1. Background
Acute myeloid leukemia (AML) is the common type of hematology malignancy in children after acute lymphocytic leukemia (ALL) (1). The prevalence rate of AML is estimated at about 30 cases per 1 million people, who are younger than 20 years old in the United States (2). In Iran, the prevalence of hematologic malignancies is ranked sixth (3). However, AML is more common in adults than children and ALL is vice versa. According to research, life expectancy in patients with AML is 5 years in Iran and 50% of patients die before 5 years. Although its prevalence is not the same everywhere in Iran, children younger than 15 years old have reported 30 cases per 100,000 (4).
Leukemia was assumed initially as an incurable disease, but over the past decade, its survival rate has exceeded 90%. Despite recent advances in the management of pediatric AML, negative behavioral and cognitive side effects may persist after chemotherapy (5). Children with AML are prone to side-effects due to illness and treatment process, which increases the risk of behavioral and emotional problems during childhood and adolescence (6). Some studies reported different side effects during and after the treatment, which include more cognitive, behavioral, and emotional problems compared to healthy peers. Various factors such as gender, age of diagnosis, cancer severity, and duration of treatment can contribute to these problems (7). Chemotherapy may directly affect children's behavioral functioning. Systemic corticosteroids, which are the main elements of successful treatment, have behavioral side effects. Mood swing, irritability, depression, anxiety, inhibition problems, and attention during treatment with corticosteroids have been reported in patients with AML (8). They also experience obvious changes in their behavioral/emotional functioning because of painful treatments, frequent visits during chemotherapy, and hospitalization, which disturbs routine activities (9). Differences in the behavioral and emotional problems that have been reported in some studies may be justified according to the different types of leukemia, the risk of recurrence, the length of the illness, the severity of the therapeutic procedures, and the measurement scale. Recent studies reported that depression and anxiety are significant problems in the immediate post-diagnosis period for children with AML. While the prevalence of anxiety lessened after the first month of therapy, depression remains a significant problem (10). Also, recent studies have shown the risk of attention, learning and social problems, anxiety, and depression in children and adolescents with childhood cancer. A recent study found that 13% to 29% of adolescents with childhood cancer experience psychological stress and emotional dysfunction (11).
Chemotherapy has a negative effect on the cognitive function of children with AML. Early studies focused on the child's general intellectual ability and confirmed that chemotherapy does not have a significant negative effect on intellectual ability. Some recent research has focused on identifying the cognitive abilities of children with AML. They showed various types of neurologic deficits, including processing speed, attention, active memory, and executive function in children with AML, who were undergoing chemotherapy (12). These deficiencies include problems in planning, processing speed, and completing multiple assignments that can disrupt educational achievement and quality of life. Collectively, they are all referred to as executive function skills (13). Executive function (EF) is a set of cognitive processes that are necessary for goal-directed behavior, such as emotion regulation, active memory, planning, and cognitive flexibility. Executive functioning has a strong association with various aspects of life, including independence, marriage, educational achievement, and employment. Previous studies have confirmed executive dysfunction in children with AML, who were undergoing chemotherapy (10). Children with AML, who undergo chemotherapy, experience more cognitive impairment compared to those, who receive radiation therapy alone. However, according to most studies, these children are also at risk of defects in attention, processing speed, and executive functions (14). Children, who undergo chemotherapy treatment for AML, are at risk of neurotoxicity caused by chemotherapy. It occurs during the treatment and, then, continues. Chemotherapy treatment protocols often use high doses of intra spinal and intravenous Methotrexate (MTX) and corticosteroids, which recent studies have shown their neurotoxic effect. This is associated with long-term problems in memory, attention, processing speed, and executive functions. These cognitive dysfunctions, which have negative effects on a child's performance and quality of life, are characterized by comparing children with AML and their healthy peers. These can make changes in brain structure and function and create dysfunction in behavior regulation and metacognitive abilities (15). Some studies have reported increased fatigue and sleep problems in children with AML, which cause a negative effect on their behavior and neurological performance. Fatigue is associated with an impairment of processing speed, attention, and memory in children with AML, who have been treated (16).
The vulnerability to adverse psychological and neurological complications is increased in patients due to treatment advancement and increased survival rates of children with AML. In most previous studies, the psychological problems of children with ALL have been studied more than AML. Metastases to the central nervous system (CNS) occurred in the participants of this study that made the psychological state more complex. Furthermore, metastasis to the CNS has different consequences form other areas (5). In addition, previous studies have focused on cognitive problems and less on the emotional and behavioral problems in children with AML. Studying the behavioral problems of these children is necessary for the rehabilitation protocol design in the future. Since boys are more susceptible to this disease than girls, the present study focused on them.
