1. Background
Cancer of cervix is the third most frequent cancer among women worldwide and is still a major concern especially in developing countries (1). Fortunately, due to hygiene measures and available screening methods, the mortality rate of the disease has been reduced, but still current estimates indicate that approximately 528,000 women are diagnosed with cervical cancer every year and about 267,000 die from the disease (1). There are various risk factors involved in development of the disease. Beside different environmental and behavioral factors like smoking, age at first intercourse, sexual behavior and partners, hygiene, and other sexually transmitted infections (2-5), several epidemiological and experimental studies have shown a strong relationship between human papilloma viruses (HPVs) and cervical cancer. In fact, HPVs are known as the major risk factors for developing cervical cancer (6-8). Human papilloma viruses belong to the family Papillomaviridae. These nonenveloped viruses have an icosahedral capsid and a single double-stranded circular DNA genome about 8 kbp in size (9). It seems that two HPV proteins, E6 and E7, are involved in the interfering with the cell cycle, leading to transformation of the host cell by inactivating p53 and retinoblastoma proteins (10, 11). Based on genomic differences, 120 types of HPV have been recognized. Depending on their association with cancerous or non-cancerous lesions, HPVs are categorized into two groups: high and low risk viruses (12). Low risk HPVs (types 6, 11, 42, 43, and 44) are related to benign lesions and high risk HPVs (types 16, 18, 31, 33, 34, 35, 39, 45, 51, 52, 56, 58, 59, 66, 68, and 70) are associated with cervical cancer lesions. HPV-16 and HPV-18 are the most common high risk HPVs reported (6, 13-16). As HPV cannot be propagated in tissue culture, its accurate identification relies on molecular biology techniques like PCR, real-time PCR, and Nucleic acid-hybridization assays (17, 18). One of the mostly used PCR approaches is based on the MY09/MY11 pair primers which targets a 450 base pair (bp) fragment within the HPV L1 ORF (19). As various genotypes of HPV have different risk of tumorigenesis, determination of distributed genotypes in population is very important in the screening and prevention programs against the disease (20). Due to the limited published data on HPV prevalence rates, it is necessary to conduct more studies to know which HPV types need to be targeted by a prophylactic vaccine in each country.
2. Objectives
The present study was conducted to determine HPV genotypes which are circulating in women’s population with cervical cancer and cervical dysplasia/metaplasia lesions using PCR and restriction fragment length polymorphism (RFLP) molecular methods.
3. Materials and Methods
3.1. Preparing of Tissue Samples
The present study was retrospectively done and the samples were collected from pathology department of three different women hospitals in Khuzestan province, Iran (southwest). These pathologically confirmed malignant samples were 72 paraffin fixed cervical biopsies which had been previously archived during 1998 to 2008 from women with cervical cancer and cervical dysplasia. The patients’ ages ranged from 32 to 90 years, with the mean age of 55 years. Ten-micron sections were duplicately prepared from each of the paraffin-embedded blocks and these samples were subjected to DNA extraction.
3.2. Extraction of DNA
First, the paraffin-embedded sections underwent a deparaffinization step using previously described method (21). Briefly, 500 μL of universal buffer (pH 9) was added to each microtube containing two ten-micron sections followed by heating at 120°C for 20 minutes. The samples were then cooled (paraffin forms a solid layer at the top of the liquid phase at this stage) and the liquid phase was harvested.
Extraction of DNA from the liquid samples was performed using phenol-chloroform method. 500 μL of phenol: chloroform: isoamyl alcohol (25:24:1, v/v) solution was added to each sample and the mixture was vortexed and centrifuged at 12000 g for 10 minutes. The supernatant was transferred to a clean microtube and equal volume of chloroform was added to the solution and the mixture was vortexed and centrifuged at 12000 g for 5 minutes. The supernatant was again transferred to a clean microtube and 0.1 volume of 3 M sodium acetate was added to the solution and the mixture was vortexed and one volume of isopropanol was added to the solution and microtubes were stored overnight at -20°C. The microtubes were then centrifuged at 12000 g (4°C) for 10 minutes, the supernatant was discarded, and the pellet was once washed with 70% ethanol. Finally, the microtubes were centrifuged, ethanol was discarded and 50 μL of distilled water was added to the pellet (21).
