1. Introduction
Pituitary gland has divided into two anterior and posterior parts with distinct embryonic origins. Pituitary tumors have mainly originated from the anterior part of the pituitary, and pituitary adenomas have been the most common types of these tumors (1). Posterior pituitary cells have contained specialized glial cells which called “pituicytes” (2). Pituicytes such as other glial cells have shown a potential of malignant transformation. These cells could produce glioma tumors like pituitary astrocytoma. Most of the posterior pituitary astrocytoma has reported as low grade tumors with a slow progression (2). In addition to these tumors, several tumors which have located in the sellar and parasellar region, might originate the different sources. Some examples of these tumors include: craniopharyngioma, meningioma, choristomas, dermoid or epidermoid cysts, hamartoma, chordoma, chondrosarcoma, metastasis and germinoma (3, 4). Besides, the location or perspective of the tumors might be erroneously made with adenoma or sellar and parasellar tumors. For example, optic nerve glioma could mimic the view of supra sellar cystic lesions or craniopharyngioma (3, 4).
Diagnosis of sellar and supra sellar masses: it might seem an easy work to diagnose preoperatively these tumors, although the majority of clinical aspects of some lesions wouldbe similar to the pituitary macroadenomas, but some of them, such as glioblastoma have shown more aggressive behavior than adenomas. There is a little report about suprasellarglioma tumors. These tumors had large sizes with areas of cystic degeneration. Approximately, half of these tumors have caused MRI contrast uptake but sometimes a number of them have shown rim enhancement (5). These tumors have been isointense or hypointense to gray matter on T1 weighted images. It might be a focal hemorrhage in imaging findings of pituitary macroadenoma, then although it can help for diagnosis but there is bleeding in many high-grade gliomas. Other imaging techniques such as scientgraphy of somatostatin receptors might be suitable to differentiate adenomas from other pathologies in the sellar and parasellar regions (6).
2. Case Presentation
A 62-year-old male without any history of previous disease, has complained from sudden frontal headache, nausea, vomiting, decreased vision in the left eye, blurred vision and double vision. These signs have gradually deteriorated. We have observed in the cranial nerves examination paralysis of the left abducens nerve (impaired movement in his left eye to the outside), visual impairment in the right eye, and a severe defect in the nasal and temporal field in the left eye. The vision activity of the left eye was 7/10, and then, 10/10 for the right eye. In fundoscopy, the retina has normal but partial atrophy that has observed in optic nerves.
