1. Background
2. Objectives
3. Methods
3.1. Patients and Sample Collection
| Variables | Benign (n = 20) | Malignant (n = 40) |
|---|---|---|
| Age (y) | ||
| > 20 | 4 (20) | 17 (42.5) |
| 20 - 40 | 8 (40) | 14 (35) |
| 40 - 60 | 6 (30) | 8 (20) |
| < 60 | 2 (1) | 1 (2.5) |
| Sex | ||
| Female | 12 (60) | 26 (65) |
| Male | 8 (40) | 14 (35) |
| Chemotherapy status | ||
| Chemotherapy + | 28 (70) | |
| Chemotherapy - | 12 (30) | |
| Grade | ||
| Low grade (grade I and II) | 25 (62.5) | |
| High grade (grade III and IV) | 15 (37.5) |
a Values are expressed as No. (%).
3.2. RNA Extraction
3.3. cDNA Synthesis and Real-time PCR
| Symbol | Gene Name HGNC | Product Length | Forward Primer (5’ → 3’) | Reverse Primer (5’ → 3’) |
|---|---|---|---|---|
| ACTB | BRWS1, PS1TP5BP1 | 137 | GATCTCCTTCTGCATCCTGT | TGGGCATCCACGAAACTAC |
| RIPK1 | IMD57, RIP, RIP-1, RIP1 | 178 | GTGCTGAAAGCCGAGATGAGTA | GCTTGTTTTGAGCTGTAGCCTG |
| RIPK3 | RIP3 | 167 | ATCCAGTAACAGGGCGACCG | TTGCGAACCTACTGGTGGGG |
| MLKL | Hmlkl | 109 | CTTTGAGGCAGTATTTACCACC | TCCCTTAGCAGAATCCACG |
| LC3A | MAP1LC3A | 135 | AGTTGGTCAAGATCATCCG | CGTCCTCGTCTTTCTCC |
| BECN1 | BECN1 | 131 | GACACGAGTTTCAAGATCCTG | TCAATAAATGGCTCCTCTCCTG |
| ATG5 | ATG5 | 176 | TGGAGTAGGTTTGGCTTTGG | AATAGTATGGTTCTGCTTCCCT |
Abbreviations: ACTB, β-actin; RIPK, receptor-interacting protein kinases; MLKL, mixed lineage kinase domain-like; LC3, microtubule-associated protein light chain 3; BECN1, Beclin 1; ATG5, autophagy-associated protein 5.
3.4. Statistical Analysis
4. Results
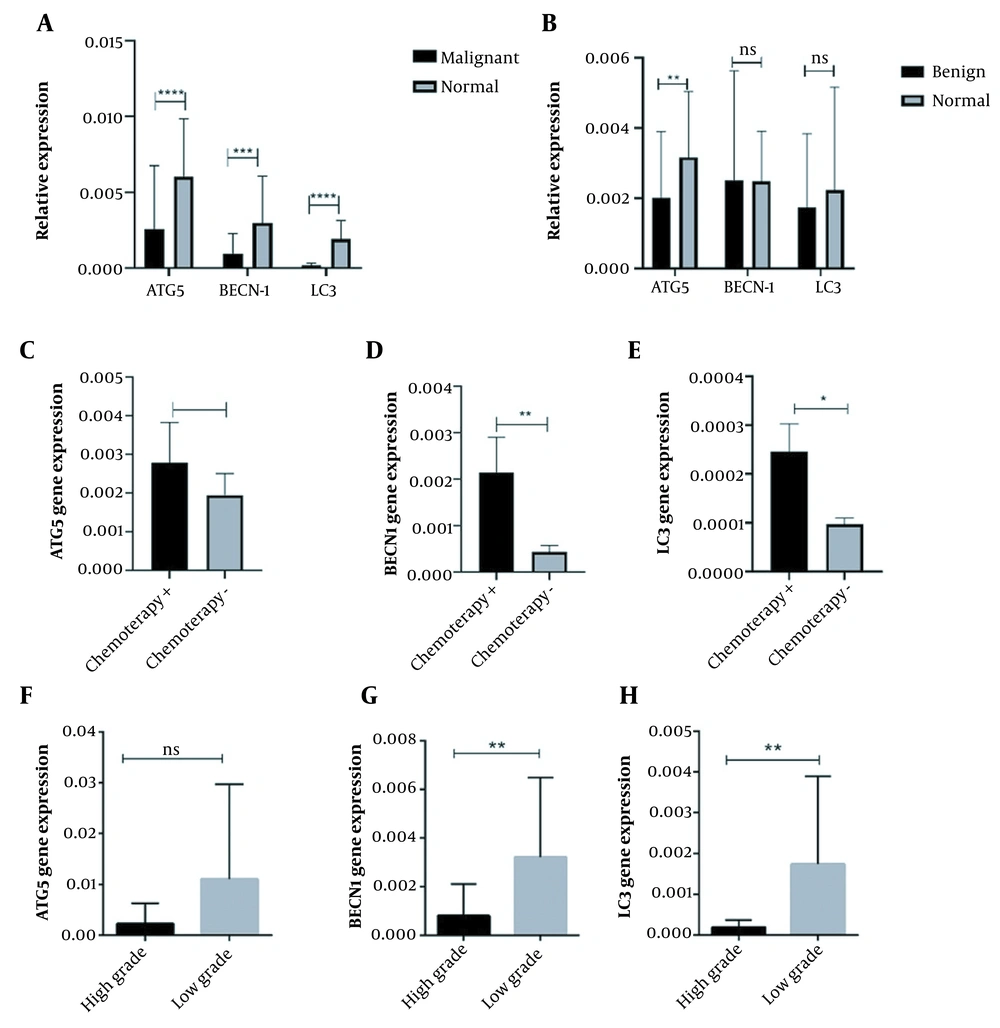
4.1. The Expression Pattern of Autophagy Main Mediators in Osteosarcoma Bone Tumors
The expression pattern of autophagy mediators in osteosarcoma tumor tissues. The autophagy-associated protein 5 (ATG5), Beclin 1 (BECN1), and microtubule-associated protein light chain 3 (LC3) expression levels were evaluated in osteosarcoma bone tumor tissues, and the decreased level of ATG5, BECN1, and LC3 expression was observed in A, malignant tumors versus normal bone tissues also B, benign bone tumors versus tumor margins except for BECN1. The C, ATG5; D, BECN1; and E, LC3 expressions were increased in chemotherapy-received tumors compared to tumors without a history of chemotherapy. The expression level of F, ATG5; G, BECN1; and H, LC3 were reduced in high-grade tumors. The statistical differences between groups are shown as asterisk (* P < 0.05, ** P < 0.01, **** P < 0.0001).
