1. Background
Ovarian cancer is the most common malignancy of all gynecologic cancers and the 7th most common type of cancers in women in the United States (1, 2). The annual incidence of ovarian cancer is estimated more than 200000 cases and 100000 women die from ovarian cancer each year around the world (3). In Iran, the age standard incidence rate of ovarian cancer has been reported as 3.1 to 3.9 per 100000 women (4, 5). The prognosis of ovarian tumors highly depends on the stage at first diagnosis and treatment. Most cases (75%) of ovarian cancer initially present with stage III or higher in the International Federation of Gynecology and Obstetrics (FIGO) staging system 2014 (6). The 5 - year survival for early stages is relatively high (87% for stage IA), but dramatically declines as the stage advances (41% for stage IIIA and 11% for stage IV). Overall survival rate for ovarian epithelial malignancies has been estimated as 46% (3).
The treatment modality of choice for ovarian cancer is cytoreductive surgery, followed by combination chemotherapy. Studies have shown that regardless of the cancer stage, post-operative response to chemotherapy is favorable (7). However, with optimal excision of the tumor mass, there is considerable chance of tumor relapse (8, 9).
Several studies have been conducted to introduce new mechanisms to increase the sensitivity of remaining tumor cells to chemotherapy. Several modifications such as genetic manipulations, immunotherapy, and addition of other chemical have been evaluated in various types of malignancies to enhance neoplastic cell sensitivity to chemotherapy (10). Among these, metformin an anti-diabetic agent from biguanides family has been widely studied for its effect on malignant cell response to chemotherapy (11-13). Researchers have reported that in vitro administration of metformin has significantly reduced cell proliferation in ovarian, prostatic, and breast neoplastic cell lines (11). The potential efficacy of metformin in the prevention and treatment of cancer was first reported in retrospective epidemiologic studies on the prevalence of cancer in diabetic patients. They succeeded to demonstrate the significantly lower incidence and cancer - related mortality in diabetic patients receiving a daily dose of 1500 to 2100 mg of metformin (14-16). Cellular and molecular effect of metformin has been linked to the activation of AMP - activated protein kinase pathway (AMPK). AMPK acts as a major cellular energy sensor, which negatively regulates metabolic pathways such as glucose consumption and fatty acid oxidation (17). Other mechanisms have been proposed for the anti-cancer potential of metformin, such as S-phase cell cycle arrest, reduced proliferation and colony formation, apoptosis induction, and reduction in insulin - like growth factor 1 receptor (18).
In 2012, two retrospective cohort studies (19, 20) reported significantly higher survival and cancer control in diabetic patients receiving metformin versus non-diabetic patients and diabetics not taking metformin, who had undergone cytoreductive surgery for ovarian epithelial cancer. These retrospective studies investigated clinical records of ovarian cancer patients and compared the 5 - year survival from ovarian cancer between the group who received metformin (case) and those who had not received metformin (control). Although mentioned studies included large study populations, the retrospective method restricted investigation of the effect of metformin receipt in non-diabetic patients with ovarian cancer who underwent chemotherapy. In the present randomized controlled trial, we studied the efficacy of metformin in combination with standard chemotherapy in improving survival and minimizing the relapse rate of ovarian cancer in non-diabetic patients.
2. Methods
This study was registered at Iranian registry of clinical trials with the code IRCT2016022726788N1.
Procedure and study protocol was approved by Medical Ethics Committee of Shiraz University of Medical Sciences with reference number 153/11/41/9p.
2.1. Study Population
This study was conducted as a randomized controlled trial in Shahid Faghihi Hospital, affiliated to Shiraz University of Medical Sciences between March 2011 and March 2013. All patients, who underwent total abdominal hysterectomy and bilateral salpingo - oophorectomy (TAH-BSO), whose pathologic evaluation of the ovarian mass confirmed the diagnosis of epithelial ovarian cancer, were invited to participate. Patients with following criteria were excluded from the study:
●Diabetic patients
●Patients with liver or gastrointestinal disorders
●Patients with renal failure
●Patients unable to tolerate metformin
●Patients in whom ovarian cancer is secondary to another cancer
●Patients who received neoadjuvant chemotherapy prior to cytoreductive surgery
After obtaining informed consent, patients were randomized in study and control groups, using an online random number generator at a ratio of 1:1.