3.3. Polymerase chain Reaction (PCR)
Analysis of the samples by PCR was done in two steps. First, quality of the extracted DNA samples was investigated using specific primers (CH20 and PC04; TAG Copenhagen Co) for human beta-globin gene. 7.5 μL of extracted DNA was added to 42.5 μL of PCR mix containing 5 U of Taq DNA polymerase, 5 μL of 10X PCR buffer, 1.5 μL of 25 mM MgCl2, 1 μL of dNTPs mix, 0.5 μL (50 pmol) of each of CH20 and PC04 primers and 33.5 of μL distilled water. The reaction was run under the following thermal cycling program: pre-denaturing at 95°C for 2 minutes; denaturing at 95°C for 1 minute, annealing at 55°C for 1 minute, extension at 72°C for 1 minute, (40 repeats); final extension at 72°C for 5 minutes. Finally, PCR products were analyzed by electrophoresis on 1% agarose gel in TAE buffer containing ethidium bromide (0.5 μg/mL). Thereafter, positive samples in this PCR assay were subjected to second PCR assay with specific primers (MY09, MY11; TAG Copenhagen Co) for L1 region of HPV genome (the sequences of the primers are presented in Table 1). Therefore, 10 μL of extracted DNA was added to 40 μL of PCR mix containing 5 U of Taq DNA polymerase, 5 μL of 10X PCR buffer, 3 μL of 25 mM MgCl2, 1 μL of dNTPs mix, 0.5 μL (50 pmol) of each of MY09 and MY11 primers and 29.5 μL of distilled water. The thermal cycling program was the same as the previous PCR. Distilled water was also used as a negative control in each run of both PCR assays.
| Primers | Size, nt | The Sequence | Product Size, bp | Target |
|---|---|---|---|---|
| 20 | 5’-CGTCCMAARGGAWACTGATC-3’ | 450 | HPV L1 region | |
| 20 | 5’-GCMCAGGGWCATAAYAATGG-3’ | 450 | HPV L1 region | |
| 20 | 5’-GAAGAGCCAAGGACAGGTAC-3’ | 268 | Human β-globin | |
| 20 | 5’-CAACTTCATCCACGTTCACC-3’ | 268 | Human β-globin |
The Primers Used in the PCR Assays for the Identification of Human and HPV Genomesa
3.4. Restriction Fragment Length Polymorphism (RFLP) Analysis
PCR products of positive samples in HPV PCR assay were purified using gel extraction kit (QiaGen) according to the manufacturer’s protocol and 10 μL of each of the purified DNA samples were subjected to enzymatic digestion at 37°C for 2 hours using RsaI restriction endonuclease (22). Thus, digestion of the PCR products obtained by primers MY09/11 can lead to the detection of the five most prevalent HPV types 6 (4 fragments of 67, 72, 149, 159 bp; indicator fragments are 149 and 159 bp), 11 (4 fragments of 26, 72, 135, 216 bp; indicator fragment is 216 bp), 16 (3 fragments of 70, 72, 310 bp; indicator fragment is 310 bp), 18 (5 fragments of 38, 85, 72, 125, 135 bp; indicator fragments are 125 and 135 bp), and 33 (4 fragments of 39, 72, 102, 236; indicator fragments are 102 and 236 bp). Afterward, the products of enzymatic digestion were analyzed by electrophoresis on 8% polyacrylamide gel (PAGE) in TAE buffer followed by staining using ethidium bromide solution to determine the number and size of digested fragments (22).