2. Objectives
The aim of the current study was to compare the behavioral and emotional disturbances and executive functions in children with AML and healthy children.
3. Methods
This research is a descriptive causal-comparative study. We investigated the behavioral/emotional disorders and executive functions in children with AML and a healthy control group. The study population consisted of all male children with AML in the age range of 7 to12 years, who had been referred to Mahak Children's Hospital in Tehran from spring to summer 2019. The diagnosis was based on the patients' medical records. The sample size was calculated based on the Morgan table, and sampling was conducted randomly through the patients' list. All the patients with AML, who were referred to the Mahak Children’s Hospital during spring and summer, were listed in the patients' list and invited to participate in the study through phone calls. The sample consisted of 58 children with AML and 58 healthy children, who were homogeneous with the control group, in terms of socioeconomic status and age. The inclusion criteria included: (1) boy children aged between 7 and 12 years, (2) the diagnosis of AML with CNS involvement, (3) treatment with chemotherapy, and (4) the lack of previous participation in psychology courses from the disease diagnosis. The exclusion criteria included: (1) having other diseases simultaneously and (2) having a history of psychiatric disorders. The gender variable was controlled because it plays an important role in the incidence of behavioral and emotional disorders among girls and boys. So, only boys participated in this study. Before beginning the study, the consent form was completed by parents that enabled them to withdraw from the research at any time. The ethical code was approved by the Iran national committee for ethics in Biomedical Research, University of Tehran. Data analysis was done by the Mann-Whitney and t-test, using SPSS V. 24. Informed consent was taken from the patients, and they were informed of the therapeutic goals before the intervention program launched. Their satisfaction was drawn to participate in the research. They were assured of the disguise of personal information and told that they could leave the research process whenever they want.
3.1. Tools
3.1.1. Child Behavior Checklist (CBCL)
CBCL is a component of the Achenbach System of Empirically Based Assessment (ASEBA) that contains 118 problem behavior items. Usually, a parent or caregiver rates child problems. CBCL includes syndrome and skills in daily life. The syndrome scale includes anxious/depressed, withdrawn/depressed, somatic complaints, social problems, thought problems, attention problems, rule-breaking behavior, aggressive behavior, and other behavior problems scale, which constitute a heterogeneous set of different problems such as behaving in a heterosexual manner, defiance, eating avoidance, school phobia, nail-biting, nightmares, overeating, weight gain, anorexia, etc. The competence scale includes activities and social skills. Generally, CBCL consists of the several following parts: (1) academic performance assessment of children and adolescents in the fields of cognitive ability, training, and education; (2) social skills assessment of children and adolescents for evaluating their adaptation with peers, siblings, and parents and how they cope with themselves; and (3) assessment of emotional and behavioral problems in children and adolescents. The internal consistency of the CBCL scale was estimated, using Cronbach's alpha coefficient. Alpha coefficient of the competence scale was relatively high and its range was between 65% and 85% for CBCL. The test reliability by using Cronbach's alpha was 0.89 for boys and 0.94 for girls. The test reliability by using split-half reliability was 0.84 for boys and 0.87 for girls. The results of the construct validity supported the 8-factor structure of this scale by using factor analysis in Iran and convergent validity of this scale with J-EPQ and Rutter behavioral problems questionnaire was satisfactory. If the index of discriminating power, which is related to variance and score distribution gets higher than 90%, the scale will be good. The obtained coefficients in the CBCL form were at the highest level (17).
4. Results
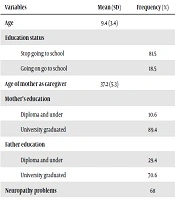
The demographic information of 58 children with AML is demonstrated in Table 1.
| Variables | Mean (SD) | Frequency (%) |
|---|---|---|
| Age | 9.4 (3.4) | |
| Education status | ||
| Stop going to school | 81.5 | |
| Going on go to school | 18.5 | |
| Age of mother as caregiver | 37.2 (5.3) | |
| Mother’s education | ||
| Diploma and under | 10.6 | |
| University graduated | 89.4 | |
| Father education | ||
| Diploma and under | 29.4 | |
| University graduated | 70.6 | |
| Neuropathy problems | 68 | |
| Dizziness | 21 | |
| Headache | 29 | |
| Seizures | 43 | |
| Fatigue | 78 | |
| Anemia | 85 | |
| Relapse experience | 43 | |
| Time diagnosis (in a month) | 37.2 (4.3) |
In this study, it was assumed that children with AML behave differently from healthy children in behavioral-emotional and cognitive-behavioral dimensions.
