1. Background
Squamous cell carcinoma accounts for more than 90% of head and neck cancers (1). Despite many advances have been made in recent decades in therapeutic fields such as surgery, chemotherapy, and radiotherapy, the 5-year survival rate is still lagging behind (2). The advanced lesions of oral squamous cell carcinoma (OSCC) often occur with pain, halitosis, difficulties in speaking, and swallow, whereas the initial lesions do not have any symptoms (3). Although the oral cavity can be directly examined, most patients are detected with weak prognosis in the advanced stages of the disease (4). It seems that delay in diagnosis is the primary reason for high mortality rates associated with this cancer (5). Therefore, an early diagnosis would lead to better treatment results and can increase the five-year survival rate up to about 80% (6).
The lack of early warning signs represents the importance of finding biomarkers with high sensitivity and specificity for early detection and screening of high-risk individuals (2). Matrix metalloproteinases (MMPs) are a family of extracellular zinc-dependent endopeptidases capable of selectively degrading various components of the extracellular matrix (ECM) and non-matrix proteins. Most of the members of this family share a catalytic domain with a conserved zinc binding site followed by a C-terminal hemopexin-like domain involved in the recognition of specific substrates. MMP-2 (gelatinase A) and MMP-9 (gelatinase B), which harbor an additional collagen-binding domain inserted in the catalytic domain that binds collagens and fibronectin. MMPs are upregulated in various types of human cancer and are associated with tumor progression including invasion, angiogenesis and metastasis, and poor prognosis. MMP-2 and MMP-9 are the most studied MMPs in cancer since their overexpression in tumor cells is linked to metastasis and advanced tumor stages. Recent investigations have shown that these 2 gelatinases play important roles in migration, invasion, and angiogenesis of tumor cells by releasing ECM-associated growth factors, modulating the activities of cytokines and chemokines, and inducing critical intracellular signalling cascades through interaction of their hemopexin (PEX) domain with cell surface receptors (7).
2. Objectives
To date, there is no study on the evaluation of serum and salivary levels of MMP-2 and MMP-9 in patients with OSCC. In this study, the protein levels of MMP-2 and MMP-9 in the saliva and serum of patients with head and neck SCC (HNSCC) were compared with those of the healthy subjects in a case-control study and their relationships with the stage and grade of SCC were determined. In addition, the salivary and serum levels of these proteins were evaluated within each group to determine whether there is a direct correlation between their serum and salivary levels.
3. Methods
In this pilot case-control study, 20 patients with head and neck squamous cell carcinoma presented to the Omid and Qaem hospitals in Mashhad, Iran from October 2014 to June 2015, were selected using purposive non-probability sampling technique. HNSCC of the case group was confirmed by histopathology and they had not received any treatment before the sampling. The control group was included 20 normal volunteers who had no history of malignancy or immunodeficiency.
Patients with immune deficiency diseases, other malignancies, renal or liver diseases were not included in the study. Additional exclusion criteria were the presence of any kind of oral malignancy in the control group during the experiment and also the patient’s dissatisfaction.
Demographic data such as age and gender; malignancy-related features including the site of involvement, symptoms and TNM (T: primary tumor size; N: regional lymph nodes involvement; M: metastasis) as well as the history of alcohol and smoking consumption were recorded in special forms based on the medical records and interviews with the patients.
3.1. Saliva Sampling
Saliva sampling was performed according to the standard technique. At 9 to 11 A.M., participants were asked not to eat, drink, smoke or use any hygienic device during at least an hour before sample collection. To prevent the salvia proteins from degradation, the samples were stored at -80°C until being analyzed (2). The saliva samples were centrifuged at 4500 g for 20 minutes to separate the cells and debris.
3.2. Serum Sampling
Blood samples were drawn before treatment. Serum was separated within an hour after blood collection; the venous blood samples were kept at room temperature for 30 minutes to be clotted, and then were centrifuged at 2000 rpm for 15 minutes. Afterwards, sera were collected and kept at -80°C until being analyzed.
