1. Context
Literally, “cure” means medical treatment that makes an illness disappeared (1). Physicians have employed various definitions for “cure” over the centuries. Graeco-Roman and Persian physicians, the pioneers in medicine, had defined “cure” as maintenance of the health like it was before the illness. This definition, called “restitutio ad integrum”, means the return of body status to complete health without any medications or special assistant for maintenance of health. Avicenna (980 - 1037 AD) wrote: “medicine is a science from which one learns the states of the human body with respect to what is healthy and what is not, in order to maintain good health when it exists and to restore it when it is lacking” (2). The above-mentioned concept was appropriate for communicable diseases. In this regard, patients affected by the majority of bacterial and viral infections, if did not die, would have returned to complete a healthy situation, as it was before the illness. In recent centuries and in particular, in the last few decades, non-communicable chronic diseases (NCDs) have emerged, and many treatment approaches and management of disorders have been discovered. This article aims to review the meaning of “cure”, particularly in diseases of the thyroid gland, in current medicine.
2. Search Strategy
The terms including “cure” and “control” with “thyroid disease”, “hypothyroidism”, “hyperthyroidism”, “thyroiditis”, and “nodular goiter” were used to search in MEDLINE for articles published between January 1980 and December 2019. After abstract screening, potential eligible articles were retrieved, and full-text articles were studied. Articles with definitions of cure or control, in particular, those related to various thyroid diseases, were included in this narrative review.
3. Results
With emerge of NCDs, maintenance of health conditions may require continuous drug(s) administration or causing another disease in order to treat the original disease (3). Physicians named this form of care “cure of the disease”, despite the fact that the status of the human body has not returned completely to like it was before the illness. On the other hand, such patients may be asymptomatic and feel well, but their body composition as well as cellular and humoral factors differ from unaffected individual. In addition, an unhealthy condition will return upon the discontinuation of any therapeutic approach, which the physician has prescribed. Nowadays, this concept of cure is employed for all relapse-free conditions, although the disease may relapse and may cause morbidity or mortality in the future (4). To distinguish these concepts, we may use “restitutio ad integrum” as a cure and controlled status.
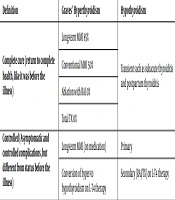
Regarding thyroid diseases, most conditions are managed as “controlled; and only in few diseases, the status of thyroid returns to the complete cured condition. Table 1 shows a classification of cure into complete and controlled cure; In fact, a complete cure is defined as return to complete health. While, controlled cure is defined as a condition that the patient becomes asymptomatic and complications of the original disease are controlled; However, the health status of the patient is different from what it was before. Interestingly, hypothyroidism management rarely results in a complete cure. To better words, there are transient hypothyroidisms such as the recovery phase of subacute thyroiditis in which thyroid structure and function return to a healthy condition. The same may happen in < 50% of women who experience postpartum thyroiditis, in which after 12 months, serum level of TSH returns to the normal range without levothyroxine treatment (5). In this line, patients with primary hypothyroidism and secondary form, which induced by ablative doses of radioiodine as well as total thyroidectomy, require lifelong levothyroxine medication for maintaining euthyroidism and therefore, are classified as under controlled hypothyroidisms (6).
| Definition | Graves’ Hyperthyroidism | Hypothyroidism | Nodular Thyroida |
|---|---|---|---|
| Complete cure (return to complete health, like it was before the illness) | Long-term MMI 85% | Transient such as subacute thyroiditis and postpartum thyroiditis | Lobectomy or RAI without L-T4 therapy |
| Conventional MMI 50% | |||
| Ablation with RAI 0% | |||
| Total TX 0% | |||
| Controlled (Asymptomatic and controlled complications, but different from status before the illness) | Long-term MMI (on medication) | Primary (on L-T4 therapy) | Total TX on L-T4 therapy |
| Conversion of hyper-to hypothyroidism on L-T4 therapy | Secondary (RAI/TX) on L-T4 therapy |
Abbreviations: L-T4, levothyroxine treatment; MMI, methimazole; RAI, radioactive iodine; TX, thyroidectomy.
aInclude benign, malignant, toxic or euthyroid nodular goiter.
In patients with nodular goiter, benign or malignant, who are euthyroid or toxic multinodular goiter, only those who undergo lobectomy or radioiodine therapy and have no need to take levothyroxine would be categorized in complete cure group. All other patients with nodular goiter who are treated with levothyroxine following total thyroidectomy or radioiodine are categorized into the control treatment modality.