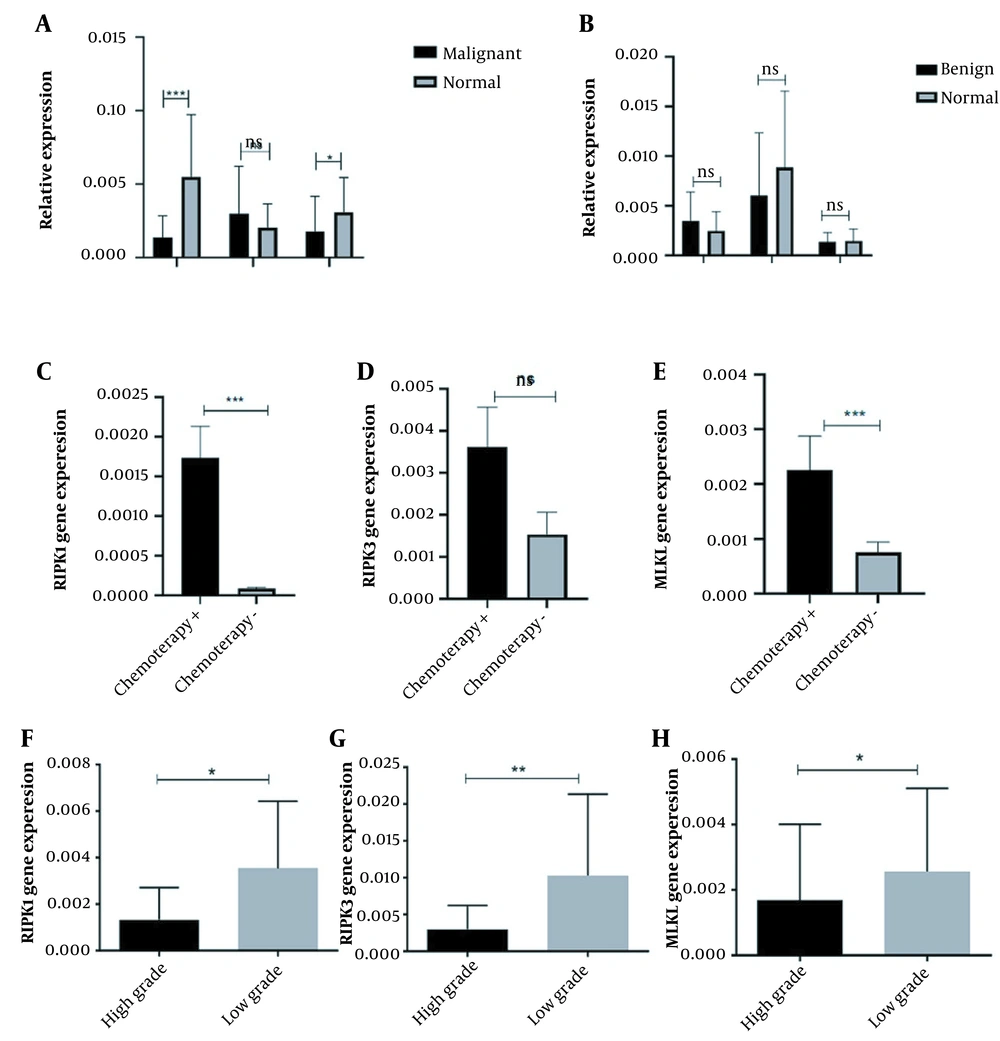
4.2. The Expression Pattern of Necroptosis Main Mediators in Osteosarcoma Bone Tumors
The expression pattern of necroptosis mediators in osteosarcoma tumor tissues. The receptor-interacting protein kinases (RIPK) 1, RIPK3, and mixed lineage kinase domain-like (MLKL) expression levels were evaluated in osteosarcoma bone tumor tissues. A, The expression level of RIPK1 and MLKL was decreased, while RIPK3 showed a non-significant increase in malignant tumors versus normal bone tissues; B, The expression level of RIPK1, RIPK3, and MLKL did not significantly change between benign bone tumors and tumor margins. The expression level of C, RIPK1; D, RIPK3; and E, MLKL was increased in chemotherapy-received tumors compared to tumors without a history of chemotherapy. The expression level of F, RIPK1; G, RIPK3; and H, MLKL were decreased in high-grade tumors. The statistical differences between groups are shown as an asterisk (* P < 0.05, ** P < 0.01, *** P < 0.001).