2.2. Study Protocol
Patients with initial complaints suggestive of ovarian tumor such as pelvic pain, abdominal fullness, weight loss, vaginal bleeding, or other non-specific systemic symptoms, whose ultrasound and CA-125 levels confirmed the diagnosis of ovarian cancer, were included in this study. Initial laboratory work-up, including complete blood count (CBC), liver function tests, BUN, and Creatinine levels were performed. After pre-operative evaluations such as cardiologic and anesthesiology examinations, patients were candidate for total abdominal hysterectomy and bilateral salpingo - oophorectomy.
Under general anesthesia, in dorsal supine position vaginal examination was performed to confirm previous findings. After the placement of a Foley catheter, patient’s legs were straightened. Iodine solution was applied to sterilize the incision site. Vertical abdominal incision was made 3 cm above the umbilicus. After entering the peritoneal cavity, systematic exploration of intra-abdominal organs such as liver, gall bladder, stomach, kidneys, small and large bowel, and para-aortic lymph nodes were examined for the presence of any metastatic lesion. The internal reproductive organs, including uterus, ovaries, fallopian tubes, and pelvic lymph nodes were visualized. Before the initiation of the resection process, peritoneal lavage was performed and samples were taken for cytologic evaluation. The uterus along with both fallopian tubes and ovaries were excised after cautious detachment of surrounding structures and vasculature. Omentectomy and para-aortic lymph node biopsy was performed. Optimal debulking was done; the goal of optimal debulking is to reduce the tumor residual < 1 cm.
Vaginal cuff was closed by absorbable suturing materials. Surgical field was irrigated and hemostasis was ensured. Peritoneum and abdominal wall layers were re-approximated. After the surgery, samples from ovarian lesions were sent to pathology lab for histopathological identification of the tumor type.
After surgery, the patients were divided in two groups as a case and control by simple random sampling method.
A 500 mg daily dose of metformin was started (gradually increasing to 1500 mg daily divided 3 times a day within a week) in addition to standard carboplatin - paclitaxel regimen (The dose of carboplatin is calculated, by using the area under the curve [AUC] and the glomerular filtration rate [GFR] according to the Calvert formula and paclitaxel was administrated by dosage (175 mg/m2 over 3 hours).
Staging of ovarian cancer was performed according to FIGO 2006 classification of ovarian cancers (21). Patients with low grade malignancies (stage I and II) received 3 to 6 courses of chemotherapy. The number courses for the case of high grade tumor (stage III and IV) varied between 6 and 9. Chemotherapy courses were administered in a monthly pattern (each 21 days). Metformin therapy was given as long as chemotherapy was needed. After each chemotherapy period, ultrasound evaluation was performed by the same radiologist, and CA-125 levels were measured by the same reference lab. After each session of chemotherapy, abdominal and pelvic exam was performed by the same gynecology resident. Relapse of the tumor was considered as elevation of serum CA-125 levels higher than 35 U/mL or recurrence of a pelvic mass in ultrasound or physical examination. During each course of chemotherapy, laboratory evaluations, including CBC, BUN, Creatinine, and liver function test were performed. Occurrence of any abnormality in these tests resulted in immediate exclusion of the patient and termination of the prescriptions. Also those patients, who suffered from gastrointestinal discomfort caused by metformin, were requested to terminate taking metformin and were excluded from the study.
After chemotherapy courses were completed, follow-up visits were performed every 3 months until 2 years and every 6 months after that. On each follow-up visit, serum levels of CA-125 marker were checked as well as ultrasound imaging and pelvic examination performed by gynecology resident.
During the follow-up period, microscopic recurrence defined by isolated rise in CA-125 level, or macroscopic recurrence defined by detecting a mass in pelvic examination or ultrasound imaging was recorded and labeled as relapsed carcinoma.
The 4 - year rate of cancer relapse and survival was compared with the age and primary stage equalized control group. Also, the pattern of CA-125 level decline during the total chemotherapy courses was compared among the group, who received metformin during chemotherapy and the control group.
2.3. Statistical Analysis Method
Statistical analysis was performed, using Chi-Square and independent t test for equality of means of the Statistical Product; survival analysis was used to evaluate the recurrence of tumor and Service Solutions (SPSS) 18, IBM corporations, Armonk, New York, U.S. P value < 0.05 was considered significant.
3. Results
Study population size was determined, using G*power software. Considering a power of 80% and P = 0.05, a sample size of 41 patients in each control and intervention group was assigned. Although, we included all patients, who met the inclusion criteria and accepted the terms and conditions of the study during the 2 - year period of patient recruitment; the calculated sample size for study groups were not achieved.