3.3. Analysis of Serum and Salivary MMP-2 and 9 Levels by Sandwich ELISA Technique
MMP-2 and MMP-9 protein concentrations in all samples were measured by Human MMP-9 Quantikine ELISA Kit (R&D Systems #DMP900, USA) and Human MMP-2 Quantikine ELISA kit (R&D Systems #DMP2F0, USA) according to their manufacture’s instructions.
The data were presented in nanograms per mL. In this experiment, serum and salivary levels of both active type (82 kD), as well as the pre-enzyme (92 Dk) of MMP-9 protein was measured in a 3.5- hour phase assay; and MMP-2 level was quantified in a 4.5- hour ELISA solid phase assay. Briefly, the microtiter plates were coated with either anti-MMP-2 or anti-MMP-9 polyclonal antibodies, and the standard solutions and samples were transferred into the wells. After washing the unbound materials, the enzyme-linked polyclonal antibody against either MMP-2 or MMP-9 antibodies was added. This was followed by another washing step and the addition of the substrate solution. The developed color was fixed and the light absorbance at 450 nm was measured by a micro-plate reader.
3.4. Ethical Considerations
The study protocol was approved by the Ethics Committee of Mashhad University of Medical Sciences, Iran. The methodology was described to all patients and the informed consent was obtained from the study participants.
3.5. Statistical Analysis
In order to data collection, data were analyzed by SPSS (version 16) software. The descriptive analysis was done by using tables and charts, while the explanatory analysis was conducted using the 2 appropriate mean comparison test (based on the normality or non-normality of the data, the t-test or Man Whitney test was used, respectively) and the Spearman correlation test. The significance level was set at ≤ 0.05 for all tests.
The assessment of the relationship between the markers and metastases, grade, location, and age was done with the independent t-test, while the relationship of the markers with the stage of the disease and correlation between the serum and salivary levels of each protein was assessed with the Pearson test.
4. Results
A total of 20 HNSCCs were identified with 9 male and 11 female patients, with the average age of 60.95 ± 15.32 years. The control group was comprised of 20 normal volunteers including 5 women and 15 men, with the mean age of 53.30 ± 4.48 years.
The tumor site was intra-oral in 11 participants (tongue, Alveolar ridge, palate) and extra-oral cavity in 9 patients (larynx, hypopharynx, and nasopharynx). Stages and grades of the tumors are shown in Table 1. Eleven patients had metastases to lymph nodes and 9 patients had no metastasis.
| I | II | III | IV | |
|---|---|---|---|---|
| Grade | 6 | 14 | - | - |
| Stage | 2 | 6 | 5 | 7 |
Number of Patients According to Grade and Stage of Tumor
Statistically, a significant difference was not observed in the salivary levels of MMP-2 in the control (2.21 ± 0.31 ng/mL) and case (2.17 ± 0.62 ng/mL) groups (P = 0.764). Although the serum levels of MMP-2 in the case group (12.48 ± 2.19 ng/mL) was higher than that in the control group (11.69 ± 2.39), but this difference was not statistically significant (P = 0.283) (Table 2).