The Kolmogorov-Smirnov test was applied to test normal distribution. It indicated the normal distribution of externalizing and internalizing problem variables in experience-based scales (P > 0.05). Therefore, an Independent t-test was used to analyze the findings of the mentioned variables and other variables were analyzed by the Mann-Whitney test.
| Variable | Healthy Children | Children with AML | t | P Value |
|---|---|---|---|---|
| Externalizing problems | 8.56 ± 1.08 | 11.57 ± 4.57 | 3.84 | 0.001 |
| Internalizing problems | 7.54 ± 1.95 | 13.57 ± 3.47 | 7.02 | 0.001 |
| General problems | 8.86 ± 5.73 | 10.37 ± 6.36 | 4.48 | 0.05 |
Abbrivation: CBCL: Child Behavior Checklist
aValues are expressed as mean ± SD.
According to Table 2, the result of the t-test indicated that the average scores of externalizing and internalizing problems in children with AML are significantly lower than the control group (P > 0.001) and general problems in these children are significantly higher than healthy children (P > 0.05).
| Variable | Healthy Children | Children with AML | Mann-Whitney U | P Value |
|---|---|---|---|---|
| Anxious/depressed | 5.86 ± 2.33 | 6.16 ± 0.79 | 268 | 0.075 |
| Withdrawn/depressed | 3.58 ± 0.65 | 14.67 ± 0.58 | 223 | 0.001 |
| Somatic complaints | 3.64 ± 1.14 | 6.47 ± 2.43 | 343 | 0.001 |
| Social problems | 4.37 ± 1.23 | 6.53 ± 1.34 | 284 | 0.001 |
| Thought problems | 6.29 ± 2.34 | 8.21 ± 1.23 | 383.5 | 0.05 |
| Attention problems | 4.86 ± 1.75 | 8.32 ± 3.45 | 566.5 | 0.001 |
| Rule-breaking behavior | 7.35 ± 0.76 | 3.76 ± 0.12 | 572 | 0.001 |
| Aggressive behavior | 9.68 ± 2.86 | 4.85 ± 2.34 | 368 | 0.001 |
| Activities | 7.24 ± 2.11 | 5.07 ± 1.47 | 298 | 0.02 |
| Social function | 7.54 ± 1.54 | 4.96 ± 0.08 | 562 | 0.001 |
| General problems | 8.86 ± 5.73 | 10.37 ± 6.36 | 321 | 0.06 |
| General competence | 16.71 ± 5.09 | 15.75 ± 4.58 | 255.24 | 0.086 |
| Emotional problems | 6.47 ± 2.35 | 6.24 ± 3.01 | 637 | 0.27 |
| ADHD | 8.16 ± 3.47 | 8.93 ± 1.28 | 604.51 | 0.145 |
| PTSD | 4.52 ± 3.24 | 5.11 ± 2.38 | 216.1 | 0.11 |
Abbreviations: ADHD, attention deficit hyperactivity disorder; PTSD, post-traumatic stress disorder.
aValues are expressed as mean ± SD.
As shown in Table 3, the participants obtained lower scores in social function, activities, aggressive behavior, and rule-breaking behavior, but there was no significant difference in PTSD, emotional problems, general competence, general problems, and anxious/depressed behaviors. Also, compared with the control group, they obtained higher scores in attention problems, thought problems, social problems, somatic complaints, and withdrawn/depressed behaviors.
5. Discussion
The main purpose of the study was the comparison between children with AML and healthy children in behavior and emotion. Patterns of psychological reaction can solve important puzzles of children with leukemia. Before explaining the results of the study, let us take a look at the history of research on the psychological problems of children with cancer. A number of studies have shown that there is no significant difference in the emotional deficit between children with leukemia and the healthy group (18). Even their psychological problems are minor compared to children with other chronic diseases (19). Other studies have suggested that although these differences are not significant, children with leukemia showed less vitality (20). The findings displayed children with cancer used to suppress their negative emotions. In other words, they are less likely to disclose their behavioral problems (21). Other studies have found that the destruction of emotions is associated with suppression or decreased levels of cortisol as an emotion-regulating hormone. Chemotherapy is a factor that plays a role in lowering cortisol levels (22). Other studies have found that behavioral perturbation syndrome can be used as a predictor for the physical deterioration of children with leukemia. The full-suppression of cortisol deactivates the immune system, causing emotional arousal and disturbing behaviors (23).
