1. Introduction
The majority of breast malignancies are primary and metastasis to the breast is extremely rare (1). Most breast metastatic diseases originate from the contralateral breast and the extramammary source is rare (2). Many reported metastatic signet-ring cell carcinomas (SRCCs) of the breast are from a gastric origin (3, 4). The breast is an unusual site for metastasis of colonic adenocarcinomas, and moreover, is its metastasis of the rare colonic signet-ring cell carcinoma (SRCC) (5). Histologically, finding tumor cells with signet-ring morphology in the breast makes different diagnoses. Misinterpreting a primary breast carcinoma composed of signet-ring cells with a metastatic SRCC of a colonic primary is possible considering the uncommon colonic metastasis to the breast (6).
2. Case Presentation
A 37-year-old woman underwent a colonoscopy in January 2020 due to a complaint of hematochezia. During the procedure, an ulcerated rectal mass causing partial obstruction was found, located 5 cm from the anal verge. Diagnosing the performed mass biopsy was adenocarcinoma with mucinous features. Radial endoscopic ultrasound evaluation showed the invasion of the tumor into the perirectal tissue staging it as T3. Spiral CT scan imaging studies of the abdomen, pelvis, chest, and mediastinum showed no evidence of metastasis. A conducted upper gastrointestinal endoscopy showed no lesion. She had no known family history of colonic carcinoma. Neoadjuvant chemoradiation therapy was conducted and the patient underwent low anterior resection surgery after 7 months. Histopathologic evaluation of the resected specimen revealed a signet-ring cell carcinoma with small vessel invasion. One regional lymph node involvement and two tumor deposits in the perirectal adipose tissue were identified. Immunophenotyping markers showed no loss of mismatched repair proteins and hence a low probability of MSI-H.
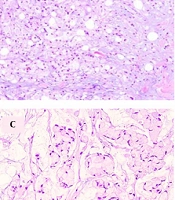
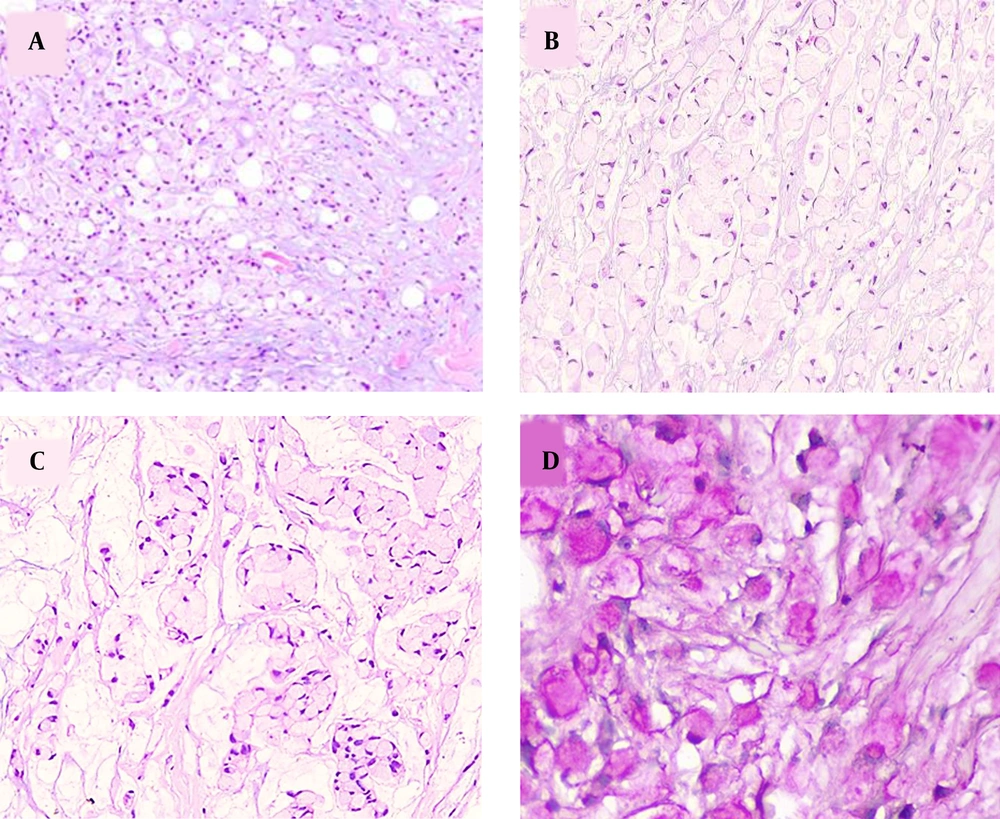
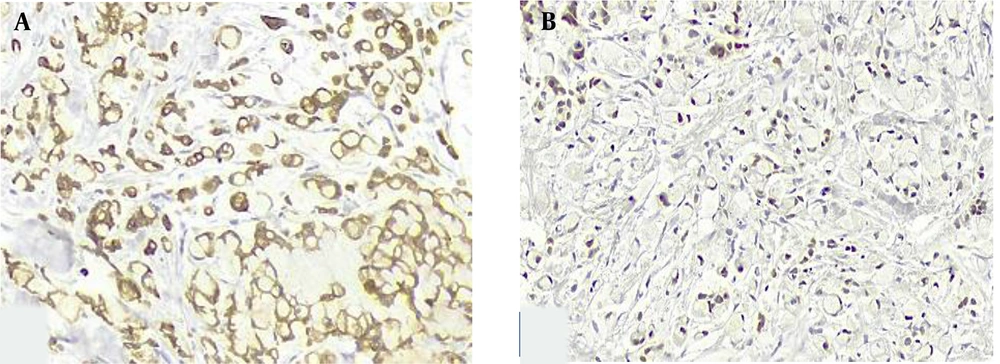
Three months after surgery, the patient came to our center with a palpable left breast mass. Diagnostic mammography showed a dense oval mass with lobulated margins, measuring 21 × 17 mm in diameter, and located at the 2 O’clock mid-zone position. Histopathologic examination of the obtained core needle biopsy specimen showed tumor cells with signet-ring cell cytomorphology among mucinous material, infiltrating strands of fibroconnective, and adipose tissue (Figure 1A). The signet-ring cells formed Indian file arrangements at some foci and clusters of tumor cells floating in extracellular mucin pools at other areas (Figure 1B and C). The tumor cell cytoplasm stained purple with mucicarmine staining, confirming the mucin nature of the intracytoplasmic content (Figure 1D). Taking into consideration the clinical history of rectal signet-ring cell carcinoma, a directed panel of IHC markers was applied. Tumor cells showed homogeneous positive staining for CKAE1/AE3, CK20, and CDX2 (Figure 2A and B). They were negative for GATA3, CK7, ER, and PR markers. These results confirmed a metastatic disease from the colonic primary. The patient underwent a partial mastectomy and axillary dissection. Axillary lymph nodes were free from tumoral involvement. Three months after surgery, the patient developed a second metastasis at the fourth lumbar vertebrae location.
