1. Background
Prostate cancer (PCa) is the second most common cancer in men worldwide. The annual incidence is 759000 in the more developed region and 353000 in less developed regions. PCa death rate varies between 142000 in more developed regions and 165000 in less developed regions (1-3). In Iran, PCa is the 3rd most commonly-diagnosed visceral cancer (4-9).
When prostate-specific antigen (PSA) levels are abnormal or nodules are palpitated on digital rectal examination, transrectal ultrasound guided biopsy of the prostate (TRUS BX) should be done (10). The result of TRUSBX provides tumor data of the Gleason score (GS) (11). The GS system is the most useful system for the histopathological assessment of PCa worldwide (12) and is an important factor for the choice of treatment (13). GS includes two scores ranging from 1 to 5; the first score presents the most common patterns and the second score presents the highest grade patterns for the tumor (14). The accuracy of GS is important because any fault in the assessment and calculation leads to inappropriate approaches and complications (15, 16).
Pathologic staging is one of the outcome predictors of PCa (17). Currently, due to widespread PCa screening by using PSA level and due to increasing the use of TRUS Bx, assessing the accuracy of biopsy in predicting pathological grading and tumor aggressiveness is very important in the outcome of pathological samples of radical prostatectomy (RP) (17, 18).
The multifocal disposition of PCa is a common finding; therefore, it is expected that different areas within the prostate tissue would have different cancer grading. This is one of the diverse sources in the pathological reports of TRUS BX and RP. These reports show 41% to 43% exact accordance between samples. Accordingly, to these findings, TRUS BX GS accuracy is bolded in choosing the treatment plan (19-21).
Some studies report a discrepancy of pathological correlation between TRUS Bx and RP regarding GS. The upgradation of GS between the TRUS Bx sample and the final specimen from RP was reported in multiple articles. The accuracy among specimens extended from 30% to 74% (18, 22-28).
2. Objectives
In this study, we compared the pathology reports of initial biopsy and final pathology of the prostate after RP to determine the discrepancy among the Iranian population.
3. Methods
3.1. Sampling
In this retrospective observational study, 105 consecutive patients with a mean age of 66.1 ± 9.6 years old, who underwent both TRUS Bx and RP in Shohada-e-Tajrish Hospital from August 2009 to October 2017, were included in the study. The exclusion criteria included the patients, who had a history of chemoradiotherapy before RP surgery and the patients, who had incomplete medical records. Finally, 12 patients, who underwent radiotherapy before surgery or those who underwent TRUS Bx in other medical centers, were excluded from the study.
3.2. Pathologic Reassessment
TRUS Bx and RP specimens were reviewed by one expert specialized uropathology. The systematic biopsy was performed through the 12 biopsy scheme (2 core from the base, mid, and apex bilaterally). The patient underwent RP through the open retro-pubic approach. Upgrading was defined as a raise in GS in pathological specimens after RP. RP specimens were examined and reviewed based on the Stanford protocol. Each prostate lobe was divided into 2 sections of anterior and posterior. Whole specimens were sliced to 5 µm and stained, using hematoxylin-eosin.
3.3. Study Outcome
Demographics data, PSA serum levels, prostate size, and pathological stage and grade were recorded. The main aim of the study was to investigate the correlation between GS of needle biopsy and RP specimen.
3.4. Data Analysis
Statistical analysis was performed, using statistical package for social sciences (SPSS V. 19). For assessing qualitative data, the chi-square test was used. Quantitative outcomes were apprised through descriptive statistics (mean ± standard deviation) and independent t-test. The statistically significant level was considered P < 0.05.
3.5. Ethical Considerations
The Ethics Committee of Shohada-e-Tajrish Hospital approved this study and let us for review of patients’ medical data. The personal data of the subjects were not disclosed and the principles of patient secrecy were respected.
4. Results
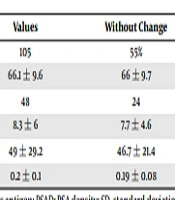
This study revealed that in TRUS BX samples, 80% had adenocarcinoma and 20% had acinar type adenocarcinoma. The final pathological diagnosis was observed in 2% of Prostatic intraepithelial neoplasia (PIN) cases, 25% of adenocarcinoma cases, 69% of acinar type cases, and 4% of non-malignant cases. In our study, 55% of cases show no change between TRUS BX and RP pathological specimens. Also, it is shown that 36% of the cases enrolled in the study have had an upgrade and the rest of them downgrade the tumor grading in the RP specimens compared with TRUS BX. The rate of abnormal digital rectal examination (DRE) and increase of PSA levels have a statistically significant correlation with the upgrading of GS (P = 0.001 and 0.02) (Table 1). The pathology reports of TRUS BX were adenocarcinoma and acinar adenocarcinoma in 80% and 20%, respectively; however, the last pathology reports were adenocarcinoma, acinar adenocarcinoma, high-grade PIN, and no malignancy in 25%, 69%, 2%, and 4%, respectively.
