancer because these patients usually have an underlying disease (4). The results of this study showed that wound infection in the case group is lower but not significant (5 vs. 20%, P = 0.34). This number was 30% in the case group and 53% in the control group in the study by Andonovska et al. (6). According to the findings of other studies, using dry AM reduced the complication and the reoperation rate (6, 22) and it is due to the anti-inflammatory properties of AM (18). We showed that in the case group the pain of patients is much lower during dressing which is in line with the findings of other studies especially in burn wounds (23). Eskandarlou et al. reported that in burn patients, the average pain score in the first 4 days after dressing with AM is much lower but after that, till 14 days the pain score is almost equal (24). This unique ability of AM to reduce pain is due to the soft mucoid lining of AM that covered the exposed nerve ending and prevented stimulation of it by an irritant (18) so it decreased morbidity of patients and decreased use of painkillers.
AM promotes wound healing by activation of cytokines, growth factors, stem cells, and mesenchymal cells (19). Different studies have reported different duration for wound healing using the AM, but studies of acute wounds and burns have reported a longest wound healing time of 4 - 12 weeks (25) and for chronic wounds, this time was up to 29 weeks (20, 26). Therefore, since our study was performed on acute wounds, patients were followed up for up to 3 months. The required time for wound healing in this study in the case group was much shorter than the control group (9.1 ± 2.2 vs. 37.8 ± 12.8 days, P-value = 0.0), and as we expected, the use of the amniotic membrane accelerated the wound healing process after surgery. However, the difference between this study and other studies was that this study was performed on full-thickness wounds of the skin, while other studies were performed on partial thickness wounds (27). In limited studies on full-thickness wounds, dry human AM was not used (28) or the wounds were not surgical (6, 23, 29). We expected that due to the positive effects of the AM on wound healing, which showed in earlier studies, the length of hospital stay in these patients reduced. In this study, the length of the hospital stay was calculated and as a result, we found that the length of hospital stay in the case group was much shorter than the control group. Based on our literature review, this variable was evaluated for the first time in our study. Since the AM reduces the rate of complications, duration of hospitalization, and wound epithelization, it reduces the number of surgeries and dressings and the amount of drug usage including antibiotics and painkillers, so the treatment costs are reduced directly and indirectly.
5.1. Conclusion
Finally, we concluded from this study that dry human AM is a safe, efficient, and applicable option to accelerate the process of secondary healing of surgical wounds and in addition can reduce wound complications, duration of hospitalization, and treatment costs.
We acknowledge that our sample size was small, since dry human AM was used to repairing surgical wounds for the first time in this study. According to the findings of the current study, we can claim that dry human AM can accelerate the healing process of this type of wound, but more studies with more sample size are needed to make a definite statement.
Another limitation of our study was that in cases where the patients who underwent surgery by different surgeons in 2 hospitals and had different surgical procedures, we had to be informed that the dressing was done by the same team, also the same team followed the patients.