1. Background
Cancer is a major problem of public health in the world and the number of cancer patients increases every year (1). The aim of therapy is recuperation or increasing life expectancy in patients with cancer. Yet in order to destroy cancerous cells, various treatments including chemotherapy, radiotherapy, immunotherapy and surgery have been used. However, a common feature of malignancy is resistance to radiation and chemotherapy which is called multi-resistancy. The resistance of cancer cells to current therapies led to designing new strategies. Viruses are rays of hope for curing a number of cancers (2).
During 90s, oncolytic viruses were used in order to treat solid tumors because they simply spread in tumorous cells and leave the least side effect in normal tissues (3, 4). By direct injection into the tumorous cells, oncolytic viruses make the immune system to stimulate. On the one hand, the failure to maintain antiviral mechanism in tumorous cells makes them sensitive to viruses while protecting normal cells from virus attack. On the other hand, autophagy is an important hypothesis for carcinogenesis and sensitivity of tumor cells to curative viruses. Autophagy is a cellular process for reducing damaged proteins and disabled parts of cells, which helps the survival of cells in high-pressure conditions (pathogen infection, lack of energy) by controlling cell hemostasis. Thus autophagy, in contrast to apoptosis, is part of the cell survival process. Irregularity in autophagy relates with different diseases, such as heart disease, nerve damage and cancer.
Autophagy gets in on the act against viruses. The phenomenon of virus removal by two-layer membrane vesicles is called Xenophagy. The involved genes in autophagy also affect innate and adaptive immunity in response to viral infection (5-8).
Beclin1 is an important, primary molecule for autophagy. This molecule interferes with Ambra1, BIF1, UVRAG proteins for increasing and with Bcl2, and Rubicon for decreasing the autophagy. It also reacts with Vps34 (PI3KIII) for forming the two-layer membrane of autophagosome (9).
In cancerous cells, autophagy is stimulated due to cells’ search for energy sources, suggesting that the control of autophagy path increases the sensitivity of cancer cells to viruses.
The high-risk subgroups of papilloma, a human virus that causes cancer, are related with carcinoma of squamous cells in cervix. When the virus enters the cell, genomic instability caused by protein expression of E6 and E7 leads to transformation of cell. In these cells, the innate and adaptive immunity is reduced. One of the innate immune mechanisms is the path of autophagy. The persistence of human papilloma virus (HPV) infection makes the expression of 3A domain in ATPase family (an anti-autophagy factor) stable and reduces the control of autophagy. There is a negative relationship between Beclin1 and high risk HPV which reveals the fact that in absence of autophagy in hrHPV infection, the cervix squamous cell carcinoma increases (10-12).
2. Objectives
In this study the degree of Beclin1 gene expression in HeLa cell (that has been transformed by E6 and E7 genes) and A549 cell (that is resistant to virotherapy in comparison with many other tumorous cells) was measured. Then the sensitivity of HeLa and A549 cells to the oncolytic VSV was evaluated by MTT assay.
3. Materials and Methods
3.1. HeLa, A549 Cell Culture and Virus Titer Determination Through TCID50
Cells from HeLa (cancer cells from cervical tumor), and A549 (human lung alveolar basal carcinoma epithelial cell line) cell lines were used to study autophagy and Vero cells (African green monkey kidney cells) were applied to propagate VSV virus, New Jersey strain.
The cells were first cultured in DMEM (Dulbeccos Modified Eagles Medium, GIBCO) environment containing 10% fetal bovine serum (GIBCO), and 1% penicillin-streptomycin 100X (GIBCO), in an incubator containing 5% CO2 at temperature of 37°C.
Next, in order to determine the virus titer using TCID50 method, the virus was inoculated on two HeLa and A549 cells in a 96-well culture plate with the logarithmic dilutions of 10-1 to 10-8. One row was left uninoculated as control. After 72 hour the microplate were checked for CPE and the virus titer was calculated using the Karber formula.
3.2. Cell Culture in 6-Well Plate for the Extraction of the Cellular RNA
The HeLa and A549 cells were cultured in 6-well plates. After reaching the proper density (90%), the cells were inoculated with 0.1 and 0.001 multiplicity of infections (MOls) of virus, leaving one row as control. Twelve hours after the incubation in the incubator containing 5% CO2 and the temperature of 37°C, the cells were harvested, and the supernatant was discarded after centrifugation. The resultant sediment was kept in Qiazol solution (Qiazol lysis reagent, Qiagen) for the extraction of cellular RNA according to the manufactor’s protocol. cDNA synthesis was carried out by random hexamer at 42°C for 1 hour (United States, Thermo Scientific Kit).