Graves’ hyperthyroidism is a rather common disease with a prevalence of 1% - 1.5% (7). The majority of patients have a prolonged course of treatment with relapsing and remitting episodes over the years (8). The strategy for Graves’ hyperthyroidism treatment is not straightforward and requires complex decision making. None of the three forms of current therapeutic approaches, i.e., radioiodine (RAI), surgery or anti-thyroid drugs (ATD), have been able to re-establish continuous normal thyroid function in all patients (9). According to the guidelines, the aim of thyroidectomy should be induction of permanent hypothyroidism (6, 9). This is due to the subtotal thyroidectomy which may be accompanied by 29% persistent or recurrent hyperthyroidism and 50% hypothyroidism; in this respect, only 19% of patients may remain euthyroid after 6 years of follow-up (10). To note, the recurrence rate after sub-ablative doses of RAI is 61% - 69% (11). In addition, one year following ablative RAI treatment, 77%, 17%, and 6% of patients were hypo-, hyper-and euthyroid, respectively; which 14% of patients might require the second RAI therapy. In this case, the rate of hypo-, hyper and euthyroidism were 86, 3.2 and 3.3%, respectively with 6.4% death after 80 months follow-up (12). Notably, both total thyroidectomy and ablative RAI therapy cause permanent hypothyroidism in the majority of patients as well as causing another disorder and requiring lifelong levothyroxine treatment for maintaining euthyroidism. Therefore, both methods fulfill criteria of controlled management. Levothyroxine replacement may cause disruption in psychological condition, lower resting energy expenditure, lower T3:T4 ratio, higher body mass index (BMI) and increased levels of serum cholesterol and LDL compared with normal controls (13-17). Some of these findings have also been reported in RAI treated Graves’ patients on levothyroxine as compared to euthyroid Graves’ patients (18). The main drawback of ATD therapy has been the high rate of hyperthyroidism relapse, mostly up to 4 years after drug withdrawal (19). The rate of recurrence has been also reported between 20% - 70% with an average of about 50% after conventional 1 - 2 years of ATD treatment (6). Recently, it has also been reported that long-term ATD treatment induces higher remission rates than conventional therapy (< 2 years) (20, 21), and two meta-analyses have shown efficacy (22) and safety (23) of continuous long-term treatment with ATD. In addition, a randomized trial in 302 patients with Graves’ hyperthyroidism has demonstrated a relapse rate of 16% in those treated with methimazole (MMI) for 60 - 120 months, as compared to 51% of patients who received conventional MMI therapy four years after discontinuation of medication (24).
Table 2 shows the remission possibility of Graves’ hyperthyroidism after a course of long-term ATD treatment. Between 12 - 22 years after treatment, only 27% of those treated with ATD for 2 years or less, remain euthyroid. 62% have recurrence, 80% have subclinical hypothyroidism, and 3% overt hypothyroidism (25). Moreover, long-term treatment with MMI for 5 - 10 years (on average, 8 years) results in only 16% recurrence 4 years after discontinuation of MMI (24). There are no data regarding the longer follow up rate of recurrence of overt hyperthyroidism, the rates of subclinical hyperthyroidism, and overt hyperthyroidism in patients who treated with long-term MMI. Given that the statistics results regarding in long-follow up of those patients who were in remission after the conventional therapy (25), if the same rate of 40% reduction in euthyroidism occurs in long-term MMI treated patients, by fulfilling the criteria for complete cure, approximately 50% of them remain euthyroid 22 years after onset of treatment.
4. Conclusions
The concept of “cure” meaning, indicates the return of thyroid status to complete health condition without any medications applied only to a few conditions in thyroid diseases. Majority of patients with hypothyroidism require lifelong levothyroxine treatment to maintain euthyroidism. In addition, many patients with benign or malignant nodular goiter undergo thyroidecomy or RAI treatment and also require permanent levothyroxine treatment. Patients with subacute or postpartum thyroiditis may attain complete cure without further treatment modalities. Among all modes of treatment for Graves’ hyperthyroidism (i.e surgery, RAI therapy, conventional ATD and long-term ATD treatment), the latter causes more percentage of Graves’ patients to attain cure, in which thyroid function returns to like it was before without received ongoing treatment.