A, discohesive tumor cells in a myxoid stroma infiltrating fibroconnective and adipose tissue (H&E ×100); B, signet-ring cells arranged in an Indian file like pattern (H&E x200); C, signet-ring cells forming poorly cohesive clusters suspended in mucin lakes (H&E x200); D, intracytoplasmic mucin is stained pinkish-purple with mucicarmine stain (H&E ×400).
3. Discussion
Primary signet-ring cell carcinoma of the colorectum is a rare tumor categorized by the World Health Organization (WHO) classification as a distinct entity of adenocarcinoma (7). Histologically, similar to other colonic adenocarcinomas, SRCC can comprise pools of extracellular mucin but is only diagnosed when more than 50% of the tumor is composed of signet-ring cells with prominent intracytoplasmic mucin.
Some studies have shown a higher prevalence of colonic SRCC in females while others have found no sex difference (8). They are strongly associated with Lynch syndrome and have a high rate of microsatellite instability (7). Consistently, these tumors are usually located at the right colon, but can also be found in the left side and rectum (8, 9). Compared to the usual colorectal adenocarcinoma, SRCC presents at a younger age, and has a higher stage at initial diagnosis (8, 9). This aggressive tumor has a high rate of lymph vascular invasion and a tendency for rapid metastasis at multiple foci (8, 10). Liang et al. showed the peritoneum as the most frequent site of colonic SRCC metastasis at presentation and recurrence, with a lower percentage metastasizing to the liver and a very rare incidence of breast metastasis (10).
Although a breast mass can be the first manifestation of a colonic metastatic disease with no known history, they are commonly associated with advanced stages of the primary tumor and as a presentation of its disseminated spread (1). Different primary breast carcinoma subtypes are known to display signet-ring cell differentiation. Pure forms of primary breast signet-ring cell carcinoma have seldom been reported yet signet-ring cells without extracellular mucin can be seen in a variety of breast tumors, among which invasive lobular carcinoma is the most common (11, 12). The latest WHO classification does not classify pure forms of SRCC with no extracellular mucin as a distinct entity of breast tumors (13). Therefore, signet-ring cells are likely a cytomorphologic feature of other breast tumor subtypes. Although metastatic breast tumors from an extramammary site comprise a small percentage of breast malignancies (14), the possibility of metastatic signet-ring cell carcinomas should always be considered when encountering signet-ring cells in a breast tumor. Among solid tumors metastasizing to the breast, gastrointestinal tract SRCC, especially of gastric origin, are a known source (3, 4). Among uncommon reported cases of metastatic colorectal adenocarcinoma to the breast, the rare SRCC subtype has seldom been described (2).