| Variables | Study Groups, ng/mL | P Value | |
|---|---|---|---|
| Case | Control | ||
| MMP-2 saliva | 2.17 ± 0.62 | 2.21 ± 0.31 | 0.764 |
| MMP-2 serum | 12.48 ± 2.19 | 11.69 ± 2.39 | 0.283 |
| MMP-9 saliva | 49.27 ± 44.50 | 44.68 ± 40.95 | 0.736 |
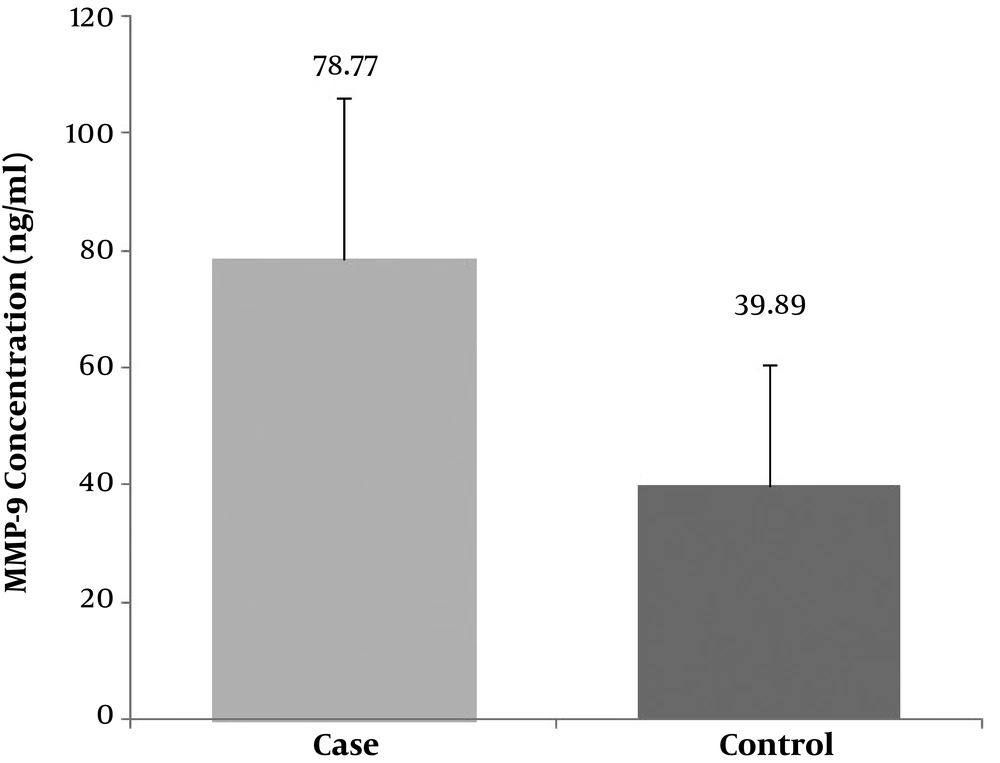
| MMP-9 serum | 78.77 ± 27.51 | 39.89 ± 20.54 | < 0.001 |
Levels of MMP-2 and 9 in the Serum and Saliva of the Case and Control Groupa
The salivary levels of MMP-9 in the case group (49.27 ± 44.5 ng/mL) were higher than the control group (44.68 ± 40.95 ng/mL), but this difference was also not statistically significant (P = 0.736) (Table 2). In contrast, the serum levels of MMP-9 in the case group (78.77 ± 27.51 ng/mL) was substantially higher than that of in the control group (39.89 ± 20.54 ng/mL) with a statistically significant difference (P < 0.001) (Figure 1).
Pearson test in the case group revealed a significant direct relationship between serum and salivary levels of MMP-2, and also between serum and salivary levels of MMP-9 [(r = 0.452, P = 0.046) and (r = 0.558, P = 0.011), respectively]; while no significant relationship was observed in the control group [(r = -0.115, P = 0.628) and (r = 0.422, P = 0.064), respectively] (Table 3).
| Groups/Variables | Type of Sample | Double t-test | Pearson Correlation Coefficient | P Value | |
|---|---|---|---|---|---|
| Saliva | Serum | ||||
| Case | |||||
| MMP-2 | 2.17 ± 0.62 | 12.48 ± 2.19 | > 0.001 | 0.452 | 0.046 |
| MMP-9 | 49.27 ± 44.50 | 78.77 ± 27.51 | 0.002 | 0.558 | 0.011 |
| Control | |||||
| MMP-2 | 2.21 ± 0.31 | 11.69 ± 2.39 | > 0.001 | -0.115 | 0.628 |
| MMP-9 | 44.68 ± 40.95 | 39.89 ± 20.54 | 0.572 | 0.422 | 0.064 |
Comparison of Levels of MMP-2, and 9 in the Serum and Saliva of the Case and Control Group
Moreover, the tumor site was subdivided into intraoral or extraoral cavity to evaluate the relationship between variables and tumor site. The results of T independent test demonstrated that serum and salivary levels of MMP-2 or 9 did not have a significant relation with tumor site and metastasis.