3.3. Beclin1 Primer Design
The Beclin1 human gene sequence was extracted from NCBI and the primers were designed in the form of exon junction using allele ID software.
The primers forward: 5' → 3' CGTGTCACCATCCAGGAACTC.
Reverse: 5' → 3' GTTTCAATAAATGGCTCCTCTCC amplified a 109 bp fragment.
3.4. Real Time PCR
The relative amount of the expression of Beclin1 gene in two HeLa and A549 cells in comparison with GAPDH internal control was carried out using Corbet device.
3.5. MTT Assay
In this method, the percentage of the vital cells in HeLa and A549 was determined based on the restoration and break down of the yellow crystals of Tetrazolium using the succinate dehydrogenase enzyme and the formation of blue crystals. HeLa and A549 cells were cultured in a 6-well plate and inoculated with 1, 0.1, 0.01 and 0.001 MOls of the virus after reaching proper density. After 24 hours, the MTT solution (Recombinant Life Idea, Tehran) was added and 2 to 3 hours later the supernatant was discarded and DMSO was added. Fifteen minutes later, the optical density was read at a wavelength of 570 nm.
4. Results
4.1. Titration in HeLa and A549 Cells
The TCID50 titer of VSV was calculated for the HeLa and A549 cells, 72 hours after virus inoculation using the Karber formula in which the virus titer was obtained to be 105 and 104.37 TCID50/mL for HeLa and A549, respectively.
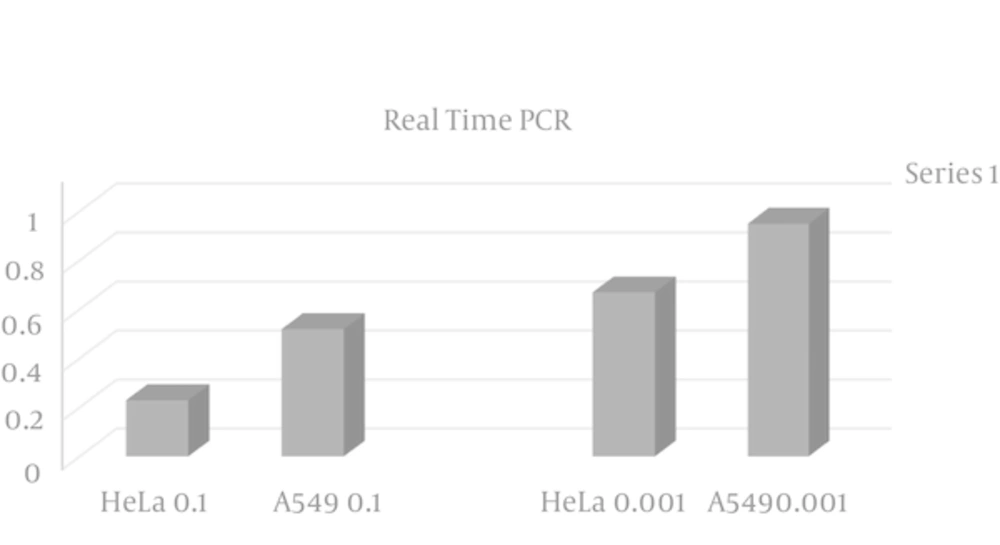
4.2. Real Time PCR
In order to study the quantitative level of gene expression, first the technical accuracy was evaluated using the melting curve. Then the amount of CT was determined using the amplification diagram.
Based on the results of REST program, from the statistical point of view, the level of Beclin1 gene expression in HeLa cell significantly declined (P ≤ 0.05) compared to A549 (Figure 1).
