1. Background
Thyroid disorders are the second most frequent endocrine disorder in women of childbearing age (1). Hypothyroidism, with a prevalence of 2% - 5%, is the most common gestational thyroid dysfunction (2, 3). According to previous reports, the prevalence of hypothyroidism (both overt and subclinical) is estimated at 4.7% in Iranian pregnant women (4). Numerous studies have addressed the adverse effects of overt hypothyroidism (OH) on pregnancy outcomes (5-7), despite controversies about the adverse fetal-maternal effects of subclinical hypothyroidism (SCH) (8). Proper screening can lead to early diagnosis of these disorders in pregnancy. However, universal screening for diagnosis of thyroid dysfunction in pregnancy is a controversial topic (9, 10).
Evidence shows that the selective high-risk case-finding approach may overlook a considerable number of gravid women with overt thyroid dysfunction (11, 12). As a result, having access to a convenient tool with an acceptable diagnostic value in detecting thyroid disorders during pregnancy has its merits. Apart from the universally advocated laboratory assessment of thyroid function, several questionnaires have been designed for diagnosis of thyroid dysfunctions (13). These clinical tools are definitely not intended to replace blood measurements, but are reliable tools in low-income settings with limited access to costly tests; they can be also useful in posttreatment follow-ups.
Although validity of these clinical tools has been confirmed in general populations, no published study is available about their validation for pregnant women. Among various available tools, the Billewicz scoring index, as a cost-effective clinical index, was validated against thyroid iodine uptake and plasma protein-bound iodine in 1969 and against thyroid-stimulating hormone (TSH), triiodothyronine (T3), and free thyroxine (FT4) in 1997 (14, 15). Despite the convenience and accuracy of this tool in diagnosis of hypothyroidism in general populations, no information exists regarding Billewicz scale verification during pregnancy; therefore, the optimal pregnancy-related cut-off value has not been established yet.
In the present study, we aimed to assess the validity of Billewicz scale in a pregnant population, selected from Tehran thyroid and pregnancy study (TTAPS) to determine the optimal cut-off point of the Billewicz index with the highest sensitivity and specificity for predicting OH.
2. Methods
Data were collected from the first phase of TTAPS, which was carried out on pregnant women, attending the prenatal clinics of Shahid Beheshti Medical University. The details of TTAPS are published in the literature (16). All women were screened for thyroid dysfunction by collecting information from their medical history, clinical examinations, and serum thyroid peroxidase antibody (TPOAb), TSH, T4, and T-uptake measurements. They were then classified as follows: normal thyroid, OH, overt hyperthyroidism, TPOAb-positive euthyroidism, SCH, and subclinical hyperthyroidism. Subjects with twin pregnancies (n, 27) or history of chronic disorders were excluded from the study. Of a total of 1,843 pregnant women, 1,264, 38, and 541 cases exhibited euthyroidism, OH, and SCH, respectively.
In this study, the Billewicz scale was applied. This scale was developed based on seven symptoms and six signs, which were typically associated with hypothyroidism. According to this scale, higher positive scores indicate a higher level of clinical hypothyroidism. Items are weighed differently, based on their frequency of occurrence (see supplementary file, appendix 1) (13). A questionnaire related to the symptoms described in the Billewicz scale was completed, and a thorough physical examination of Billewicz signs (i.e., slow movement, coarse skin, cold skin, periorbital puffiness, bradycardia, and delayed ankle reflex) was performed. After collection and centrifugation of fasting blood samples, the sera were sent to the Research institute of endocrine sciences of Shahid Beheshti University of Medical Sciences for laboratory assessments.
Using radioimmunoassay and immunoradiometric assay, T4 and TSH levels were measured, respectively by commercial kits (Izotop Kit, Budapest Co., Hungary) and a gamma counter (Dream Gamma-10, Goyang-si, Gyeonggi-do, South Korea). T-uptake and TPOAb were also determined by the enzyme immunoassay (Diaplus Kit, San Francisco, CA, USA) and immunoenzymometric assay (Monobind Kit, Costa Mesa, CA, USA), respectively, using a calibrated ELISA reader (Sunrise, Tecan Co., Salzburg, Austria). The inter- and intraassay coefficients of variation for T4, T-uptake, TSH, and TPOAb were 1.1%, 3.9%, 2.2%, and 4.3% and 1.9%, 4.7%, 1.0%, and 1.6%, respectively.