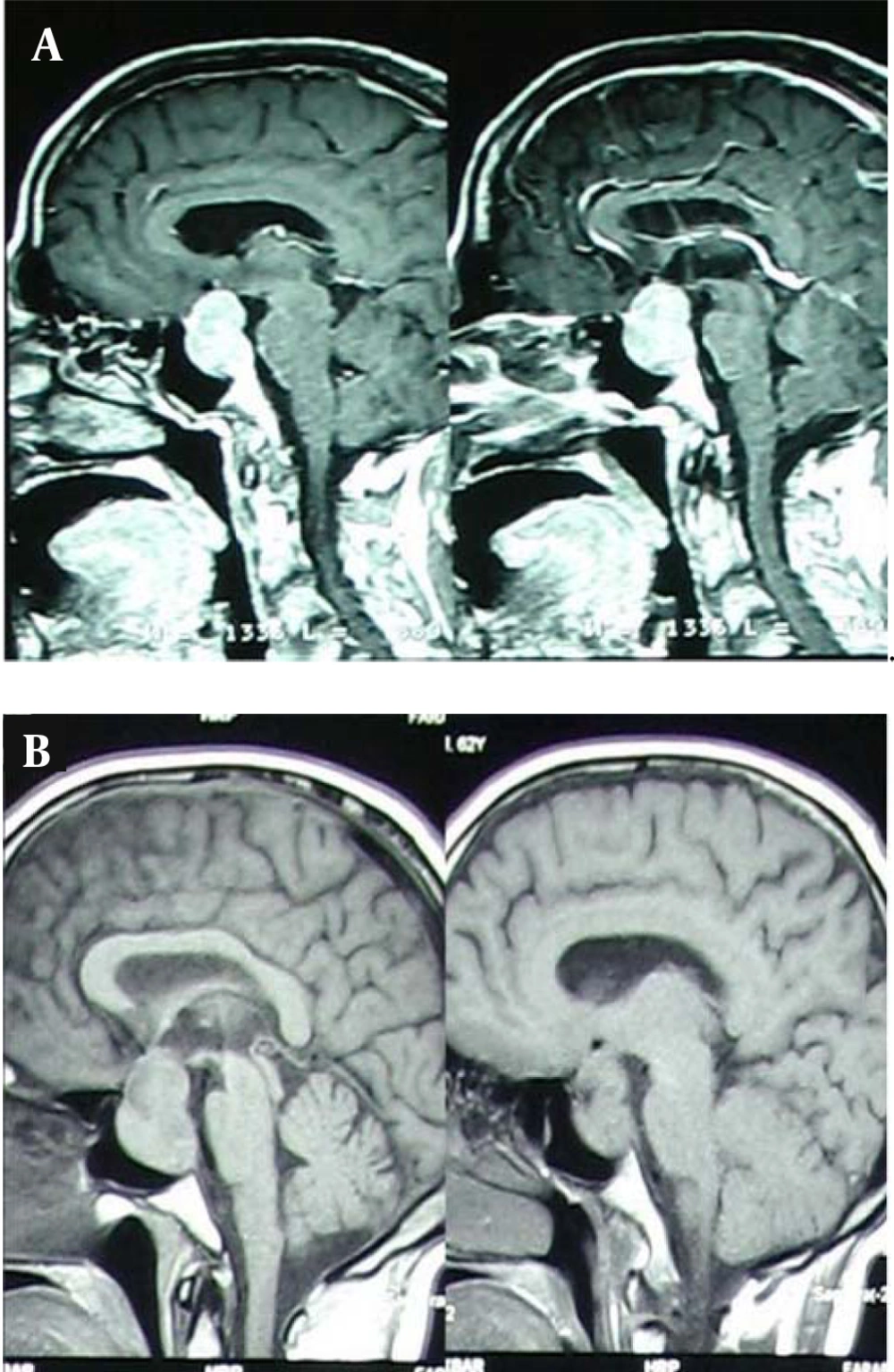
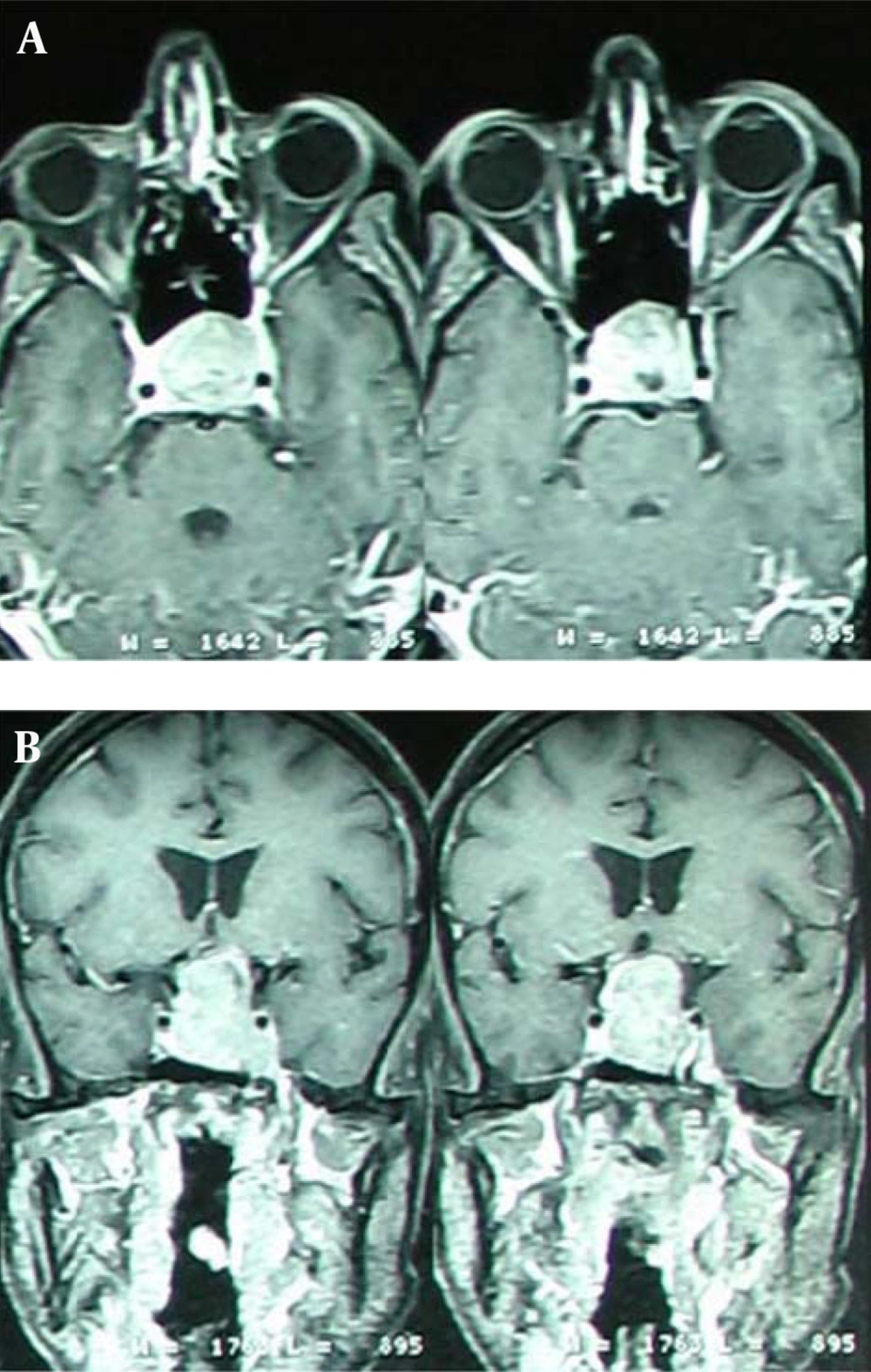
In skull radiographic, there was an expansion of sellaturcica and a lesion with clear border on T1; heterogeneous; iso - to hypotense; size about 3/5 × 2/5 cm with no surrounding edema that it has homogeneously attracted contrast Lesion, that shown a signal hypointensity on T2 with heterogeneous enhancement.
In the coronal sections, the tumor expanded to the suprasellar region and optic chiasmatic that has compressed especially on the left side. Also, it has found an enhancement of the carotid artery without obstructive symptoms and brief extension into the left cavernous sinus (Figures 1 and 2). Clivus and cavernous sinus were intact.
There was not a compression on the forth ventricle or pons. There was also a shift of the middle cerebral artery. The serum levels of GH = 0.1; IGF = 300; prolactin = 7.8; TSH = 0.5 were normal. The serum concentration of ACTH was low (ACTH = 1). Serum Levels for LH and FSH were in normal range, 1.3 and 10 mIU/mL, respectively.
Clinical diagnosis has confirmed the pituitary adenomas. After clinical diagnosis, a partial tumor resection has conducted. The surgery has done by a transsphenoidal approach. We have faced to a creamy soft tissue tumor in sella turcica that was relatively hypervascular with tendency to bleeding. There was no any evidence of bone invasion.