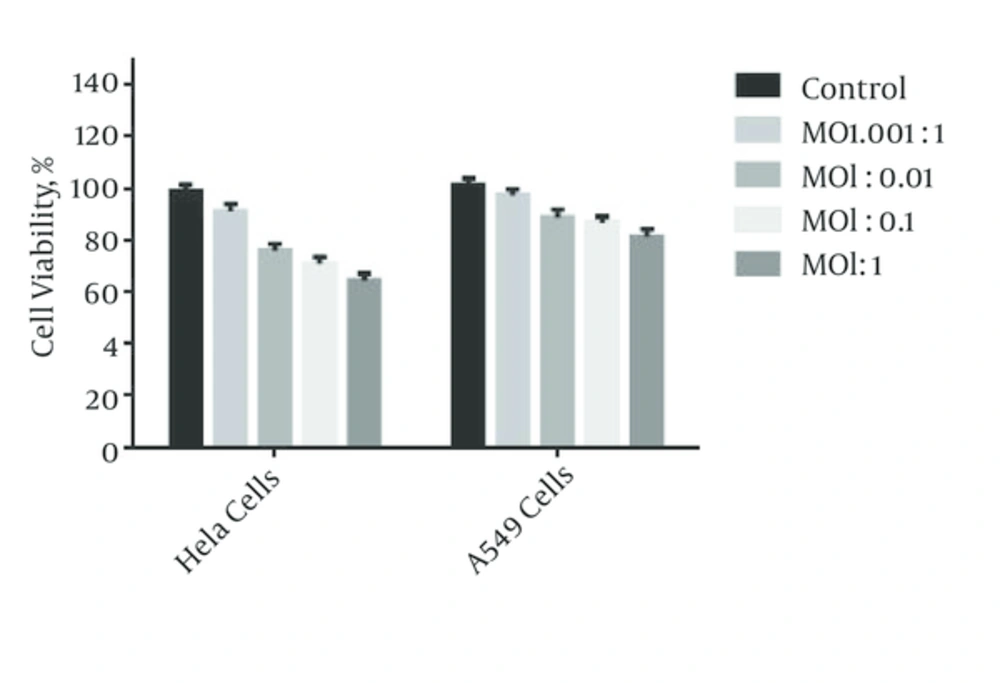
4.3. MTT Asay
The percentage of vital cells 24 hours after virus infection at different MOIs was evaluated. Based on the results of the MTT test for the two HeLa and A549 cells, the percentage of the vital cells in A549 was higher compared to that of HeLa (Figure 2).
5. Discussion
Autophagy plays a complicated role in the production of tumor and response to treatment. In the early stages of tumorgenesis, autophagy can serve as a suppressor and reduction of autophagy has been observed with the transformation of tumor cells (13).
In this case, inducing autophagy seems useful for the prevention of cancer, although at the stage of tumor creation, autophagy contributes to tumor development and cancer cells are reinforced by autophagy to survive under treatment and metabolic stress. On the other hand, autophagy is considered as an innate antivirus mechanism against viral infection.
Data suggest that autophagy is directly activated by VSV infection, most likely via the surface glycoprotein VSV G, and thus activation does not require viral replication. Shelly et al. showed that VSV RNA is not the sensor for autophagy in flies (5).
Malilas et al. in 2013 showed that A549-CUG2 cells, which harbor a novel oncogene mediating stat1 activation causing resistance to infection of vesicular stomatitis virus became susceptible to VSV infection upon treatment with Beclin1 siRNA (14).
Most cancer cells, including HeLa, are sensitive to virus therapy due to defects in the innate immune system (including autophagy) while the activity of the autophagy mechanism can be interpreted as a contributing factor to the resistance or lack of proper response from some cancer cells to virus treatment. In a number of studies on various cancers, inhibition of autophagy by drugs including 3MA (Inhibition PI3KIII), cancer cells have sensitized to a wide range of treatment models, including radiation theraphy, hormonal therapy etc. By inhibiting autophagy, viruses are protected from the xenophagy phenomenon which helps the viruses to destroy cancer cells (5-7, 13, 15-17).
Among the most important evidence of the role of autophagy in tumor suppression is a study conducted on Bcl2 protein and Beclin1. Anti-apoptotic Bcl-2 interacts with the BH3 domain of Beclin1, which results in autophagy reduction and tumor progression. Elimination of Beclin1 is frequently detected in breast, ovarian, prostate cancer in humans and mice (9).
In the present study, a comparison was made between the activity level of Beclin1 gene, as the most important factor for the commencement of the autophagy process, in two HeLa and A549 cells. The reduced expression of Beclin1 was expected for HeLa due to the activity of E6 and E7 genes in the autophagy process. A549 cell is reported to be resistant to virus treatment compared to many other tumor cells. The results showed that autophagy was reduced in HeLa cells compared to A549 and perhaps the heightened sensitivity of HeLa cells to the death due the VSV virus can be attributed to the reduction of autophagy in these cells, although further investigation is needed to fully evaluate this connection (6).
In conclusion, it can be proposed that for the development of successful autophagy strategies against cancer, there is a need for a better understanding of cell mechanisms in connection with the stages of tumor, cell type and genetic factors. In addition, we must be assured about the special autophagy pathways activated or inhibited in various anti-cancer treatments.
It seems possible to increase the sensitivity of tumor cells to virus treatment or other treatments by finding ways to reduce autophagy (7, 13).