Medical information from 70 patients were recorded and analyzed in this study. The study and control groups consisted of 30 and 40 patients, respectively, aged between 30 and 80 years old. Statistical analysis for comparison of influencing factors, including age, tumor histologic type, stage and grade, and number of previous pregnancies between groups revealed no significant difference. Following tables describe these results (Tables 1 and 2).
| Group | Minimum Age | Maximum Age | Mean Age | P Value |
|---|---|---|---|---|
| Metformin + Chemotherapy | 36 | 80 | 49.7 | 0.654 |
| Chemotherapy | 30 | 62 | 47.5 |
| Variable | Frequency in Case Group | Frequency in Control Group | P Value |
|---|---|---|---|
| Tumor type | 0.543 | ||
| Serous | 24 | 25 | |
| Mucinous | 4 | 2 | |
| Endometrioid | 2 | 3 | |
| Tumor stage | 0.686 | ||
| I | 8 | 7 | |
| II | 2 | 2 | |
| III | 20 | 21 | |
| Tumor grade | 0.622 | ||
| I | 7 | 7 | |
| II | 2 | 4 | |
| III | 21 | 19 | |
| Menstrual cycle | 1 | ||
| Normal | 2 | 3 | |
| Abnormal | 28 | 27 | |
| Gravid | Mean : 4.1 | Mean : 4.7 | 0.553 |
As indicated in Table 2, the most prevalent tumor in both groups type was papillary carcinoma (83%). Most of the patients (70%) were presented at stage III of ovarian cancer, and the most tumor grading was grade 3 (63%).
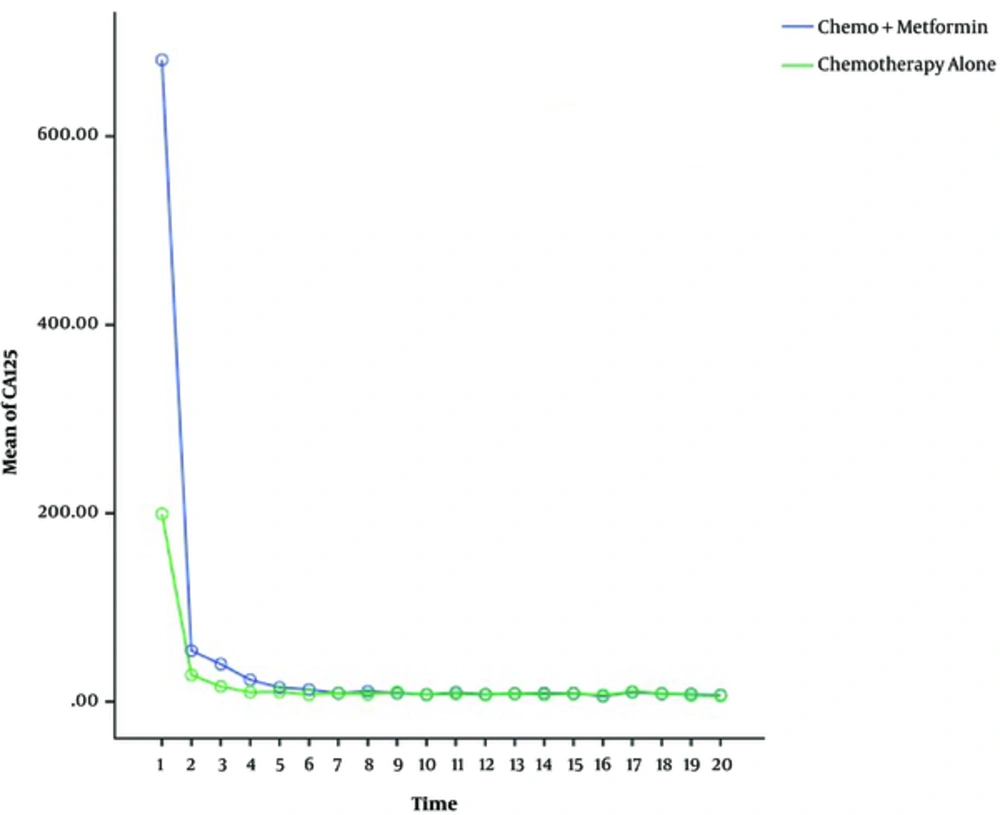
Statistical analysis for evaluation of variation among two groups regarding the pattern of CA-125 decline was performed by general linear model for repeated measures. With P value = 0.332, the difference among two groups was not significant.
Statistical analysis regarding CA-125 level decline within each group was significant (P = 0.000) for both case and control groups, indicating that the decline of CA-125 levels was significant during chemotherapy (Figure 1).