4. Results
Although the collected sample had different statistical characteristics, chi-square test indicated that there was no significant difference between the various groups in all comparisons (P value = 0.05). 45 out of 60 samples (75%) had cervical cancer, whereas, the rest showed cervical dysplasia/metaplasia (25%). These patients were of two groups: those who lived in downtown or rural areas and those who were residents in urban areas. Fifteen patients (25%) with cervical cancer and six patients (10%) with cervical dysplasia were people who lived in downtown or rural areas, while thirty patients of urban people (50%) had cervical cancer and nine patients (15%) had cervical dysplasia. Statistics are given in Table 2.
4.1. PCR Assays
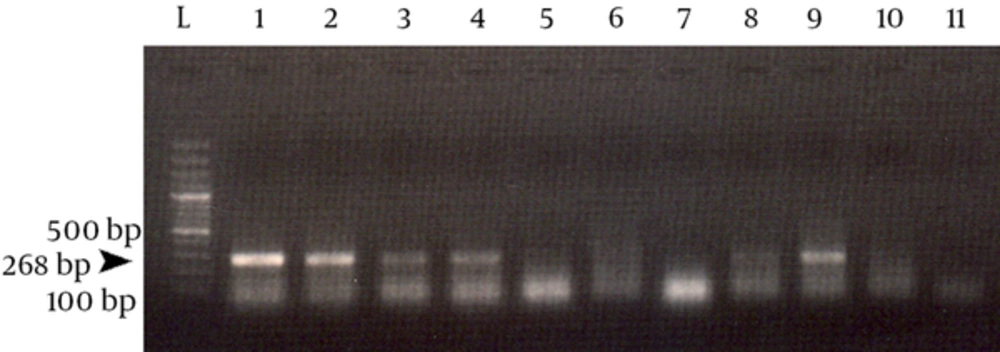
Specific sequence of human β-globin was successfully amplified in the first PCR assay from 60 out of 72 extracted DNA samples. This PCR yielded a 268 bp DNA fragment which confirmed efficient DNA extraction from positive tissue samples. Figure 1 shows an sample result of this PCR.
4.2. HPV PCR
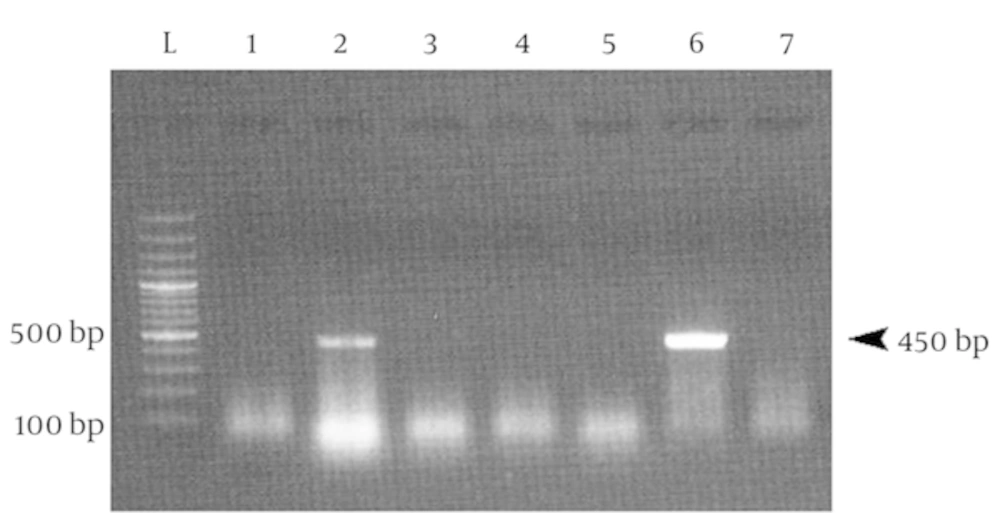
Positive DNA samples in first PCR were then subjected to HPV PCR. This PCR assay targeted a 450 bp DNA fragment in HPV genome. An agarose gel electrophoresis of this PCR assay is shown in the Figure 2.