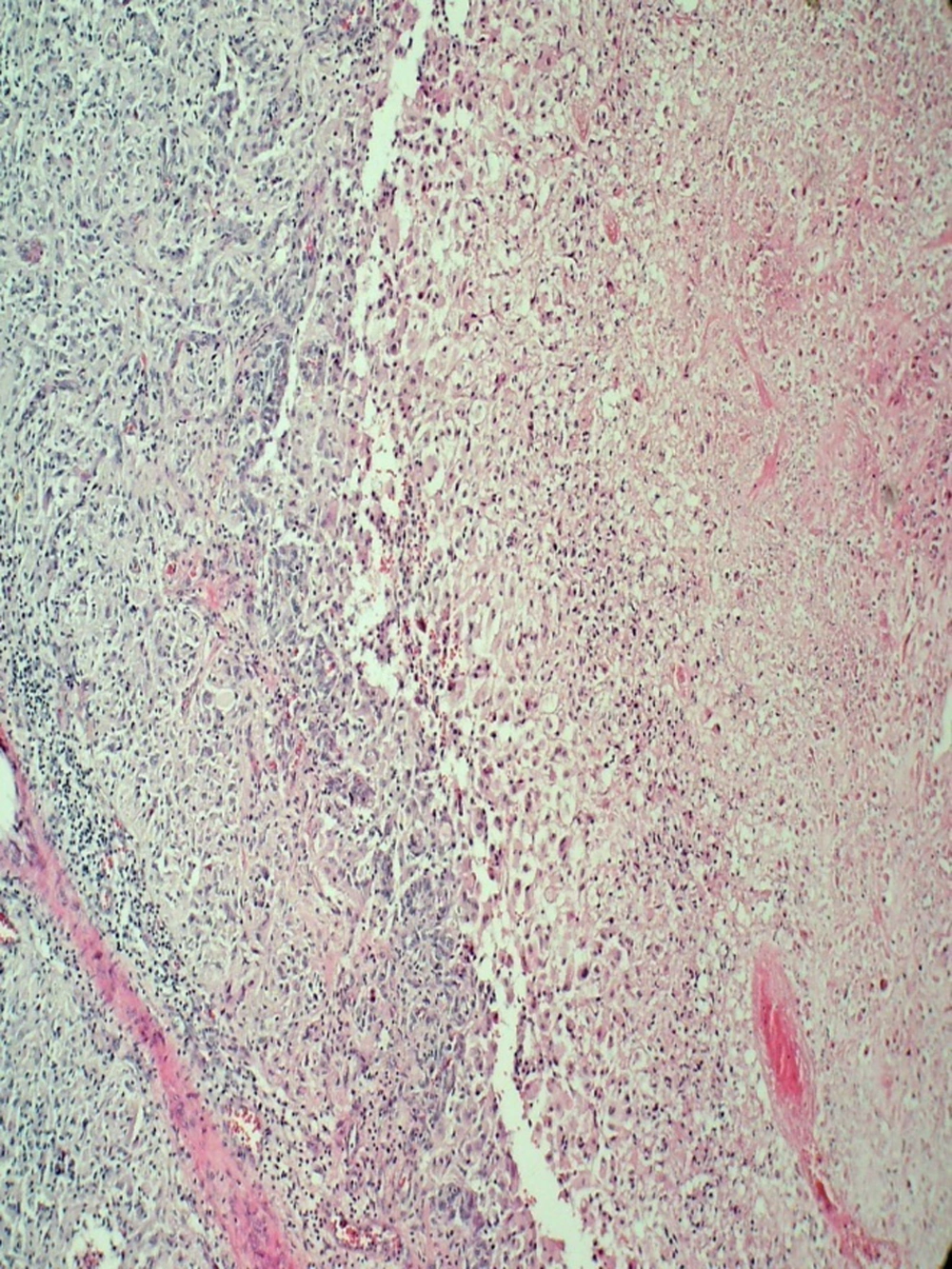
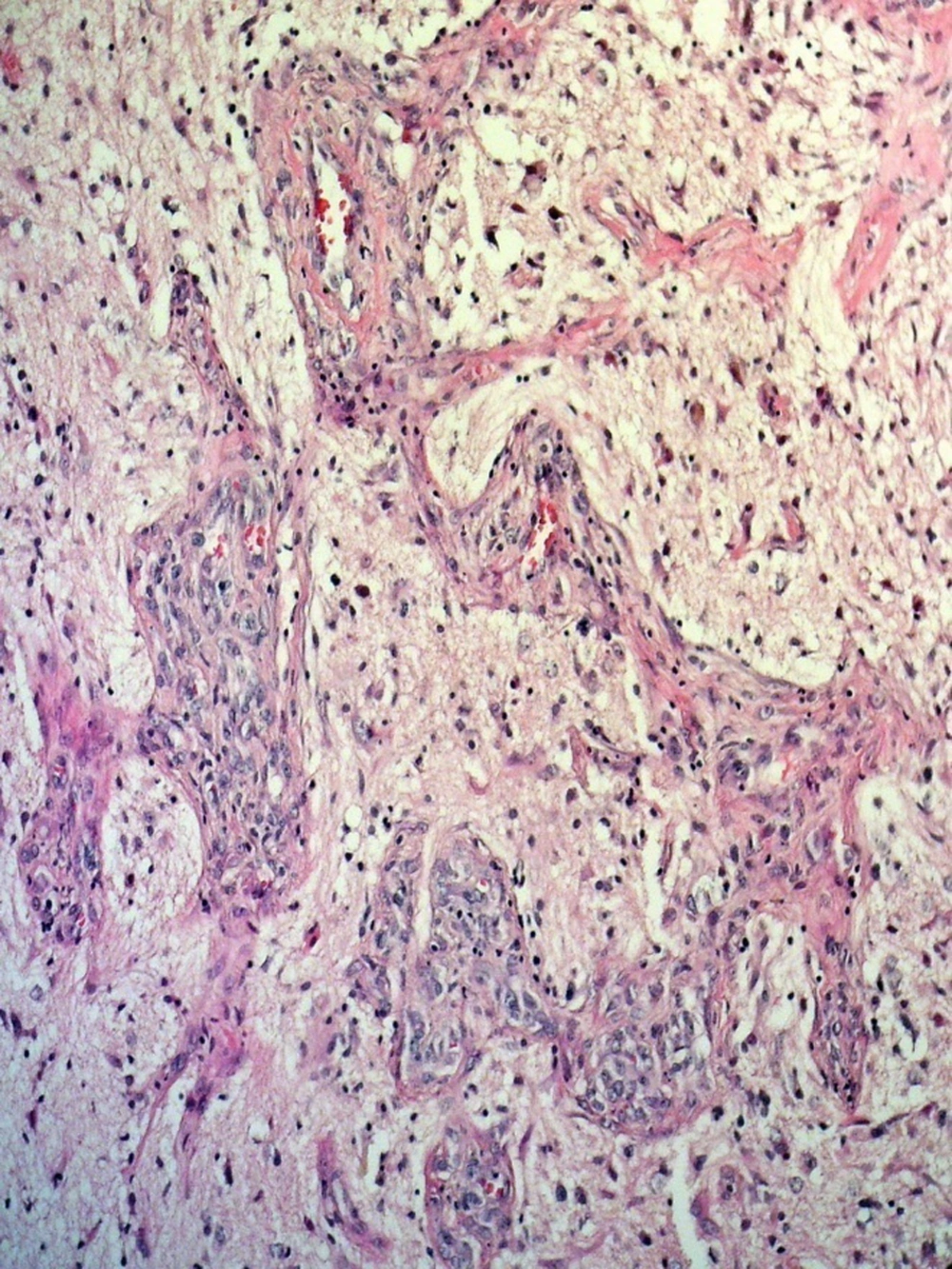
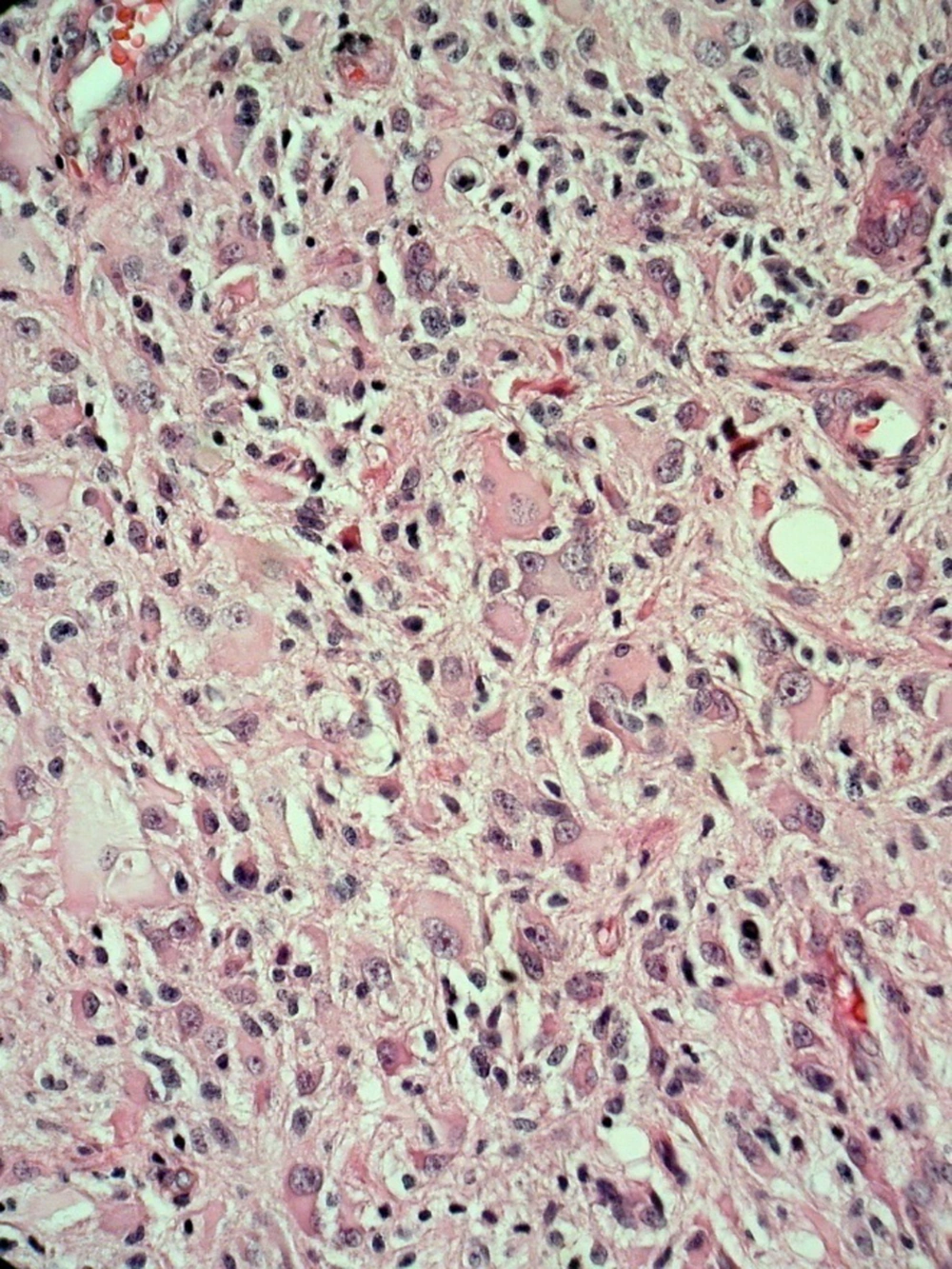
The surgery has done by a transsphenoidal approach. On surgical operation we have found a soft creamy soft tissue tumor in sella turcica without any evidence of bone invasion. Partial resection has done and no complications have found after surgery. The pathologist has diagnosed a glioblastoma multiform due to neoplastic tissue with severe atypia proliferation and extensive necrosis in the microscopic slides. This diagnosis has confirmed using immunohistochemistry technique. Microscopic examination has revealed proliferation of mitotically active highly atypical cells in a fibrillary background with tumoral coagulative necrosis and capillary proliferation, some with glomeruloid feature (Figures 3 - 6). Regarding localization of the tumor, immunohistochemical staining has performed to establish the true nature of the neoplastic cells. These cells have shown strong positive reactivity with GFAP (Glial Fibrillary Acidophilic Protein, Figure 4) and were negative for epithelial markers of cytokeratin (CK) and epithelial membrane antigen (EMA). Synaptophysin was also negative in tumoral cells. So the tumor has diagnosed as glioblastoma multiforme.
3. Discussion