Differentiating primary from metastatic SRCC and assigning the source of its origin has critical value since it can lead to different treatment strategies. In addition to histopathologic features and IHC markers used for such discrimination, considering clinical and imaging findings, and obtaining a history of a prior carcinoma are useful. The previous history of colonic or gastric tumors can guide toward a metastatic disease. If there is no history of a known tumor, a concise imaging or endoscopic evaluation should be conducted and their findings sought.
Among imaging studies, mammographic evaluation can be of value in differentiating primary from metastatic breast malignancies. The latter usually displays as a well-circumscribed mass with no evidence of microcalcification, spiculation, or skin thickening on mammographic images (15, 16).
Histologically, signet-ring cells are recognized by abundant intracytoplasmic mucin which pushes the nucleus to the periphery in a rim-like manner, giving it the popular signet-ring appearance. Histiocytes and melanoma tumor cells can be mistaken with signet-ring cells. Periodic acid-Schiff and mucicarmine, stain the intracytoplasmic mucin content of the signet-ring cells and help in their distinction from such mimickers. The presence of accompanying extracellular mucin with signet-ring cells can be seen in invasive lobular carcinoma, invasive breast carcinoma of no special type, mucinous carcinoma, and rarely in other subtypes of breast tumors. Mucinous carcinoma of the breast, in particular, can show pools of extracellular mucin with floating clusters of tumor cells, in varying shapes and sizes, which are separated by fibrous septa. Yet the presence of intracytoplasmic mucin with signet-ring morphology without extracellular mucin is not a feature of mucinous carcinoma. Therefore, if a tumor consists of only signet-ring cells without evidence of extracellular mucin, it is not classified as mucinous carcinoma.
Based on cytomorphology alone, delineating a metastatic breast SRCC with no clinical history can be problematic due to similar morphologic features of SRCC in different sites (12, 17). Attention to some morphologic features may guide in distinguishing a metastatic tumor. In addition to an extensive lymph vascular invasion, clues to a metastatic tumor are a discernable periductal or perilobular tumor distribution and an unexpected histologic pattern. In contrast, the presence of an in-situ component may help in directing the pathologist towards a primary breast tumor, but caution is required since a distended lymph vascular invasion by a metastatic tumor may simulate an in-situ carcinoma. Endothelial IHC markers highlight the endothelium of the vascular spaces and can be helpful when suspicion exists. Recognition of such diagnostic features might not be possible on examining a small core needle biopsy. As for our case, no recognizable breast tissue was presented in the given sample and only tumor cells were seen splitting some strands of fibroconnective tissue. History of colonic carcinoma, raised our suspicion of the possibility of a metastatic tumor which was further confirmed by the performed IHC panel. When a patient has a history of a known tumor in another site, it is helpful to re-examine the first specimen, if possible, and compare the histopathologic features of both tumors. Similarities in histopathologic characteristics are in favor of metastatic disease.
IHC study has important significance in differentiating primary from secondary breast SRCC and in determining tumor origin since SRCCs of various sources have the same cytomorphologic features. CK7 and CK20 expression patterns are most widely used to distinguish gastrointestinal metastatic sources from breast SRCCs (18). Useful breast-related markers include CK7, GATA3, GCDFP-15, ER, and MUC1. Despite the exclusive expression of ER in breast carcinoma, it can be negative in 20% of breast SRCCs and thus IHC study for ER alone may not differentiate SRCC of the breast from gastric or colon SRCC (12).
Most primary breast carcinomas are CK 7(+)/CK 20(-) compared to metastatic colorectal carcinomas which usually display CK 20(+)/CK 7 (-) and CDX2(+) staining patterns (16). Yet some studies have shown a lack of sensitivity for CK7 and CK20 in this distinction (12). The use of ER and MUC1 as markers of breast SRCC in combination with MUC2 and CDX2 as gastric and colonic SRCC stains can increase the sensitivity and specificity in diagnosing the site of SRCC origin (12).
CK20 and CDX2 cannot reliably distinguish between an upper and lower gastrointestinal source of breast metastatic SRCC, since these markers can be positive in both origins (16). CDX2 nuclear positivity however is heterogeneous and strong in colonic SRCC, unlike gastric SRCC where it is more heterogeneous (12). SATB2 is a sensitive and specific marker for colonic carcinomas and Hep Par 1 expression distinguishes gastric from colonic and breast SRCCs (16).
Colonic SRCC behaves more aggressively than the usual colorectal adenocarcinoma with a low survival rate (19). Its breast metastasis implies a disseminated disease and a more dismal prognosis. Systemic chemotherapy is the main applied therapy. Surgical management of the metastatic breast mass is controversial. Some avoid it due to the risk of skin seeding, tumor recurrence, or short lifespan yet others perform mass excision limited to palliative therapy or simple mastectomy (1, 16, 20).