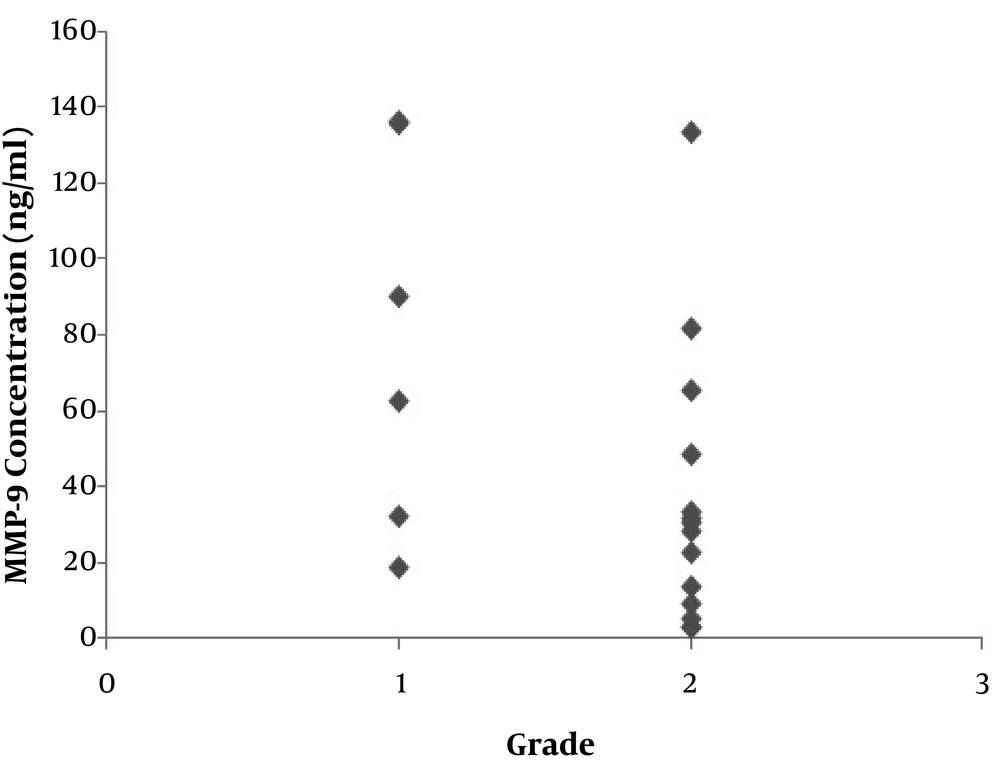
Regarding the histopathologic grade of tumors, t-test revealed that there was no significant relation between serum levels of MMP-2 or 9 as well as salivary levels of MMP-2 with the grade of the disease; whereas the relation between salivary levels of MMP-9 and the tumor grade was statistically significant (P = 0.045) (Figure 2).
In addition, the results of ANOVA and Spearman tests demonstrated that there was no significant relationship between serum and salivary levels of MMP-2 or 9 with tumor stage.
As presented in Tables 2 and 4, the serum levels of MMP-9 showed no correlation with clinical parameters such as the tumor location, clinical stage, histopathological grade, and lymph node metastasis. Interestingly, the salivary levels of MMP-9 were significantly associated with the tumor histopathological grade (Table 4). No correlation was found between salivary or serum levels of MMP-2 and any of the clinical parameters.
| Tumor Site | Grade | Stage | Metastasis | ||
|---|---|---|---|---|---|
| P Value | Correlation Coefficient | ||||
| Salivary MMP-2 | 0.717 | 0.873 | 0.217 | 0.289 | 0.377 |
| Serum MMP-2 | 0.735 | 0.539 | 0.265 | 0.262 | 0.249 |
| Salivary MMP-9 | 0.677 | 0.045 | 0.414 | -0.193 | 0.874 |
| Serum MMP-9 | 0.372 | 0.250 | 0.759 | -0.073 | 0.977 |
Serum and Salivary Levels of MMP-2 and 9 in SCC Patients in Relation to the Clinicopathological Features of Tumor
5. Discussion
Recent achievements in the field of biological research have provided a better understanding of the molecular processes involved in the pathogenesis and progression of HNSCC and led to the identification of a large number of biomarkers. However, further studies are needed in order to make the clinical application of these biomarkers possible.