26 out of 60 samples (43.3%) were HPV positive. 17 cases of these HPV positive samples were from urban people and 9 cases were from rural people (Table 2). Moreover, the samples were classified into seven groups based on women’s age from 32 to 95 years old. Most HPV positive samples belonged to women aged from 48 to 55 and 55 to 63 years. However, 11 out of 45 samples with cervical cancer and 5 out of 15 cervical dysplasia/metaplasia samples belong to 48 - 55 years old group, while 55 - 63 age group only contained 8 samples with cervical cancer. Meanwhile, the samples were categorized into four groups based on women’s first-marriage age from 12 to over 23 years. 13 out of 26 HPV positive samples were from the group with the first-marriage age of 12 - 16 years. Detailed statistics of the tested samples are presented in Tables 3 and 4.
| Age Group | HPV Positive | HPV Negative | Cervical Cancer | Dysplasia/Metaplasia |
|---|---|---|---|---|
| 4 (6.7) | 4 (6.7) | 6 (10) | 2 (3.3) | |
| 3 (5) | 7 (11.7) | 7 (11.7) | 3 (5) | |
| 6 (10) | 10 (16.7) | 11 (18.3) | 5 (8.3) | |
| 6 (10) | 2 (3.3) | 8 (13.3) | 0 (0) | |
| 5 (8.3) | 6 (10) | 10 (16.7) | 1 (1.7) | |
| 1 (1.7) | 5 (8.3) | 2 (3.3) | 4 (6.7) | |
| 1 (1.7) | 0 (0) | 1 (1.7) | 0 (0) | |
| 26 (43.3) | 34 (56.7) | 45 (75) | 15 (25) |
Distribution of the Samples Based on Their Age Groupa
| Marriage Age | HPV Positive | HPV Negative | Cervical Cancer | Dysplasia/Metaplasia |
|---|---|---|---|---|
| 13 (21.7) | 16 (26.7) | 22 (36.7) | 7 (11.7) | |
| 8 (13.3) | 8 (13.3) | 11 (18.3) | 5 (8.3) | |
| 2 (3.3) | 7 (11.7) | 8 (13.3) | 1 (1.7) | |
| 3 (5) | 3 (5) | 4 (6.7) | 2 (3.3) | |
| 26 (43.3) | 34 (56.7) | 45 (75) | 15 (25) |
Distribution of the Samples Based on Their Marriage Age
In addition, 15 and 41 cases of the study population were affected by dysplasia/metaplasia and squamous cell carcinoma, respectively. In addition, 4 cases showed carcinoma in situ and adenocarcinoma. These data are summarized in Table 5. These results showed that percentage of HPV positive samples in SCC samples were higher (48.8%) than dysplasia/metaplasia samples (26.6%).
| Pathologic Lesion | HPV Positive | HPV Negative | Total |
|---|---|---|---|
| 20 | 21 | 41 | |
| 1 | 1 | 2 | |
| 4 | 11 | 15 | |
| 1 | 1 | 2 | |
| 26 | 34 | 60 |