Pituitary gland has divided into two anterior and posterior parts with different embryonic background. Posterior pituitaryhas also named “pituicytes”. Pituicytes and modified glial cells have located in the pituitary stalk and neurohypophysis and originated from empendymal cells. There would be five types of glial cells: major cells, dark cells, oncocyte cells, ependymal cells and granular cells (7, 8).
A review of the literature has revealed only a few case astrocytoma of hypophysis, and there has been scanty discussion of the imaging findings of pituitary astrocytomas. We have found only a handful number of suprasellar malignant gliomas have been published and only one case report of sellar and supracellar GB masquerading as a pituitary macroadenoma (9). Although these tumors were indistinguishable from other pituitary tumors, but pituitary adenomas would be distinctive due to the spindled view of the cells. On the other hand, immunohistochemistry studies have shown presence of GFAP immunoreactivity, neuroendocrine markers and pituitary hormones in these tumors (10). Regarding the pituitary adenomas were the most common types of pituitary tumors, and MRI images for non-adenoma tumors were similar to adenoma tumors; it should consider for a correct diagnosis. In the other words, posterior pituitary tumors should consider by radiologist and clinician before treatment (11, 12). Because of their scarcity, it was hard to ascertain the prevalence of adenoma tumors. Reports have shown that it has happened in the young and middle-aged subjects with signs of headaches, an intracranial focal lesion with a reduction in hypophysis hormones. Visual symptoms have not expected in these tumors, however, in our patient there was a visual symptom because of a compression of the optic chiasm. Our patient had optic atrophy at the presentation that was partial. Weather the behaviors of hypophysis GB would be different from other part of CNS, that mimicked a slow growing features was a question to be proved. Expansion of sellaturcica was another radiologic finding that boosts this hypothesis.
These tumors have shown a signal hypointensity on T1; low to moderate on T2. In our patients, the tumor was hyperintense in T2-weighted images but unlike glioblastoma that has characterized with central necrosis, this tumor has absorbed dye in the central part, and caused to happen an error diagnosis by radiologist. Although it has not been possible to differentiate the anterior or posterior origin of the tumor based on imaging findings, authors recommend a specific attention to the posterior pituitary tumors for diagnosis.
Information about this tumor should consider by the clinicians, radiologists and pathologists in the diagnosis of sellaturcica region for a correct diagnosis and treatment and less likely to recognize the error of tumors of the sellaturcica region.