In this research, we compared the behavioral and emotional disorders of children with AML to healthy children. However, the difference between ill and healthy children was not the same in all dimensions. For example, PTSD, emotional problems, general competence, general problems, and anxious/depressed behaviors demonstrated no difference with healthy peers. This finding is in line with previous studies such as Tillery et al. (18), Murphy et al. (24), and Han et al. (25). According to the findings, children with AML obtained lower scores in aggressive behavior and rule-breaking behavior compared to the control group. The unexpected finding may be due to the treatment improvements in recent decades, which reduced the negative effects of illness and treatment on various aspects of a child's life. Despite advanced therapies, these children are less likely to be hospitalized than before, which can improve their psychological experience. Also, increasing parental awareness and families about the disease, as well as reducing the impact of negative stereotypes of peers in relation to chronic diseases in recent decades may help increase the social acceptance among peers and community; also, it makes them most engaged in all kinds of activities and interactions like their peers.
Children with AML obtained higher scores in attention problems, thought problems, social problems, somatic complaints, withdrawn/depressed behaviors, and low social function. This finding is consistent with the study of Tamnes et al. (26), which showed that children with AML have higher scores in anxious/depressed behaviors, withdrawn/depressed behaviors, and social and internalizing problems variables compared to healthy children. Graef et al. (27) also found that childhood cancer survivors have more anxiety, depression, and antisocial behaviors. Warris et al. (28), Viola et al. (29), and Plas et al. (30) also showed more behavioral and emotional problems in children with AML after the treatment, which is consistent with the present study.
Metastasis to CNS affects brain activity of participants. CNS involvement can be regarded as a considerable factor in exacerbating the behavioral-emotional problems of patients. In the present study, the participants received intensive chemotherapy because of metastasis. Patients undergoing high-dose chemotherapy experience more mental and social difficulties and need rehabilitation (31).
Compared with other kinds of leukemia, AML is intensive and need aversive chemotherapy. A high dose of pharmacy could affect cognitive function such as attention and thinking process. A meta-analysis showed that the physical, depressive, social, and role function of patients with AML depends on their treatment (31). The higher the dose of chemotherapy, the worse the performance. This factor may also explain the difference between the results of this study and other studies. Children with AML have a worse prognosis. They receive higher doses of chemotherapy and more hospitalization. These factors can explain psychological problems in this group. On the other hand, mothers, who are overwhelmed by the burden of the disease, might be excessively sensitive to their child's behaviors and report their performance with bias and over protection.
Painful medical procedures, prolonged periods of illness, and physical problems are probably related to cognitive deficits that these children may experience after diagnosis. The diagnosis of childhood cancer is one of the most stressful experiences for children. Also, these children are being exposed to social and educational deprivation and sometimes overprotection that prevents independence.
Depression may be caused by illness-related deprivations, reduced child engagement in academic performance, and treatment side-effects that often affect the brain structure and function in these children. Withdrawal from school and limited interaction with peers during childhood reduce behavioral and cognitive development and acquisition of cognitive abilities. Furthermore, depression and low activity may be due to the conditions of hospitalization. Sometimes the environment of the hospital would be unpleasant for children; therefore, emotional and behavioral problems may be due to independent cancer and its treatment.
Children with other types of leukemia have been less studied. In addition, most prior studies had focused on the behavioral disruption in cancer survivors and paid less attention to active patients. If all children with cancer are equally studied, information about children with different characteristics will be lost. This study has attempted to pay more attention to details.
One of the limitations of this study is associated with CBCL that parents or nurses report about a child’s behaviors. Usually, there is a gap in other reports and reality (32). Measuring behavioral problems with more accurate tools may lead to different results. For example, experimental methods that directly measure children's behavior are preferred to questionnaires. In addition, one of the positive points of the study was the control of affective variables such as sex, stage of cancer, and location of metastases. Anyway, strict in selective samples could reduce the generalizability of the results. In the upcoming investigates, it is recommended to compare the psychological status of girls and boys with AML and patients with nervous system involvement with patients without metastases. One of the research limitations was the small sample size. It is recommended to consider a larger sample in future studies. Also, the severity of leukemia problems should be compared with other chronic diseases in future studies.
5.1. Conclusions
The results indicate that some factors associated with illness (such as anemia, hormone dysregulation, and immune system deficit) and treatment (chemotherapy) caused some behavioral, emotional, and cognitive problems in these children.