2.1. Definitions
Overt hyperthyroidism was defined as TSH level < 0.1 μIU/mL and FT4I > 4.5. OH was defined as TSH > 10 μIU/mL or TSH > 2.5 μIU/mL and FT4I < 1. SCH was defined as normal FT4I (1-4.5), despite elevated TSH (2.5-10 μIU/mL). Finally, subjects with TPOAb > 50 IU/mL were considered TPOAb-positive. It should be noted that when we first designed TTAPS and initiated data acquisition (from September 2013 to February 2016), the available guideline was the previous American thyroid association guideline (10). Accordingly, SCH was defined based on the common TSH cut-point of 2.5 μIU/mL instead of the recently recommended cut-point of 4.0 μIU/mL (17). In the Billewicz questionnaire, more than a 1.5-fold increase in weight (e.g., more than 8 kg in the second trimester for those with a normal prepregnancy BMI) was considered as abnormal weight gain. Also, bradycardia was defined as pulse rate < 75 bpm.
2.2. Ethics Approval
Written informed consents were obtained from all the participants, and the study was approved by the ethics committee of research institute of endocrine sciences (code, IR.SBMU.ENDOCRINE.REC.1396.383).
2.3. Statistical Analysis
Continuous variables were assessed for normality using one-sample Kolmogorov-Smirnov test. Categorical variables, expressed as percentages, were compared using Pearson’s Chi square test. Normal continuous variables were compared between the three groups, using one-way ANOVA test and expressed as mean (standard deviation). Variables without a normal distribution were expressed as median (interquartile) and compared using Kruskal-Wallis test.
First, we classified our participants into three groups, using the cut-off values reported by Billewicz and colleagues: score ≥ +25 for OH; score of -30 to +25 (above -30 and below +25) for SCH; and score ≤ –30 for excluding hypothyroidism. A receiver operating characteristics (ROC) curve analysis was performed, and the optimal cut-off value was identified, using the Youden index for differentiating OH from euthyroidism. In fact, the Youden index maximizes the difference between sensitivity and 1-specificity (Youden index: sensitivity + specificity-1). Therefore, by excluding SCH patients and maximizing sensitivity + specificity-1 across various cut-off points for the Billewicz index, the optimal cut-off point was calculated to distinguish between healthy subjects and patients with OH (18). Data were analyzed using SPSS version 22, and level of significance was set at P < 0.05.
3. Results
The mean (SD) age, BMI, and gestational age of the participants were 26.6 (5.3) years, 25.1 (4.6) kg/m2, and 11.8 (4.1) weeks, respectively. Table 1 presents the characteristics of the study population according to their thyroid status. The median and interquartile range for TSH in euthyroid, OH, and SCH women were 1.46 (0.81 - 1.91), 7.92 (3.32 - 11.24), and 3.79 (3.06 - 4.96) μIU/mL, respectively. These values show that women with OH had significantly higher TSH levels, compared to the other two groups.