| Variable | Values | Without Change | Upgrade | P Value |
|---|---|---|---|---|
| No. Pts | 105 | 55% | 36% | |
| Age, mean ± SD | 66.1 ± 9.6 | 66 ± 9.7 | 66.4 ± 8.2 | 0.9 |
| Abnormal DRE, % | 48 | 24 | 23 | 0.001 |
| PSA, mean ± SD | 8.3 ± 6 | 7.7 ± 4.6 | 10.1 ± 7.4 | 0.02 |
| Mean prostate, volume ± SD | 49 ± 29.2 | 46.7 ± 21.4 | 51.3 ± 36 | 0.7 |
| PSAD | 0.2 ± 0.1 | 0.19 ± 0.08 | 0. 2 ± 0.17 | 0.6 |
Abbreviations: DRE, digital rectal examination; PSA, prostate-specific antigen; PSAD: PSA density; SD, standard deviation.
5. Discussion
The most important diagnostic test that leads to choosing the most appropriate approach is the pathological examination of biopsy samples. Therefore, it is of great importance to recognize the factors that affect biopsy samples results. Some studies show incompatibility between histopathological specimen examination of RP and TRUS Bx (18).
In our study, 55% of the cases show no change between TRUS BX and RP pathological specimens. Also, it is shown that 36% of the cases enrolled in the study have had an upgrade of the tumor grading in the RP specimens. We found that patients with abnormal DRE and high PSA levels are more prone to upgrade and have a statistically significant correlation with upgrading respectively (P = 0.001 and 0.02).
In 1998, Cecchi et al. evaluated tumor clinical grading and pathologic stage in association with GS and PSA levels. The study had 72 men enrolled. The patients underwent TRUS Bx and RP. Only 47.2% of the cases had the same GS in biopsy and final pathology; 37.5% of the cases were undergraded and 15.2% were overgraded. Clinical and pathologic stage was similar in 30.5% of the patients; 61.1% of the patients were understaged and 8.3% were overstaged. In conclusion, the GS in needle biopsy may be useful in predicting stage and grade (20), which was not included in our study. Today, with the advancement of imaging techniques, expert skills, and an increase in the number of biopsy cores from 6 to 12, the differences of pathologic reports between TRUS Bx and RP will be reduced. In the study of Khoddami conducted in 2016 in Iran, GS of needle biopsy and prostatectomy samples was the same in 68.2% of cases, while 31.8% had a difference of 1 or 2 in GS. Sensitivity and the positive predictive value was 86% and 79% in low-grade, 67% and 75% in intermediate, and 80% in high-grade tumors. In general, the reliability of Gleason grading from the needle biopsy was satisfactory in predicting the final pathology. The moderate-risk group was the most difficult to diagnose in needle sampling (19).
In the survey of Niroomand et al. in 2016, 36.7% of patients had similar GS in needle biopsy and pathology sampling. The weight of similarity was 46.4% and 33.3% among those with and without the perineural invasion (PNI). The highest similarity was in GS of 7 (% 63.6) and the lowest was in GS 8 to 10 (25%); however, there was no significant difference in patients with PNI (P > 0.05). Although the presence of PNI in the sample of TRUS Bx is associated with higher surgical procedures, PNI is not considered an independent factor in the classification of risk factors (21), which is similar to the results obtained in our research.
In 2012, Fine et al. revealed that pathologic results from needle biopsy and RP were similar and needle biopsy was reliable in the diagnosis of PCa. In our study, this theory was demonstrated (29). In the study conducted by Ojea in Spain in 2003, 97 men with PCa were survived, showing that the pathologic results of TRUS Bx and postoperative RP were statistically corresponding. These results were similar to those of our study (30). Bulbul et al. in 2007 conducted a study with 44 patients and showed that in unilateral tumors, the efficacy of cancer diagnosis by postoperative pathology is higher than primary TRUS Bx results. We did not focus on the unilaterality and bilaterality of tumors (17).
A comparative study of Lopez et al. conducted in 2006, which enrolled 1357 patients, revealed a great number of preoperative biopsies that were corresponding with RP pathology specimens (31). In comparison, our study showed the same result for every subject enrolled.
The aim of this study was the comparison of the pathology reports of initial biopsy and the final pathology of the prostate after RP to determine the discrepancy among the Iranian population. It is generally concluded that initial biopsy and RP pathological results are similar in our investigation. Lastly, we suggest further studies to confirm these findings. We recommend using a higher number of enrolled subjects, carrying out the study in a prospective manner, and taking into consideration other factors that can improve differential of prostate malignancies. This article is a retrospective study and it is better to be performed prospectively with the increase of the cases.
5.1. Conclusions
It is generally concluded that the initial biopsy with the final pathology of RP is similar in our investigation.