Descriptive Statistics of the HPV Positive and Negative Samples Based on Their Pathologic Lesions
4.3. RFLP
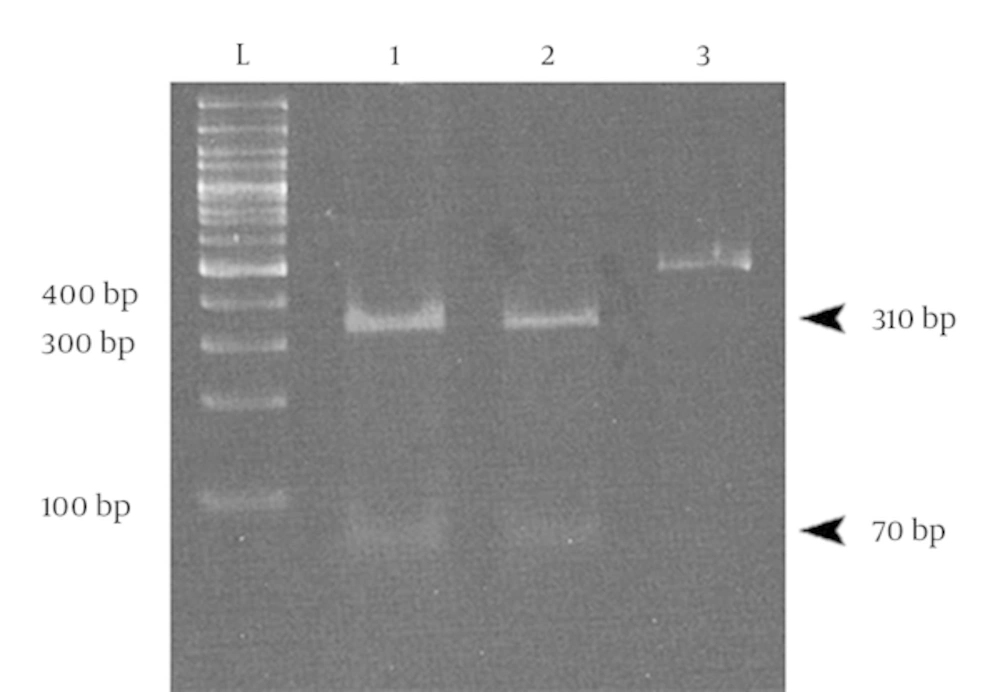
All 26 HPV positive samples were analyzed by digestion with RsaI restriction enzyme. Digestion with this enzyme leads to the identification of five types of the virus including types 6, 11, 16, 18, and 33 (22). Polyacrylamide gel electrophoresis (PAGE) showed that digestion of the products of HPV PCR assay led to the production of two 310 and 70 bp DNA fragments (although there were actually 3 fragments of 70, 72, and 310 bp, the difference between 70 and 72 bp fragments was not recognizable, and thus the electrophoretic pattern consisted of the two DNA fragments). These results indicated that all 26 human papilloma viruses were HPV type 16. A sample result of PAGE is presented in the Figure 3.
5. Discussion
Cancer of cervix is among the most major concerns of women’s health. There are different risk factors involved in development of this lesion and it has been shown that one of the major risk factors is infection with human papillomavirus (6-8). Prevalence of this disease in developing countries is higher than in developed countries probably because there are no organized programs for screening and early diagnosis (23). Few studies have been conducted to determine the epidemiology and effective risk factors on HPV infected cases in the Middle East countries and there are only sporadic available data about this cancer from Iran. Besides, there is no organized screening, vaccination, or eradication program to control cervical cancer. As a result, it is certainly difficult to take proper preventive measures against the disease without organized statistics. Although there are some assays like Pap smear test which are very applicable, but the results could be false negative (24). In addition, knowing the prevalence of HPV infections provides invaluable data towards managing, controlling, and prevention of cervical cancer in women population. Meanwhile, determination of the most prevalent HPV type is very important in preparing effective vaccines against viral infection in different regions and/or countries. Although there are some approved vaccines being used against HPV infections (25-27), type(s) of prevalent papilloma viruses should first be identified to administer the most appropriate vaccine.