We determined recurrence of the tumor within 4 - year follow-up after chemotherapy (with or without metformin) by definition of:
CA-125 level significant rise
Ultrasound evidence of lymph node, liver, or pelvic mass
Detection of lymph node, vaginal lesion, or pelvic mass in pelvic exam
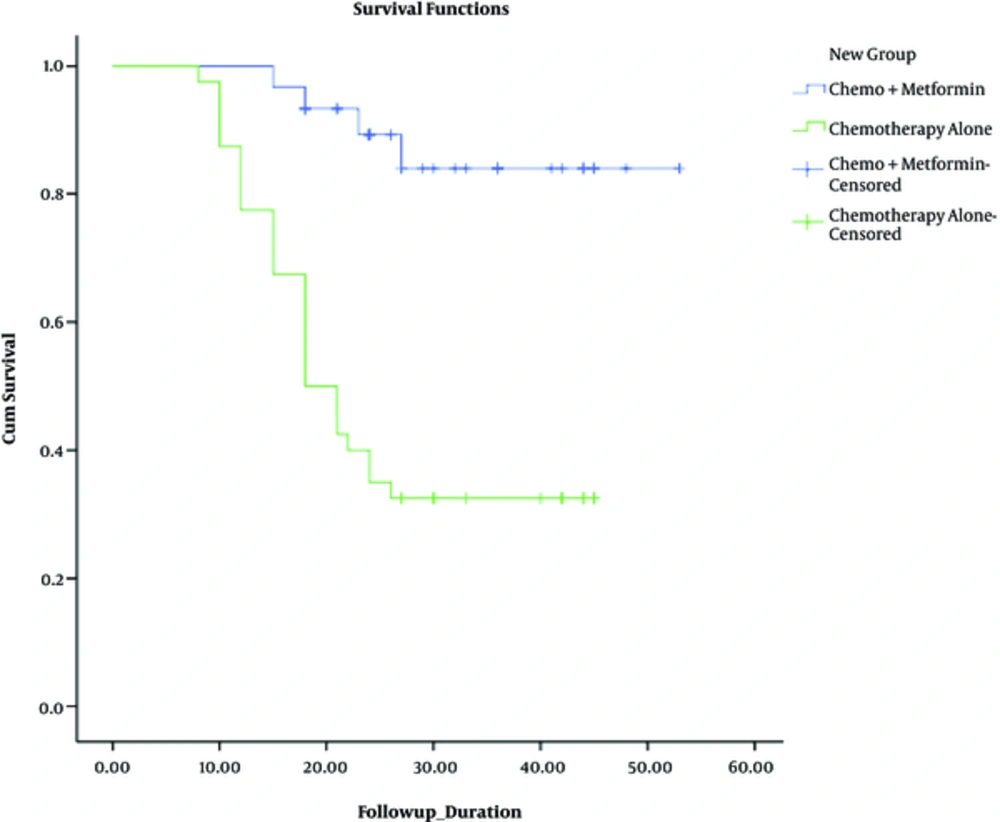
The isolated elevation of CA-125 levels was labeled as microscopic recurrence and mass detection in ultrasound or examination regarded as macroscopic recurrence. Among 30 patients of the group treated with metformin during chemotherapy, 4 - year rate of recurrence was 13.3% (4 patients). Within the control group, 27 out of 40 patients developed recurrence of malignancy (67.5%) (Figure 2). This difference was statistically analyzed by Chi-square test. With P value = 0.000, the difference was statically significant.
The mean survival of patients, who used metformin, was 48 months (with 95% CI: 43.4 - 52.5), while the mean survival who received chemotherapy alone was 25.7 (with 95% CI: 21.3 - 30).
Another factors, which were significantly predictive of recurrence and included in this study, were FIGO stage (P = 0.005) and tumor grade (P = 0.029), while the histology of tumor was not a significant predictor of recurrence.
Adverse effects of metformin such as abdominal discomfort, nausea/vomiting were evaluated within the study group. None of the patients, who received metformin, had any complaint of nausea/vomiting or developed signs of hypoglycemia. Only 6 patients complained about abdominal discomfort, which comprised 20% of the study population.
Other possible side effect of using metformin, which was evaluated in this study, was weight loss. To detect the occurrence of this adverse effect, weight of patients in both groups was recorded before and after the treatment. An average weight gain was detected in both groups. The mean value of weight gain during therapy was 5.5 kg and 2.7 kg in case and control groups, respectively. Statistical analysis by independent t test showed that this difference was significant (P = 0.000).