MMP-2 and 9 are proteases which play a role in removing the extracellular matrix through collagen IV decomposition and are thus involved in the invasion of tumors and metastasis.
MMP-9 plays a role in inflammation, wound healing, tissue remodeling, movement of matrix-bonded growth factors, and cytokine’s processing (8). MMP-2 (gelatinase A) is expressed at high levels during the growth. This enzyme increases at the sites of tissue damage, inflammation and in the stromal cells of the invading edge of the metastatic tumors.
High expression levels of MMP-9 and MMP-2 was shown in different cancers including HNSCC (9-11). Most of these studies have focused on tissue samples and showed an increased expression of these proteins in tumoral tissues. While, most published research in the field of cancer biomarkers have focused on blood components such as serum, plasma, and urine. Saliva as a source of biomarkers has captured less attention (12). In addition, advantages of the salivary sample can be noted due to the ease of collection, no need for special equipment and a trained person for sampling, and non-invasive and simplicity nature of the sample collection. Thus the establishment of the salivary diagnostic methods in these patients is highly valued (5).
According to the results of the present study, the mean serum levels of MMP-9 in patients with HNSCC were considerably higher than normal subjects. Further, serum and salivary levels of either MMP2 or 9 in the case group were significantly correlated; but this correlation was not observed in the control group.
In the study conducted by Shpitzer et al. (13) MMP-2 and MMP-9 proteins were measured in salivary samples of OSCC patients in which the tumor location was in the lateral border of the tongue. The salivary concentration of MMP-2 and MMP-9 in the case group were higher than the control group. However, in the present study, although the salivary levels of MMP-9 were higher in case subjects, the difference was not significant. In another study in which the MMP-9 concentration was measured in the salivary samples of patients with tongue SCC, its concentration was considerably higher in the patients in comparison with the control subjects (14).
Regarding the serum levels of these proteins in studies of Wang et al. (15), Cheng et al. (16), and Ranuncolo et al. (17), the MMP-9 concentration was reported to be significantly higher in patients with HNSCC compared to the control group. These results were similar to the results of the present study in which serum concentration was significantly lower in healthy subjects than SCC patients.
In the other studies (18-22), the MMP-9 protein was represented as a better marker compared with MMP-2 for the evaluation of metastasis and malignant changes and for assessing the clinical features, the prognosis predicting factor, and the accurate grading of tumors. Moreover, in Patel et al. (23, 24) and Xu et al.’s (25) studies, the incidence of MMP-2 was associated with lymph node metastases. The disagreement between these studies might be the result of the variety in tumor locations, the number of samples, population differences, and the diversity in sensitivity of the applied techniques.
As it has been observed in several studies, the serum levels of MMP-9 in patients with HNSCC are significantly higher than that of healthy and treated patients, but this difference was not observed for MMP-2 levels, which is consistent with the results of the present study. Therefore, although there are limited studies in this area, it seems that MMP-2 is not a reliable marker for diagnosis and prognosis of HNSCC.
Due to the limitations of the present study such as small sample size, high diversity in tumors locations, and the lack of age/sex matching between the 2 groups, we suggest that for future studies, researchers use a larger population in which these parameters are matched.
The aim of this study was to evaluate the levels of MMP-2 and 9 in serum and salivary of HNSCC patients and compare them with a healthy group. Consistently with previous studies, the results of this study demonstrated that the serum levels of MMP-9 were considerably higher in patients compared with the control group. Although the levels of MMP-9 in salivary were higher in the case group, it showed no significant difference with the control group; however, it was correlated with the tumor histopathological grade. Furthermore, the salivary levels of this proteinase significantly correlate with the serum levels in the case group. In the present study, the serum and salivary levels of MMP-2 were not significantly different between case and control group. However, there was a significant correlation between its serum and salivary levels in the case group.
Since previous studies have also found the high levels of MMP-9 marker in the patients’ serum, it appears that if this conclusion would be proved in further studies, the serum levels of MMP-9 can be a reliable marker for diagnosis and analysis of the response to treatment in patients with HNSCC. Given the scarcity of the studies on the serum and salivary levels of MMP-9 and MMP-2 in patients with HNSCC, more extensive studies are needed in future.