Several studies have been conducted to determine prevalence of HPV infections in different groups of patients, the most prevalent types of the virus with their geographic distributions, and risk factors involved in development of cervical cancer. In an extensive epidemiological study, Bosch and colleagues tried to determine whether the association between HPV infection and cervical cancer is consistent worldwide and to investigate geographic variation in the distribution of HPV types. They tested over 1000 histologically confirmed specimens from patients with invasive cervical cancer at 32 hospitals in 22 countries by PCR-based assays, capable of detecting more than 25 different HPV types. Their results showed that 93% of the tumors were infected with HPV DNA with no significant variation in HPV positivity among countries. HPV 16 was present in 50% of the specimens, and were found to be the predominant virus type in all countries (6). Munoz and colleagues performed a study to broaden the knowledge of high risk HPV types. They pooled data from 11 case-control studies from nine countries involving 1918 women with histologically confirmed squamous-cell cervical cancer and 1928 control women. HPV DNA was detected in 1739 of the 1918 patients with cervical cancer (90.7%) and in 259 of the 1928 control women (13.4%). The most common HPV types in patients, in descending order of frequency, were types 16, 18, 45, 31, 33, 52, 58, and 35 (16). In another study performed by Sadeghi et al. in 2008, 52 paraffin-embedded blocks with cervical cancer and 52 paraffin-embedded cervical tissue blocks with normal histology were assayed and the results showed that 30.7% of samples with cervical cancer were infected by HPV, while there was not any positive sample in healthy specimens (28). Maleknejad et al. tried to delineate HPV infections in 64 paraffin-embedded samples from patients with cervical cancer. 59.4% of samples were infected with HPV DNA, 34.4% of which were HPV 16 and the rest (25%) were HPV 18 (29). Han et al. performed a study on young women (< 35 years old) in Korea. The results depicted that 84.5% of samples were infected by HPV 16 and HPV 18, but the most prevalent virus was HPV 16 (30). In a survey, Yang and colleagues investigated correlation of viral factors with cervical cancer in Taiwan. 27 cervical cancer biopsy specimens and 29 normal cervical scrapings were tested and they found that 18 (66.7%) out of 27 cervical biopsies were infected by HPV DNA. Among those, HPV type 16 (9 cases) was the most closely associated with cervical cancer and type 18 ranked second (31). In another research, Schellekens and colleagues tested 74 cervical cancer specimens in Indonesia. HPV DNA of 12 different HPV types was detected in 96% of the specimens. The two most common types were 16 (44%) and 18 (39%), which also indicated high prevalence of HPV 16; however, there are some data which reported HPV 18 as the most common virus type in this region followed by HPV 16 (6, 32).
Considerably, HPV 16 was the most or one of the most prevalent types of the virus in many studies. The results of the present study were also in agreement with the previous findings. In this study, it was shown that 43.33% of patients with cervical cancer or cervical dysplasia were infected by HPV DNA. Unfortunately, as we did not have access to a reference HPV DNA genome, we did not incorporate a positive control. However, it seems that MY09/MY11, which is one of the mostly used pairs of primers, has been proved to be a highly sensitive and specific primer set for detection of HPV DNA (33). Nevertheless, it would be better to use another pair of primers e.g. GP5+/GP6+, to increase sensitivity for detection of HPV DNA in the examined samples (34). Meanwhile, RFLP analysis by RsaI restriction enzyme showed that all (100%) HPVs belonged to one of the most prevalent oncogenic HPVs, HPV type 16. The results of this study confirmed that PCR could be a useful assay to detect HPV viral DNA and subsequent RFLP analysis may help to identify different HPV types circulating in a population. Therefore, it seems that HPV 16 is one of the essential types of the virus which should be included in vaccines against the disease. Meanwhile, although chi-square test did not show any significant difference between various groups of people, there are some points worthwhile to notice. Most of cervical cancer and HPV positive samples belonged to the groups aged from 48 to 71 years, especially 48 - 55 years. In fact, the number of samples was not equal in different sample groups; however, this may also imply on the higher frequency of patients who referred to hospitals in those age groups. It should be considered that previous studies reported that HPV infection is most common in sexually active young women, 18 to 30 years of age and there is also a sharp decrease in prevalence after 30 years of age. However, cervical cancer is more common in women older than 35 years, suggesting infection at a younger age and slow progression toward cancer (15, 35). This difference may be a reflection of social behaviors and traditions, and women’s first sexual intercourse in the region which usually occurs in older ages compared to other regions. Besides, most cases of cervical cancer (36.7%) and HPV positive samples (21.7%) belonged to women who got married before 17. The correlation of marriage age and the incidence of the disease is noticed in some studies (36, 37). However, some researchers did not find any correlation between marriage age and the incidence of the disease (38). These data show that there is a need to perform more studies to increase the knowledge about biology of different HPV types and involved risk factors in the development of the disease. The results of the present study can be guidance for future programs regarding screening and managing of this major disease and also for taking appropriate vaccination strategies against the prevalent types of the virus.