4. Discussion
Even with full remission from primary ovarian epithelial cancer after successful cytoreductive surgery and chemotherapy, recurrence of the disease occurs in about 60% to 70% of cases (22, 23). In the present study, we aimed at evaluating the efficacy of additional metformin administration to standard post-surgical chemotherapy. Metformin is an anti-diabetic agent, which has been used for the management of diabetic patients around the world. After publication of studies, which revealed significantly lower incidence of cancers among diabetic patients who received metformin compared to diabetics on other anti-diabetic treatments (14), metformin was investigated for its new potential in treatment and prevention of cancer. Bimolecular and cellular evidences have been published on animal models and human cancer cell lines that revealed metformin can inhibit growth and development of tumor cells (24). Various types of cancers and cell lines have been studied for the suppressive effect of metformin on tumor growth and cell proliferation such as mammary tumors in HER-2/neu transgenic mice (25), in mouse tobacco induced lung carcinogenesis (26), and breast/pancreas cancer in animal models (27).
In this case-control human study, on non-diabetic population, we compared a study population, consisting of 30 non-diabetic women with ovarian epithelial cancer who received a daily dose of 1500 mg metformin, divided 3 times a day during the courses of their chemotherapy with standard Carboplatin/Paclitaxel regimen with a case by case matched control group of ovarian epithelial cancer patients from October 2009 till October 2014, who received the same standard chemotherapy courses without combined administration of metformin. The results showed significantly lower rate of recurrence (13.3% vs 67.5% in the control group) within 3 to 4 years of follow-up. We are going to follow the patients till 5 years to compare the 5 - year recurrence rate of malignancy among the groups.
By the hypothesis that metformin increases the cytotoxic effect of chemotherapy agents on ovarian tumor cells, we expected to identify significantly faster pattern of improvement of serum CA-125 in the metformin treated group during chemotherapy courses, but due to small sample size and restrictions of patient recruitment, general linear tests could not be performed for our results. Although, it can be noticed that the regression line of CA-125 levels in the case group is located below that of the control group during the chemotherapy.
One of the mechanisms, by which metformin is thought to assert its anti-neoplastic and tumor - preventive effect, is its role in regulating cellular energy availability (28, 29). In a study by Phoenix et al., they evaluated the effect of metformin administration along with restricted, moderate, and high energy intake diet on murine primary and metastatic breast cancer invasion and growth. The results showed that dietary energy restriction achieved by metformin treatment significantly suppressed tumor growth and metastasis. This suppression is linked to reduced IGF-1 level and downstream signaling activities (29).
Other cellular metabolic mechanism of anti-cancer potential of metformin is found to be through the activation of liver kinase B1 (LKB1) and AMP-activated protein kinase (AMPK) signaling pathway. LKB1 is a regulator of gluconeogenesis in hepatic cells, but within the epithelial cells, it has previously been known as a tumor - suppressor gene; As in Peutz - Jeghers syndrome that impaired the function of LKB1, gene results in the development of multiple gastrointestinal polyps and significantly higher risk of various epithelial cancers (30). Metformin activates LKMB1/AMPK signaling pathway by inhibiting I of oxidative phosphorylation within mitochondria. The activation of LKMB1/AMPK signaling pathway down-regulates AKT/mTOR pathways. These intracellular alterations made by metformin, finally result in regulation of protein synthesis, fat metabolism, cell proliferation, and growth within tumoural cells (31, 32).
In 2011, Rattan et al. conducted a study for the first time, which showed significantly increased cytotoxicity of Cisplatin when administered in combination with metformin on ovarian cancer cell of murine models (13).
In a recent case - control human study, Kumar et al. performed a retrospective study, recruiting 72 cases consisted of diabetic patients, who received metformin monotherapy for their diabetes control and 143 randomly selected age-matched controls, all selected from women with epithelial ovarian cancer, who received standard chemotherapy after cytoreductive TAH-BSO surgery. The results of that study was highly suggestive for favorable efficacy of metformin combination therapy in minimizing the relapse rate of ovarian epithelial cancer, but confirmatory cohort prospective studies was suggested (19).
4.1. Conclusions
As a pilot study, our project was successful in revealing the surprising potential of metformin in the enhancement of efficacy of chemotherapy after cytoreductive surgery for ovarian epithelial cancer and reducing recurrence rate. We recommend further clinical investigations, including larger study populations and longer follow-up period to validate these results. Also, cellular studies on epithelial ovarian cancer cell lines can be performed, using case and control groups to compare cytotoxic effects of Carboplatin/Paclitaxel regimen with and without metformin treatment.