| Characteristics | Euthyroidism (n, 1264) | Subclinical Hypothyroidism (n, 541) | Overt Hypothyroidism (n, 38) | P Value |
|---|---|---|---|---|
| Age, yb | 26.68 ± 5.30 | 26.32 ± 5.16 | 27.84 ± 5.01 | 0.140 |
| Body mass index, kg/m2b | 24.85 ± 4.53c | 25.62 ± 4.75d | 27.47 ± 5.47 | < 0.001 |
| Gestational age, wke | 11 (8 - 14)d | 12 (8 - 16) | 10 (7 - 14) | < 0.001 |
| Gestational age < 14 wkf | 880 (69.81) | 332 (61.5)d | 25 (65.8) | 0.003 |
| Primigravidaf | 442 (35) | 213 (39.4) | 16 (42.1) | 0.156 |
| TSHc | 1.46 (0.81 - 1.91)d | 3.79 (3.06 - 4.96)g | 7.92 (3.32 - 11.24)c | < 0.001 |
| T4e | 10.5 (8.8 - 12.5) | 10.3 (8.7 - 12.4) | 10.2 (8.4 - 13.02) | 0.320 |
| FTIe | 2.9 (2.5 - 3.4) | 2.8 (2.3 - 3.2)d | 2.8 (2.3 - 3.22) | 0.420 |
| TPOAbe | 4 (2 - 8)d | 7 (3 - 28)g | 46 (5.5 - 445)c | < 0.001 |
| T3 uptakee | 28 (25 - 30)c | 27 (24 - 29)d | 26.5 (23 - 29) | < 0.001 |
Characteristics of the Study Population According to Their Thyroid Statusa
The mean±SD age of OH, euthyroid, and SCH women was 27.84 ± 5.01, 26.68 ± 5.30, and 26.32 ± 5.16 years, respectively. Euthyroid women had a significantly lower BMI, compared to OH and SCH groups (24.85 ± 4.53 versus 27.47 ± 5.47 and 25.62 ± 4.75). The prevalence of TPOAb positivity was 3.6%, 18.9%, and 50% in euthyroid, SCH, and OH groups, respectively. In addition, the median and interquartile range of TPOAb in euthyroid, OH, and SCH groups were 4 (2 - 8), 46 (5.5 - 445), and 7 (3 - 28) μIU/mL, respectively (Table 1).
Distribution of various risk factors for thyroid disorders showed that history of levothyroxine treatment and history of thyroid dysfunction were the two most common risk factors for OH in women. Also, age ≥ 30 years and thyroid antibodies, primarily TPOAb, were the most common risk factors for SCH (Table 2). Among euthyroid pregnant women, 491 (38.5%) exhibited at least one sign or symptom of the Billewicz index. Based on the findings, 257 (47.5%) and 34 (89.5%) SCH and OH women had more than (or equal to) one Billewicz sign or symptom, respectively. The mean ± SD Billewicz score was -17.11 ± 13.63 in the OH group, which is significantly different from that of euthyroid (-41.16 ± 11.16; P < 0.001) and SCH (-40.1 ± 11.2; P < 0.001) women.
| Risk Factors | Euthyroidism (n, 1264) | Subclinical Hypothyroidism (n, 541) | Overt Hypothyroidism (n, 38) | P Value |
|---|---|---|---|---|
| History of thyroid dysfunction | 26 (2.1)b | 35 (6.5)c | 22 (57.9)d | < 0.001 |
| History of levothyroxine treatment | 7 (0.6)b | 1 (0.2)c | 23 (60.5)d | < 0.001 |
| History of treatment with radioactive iodine | 2 (0.2) | 0 (0.0) | 0 (0.0) | - |
| History of therapeutic head or neck irradiation or thyroid surgery | 0 (0.0)b | 2 (0.4) | 1 (2.6)d | - |
| Family history of autoimmune thyroid disease or thyroid | 127 (10.1)b | 76 (14)c | 11 (28.9)d | < 0.001 |
| Goiter | 25 (2)b | 6 (1.1)c | 4 (10.5) | < 0.001 |
| TPOAb | 45 (3.6)b | 102 (18.9)c | 19 (50)d | < 0.001 |
| Type I diabetes mellitus or other autoimmune disorders | 13 (1)b | 0 (0.0) | 0 (0.0) | - |
| History of miscarriage | 200 (15.9) | 89 (16.5) | 6 (15.8) | 0.951 |
| History of preterm delivery | 21 (3.0) | 8 (3.0) | 0 (0.0) | 0.758 |
| Infertility | 62 (4.9) | 23 (4.3)c | 8 (21.1)d | < 0.001 |
| Morbid obesity (BMI ≥ 40) | 7 (0.6) | 1 (0.2)c | 2 (6.1)d | - |
| Age ≥ 30 y | 387 (30.7) | 144 (26.6) | 16 (42.1) | 0.054 |
Risk Factors for Thyroid Disorders in the Participants According to Their Thyroid Statusa
Weakness/fatigue and laziness/sleepiness were the two most common symptoms in all the groups (Table 3). In total, 34 out of 38 women with OH showed delayed ankle reflexes; this sign was observed in six out of 541 women with SCH and 10 out of 1,264 euthyroid pregnant women. In addition, pulse rate < 75/minute was observed in 23.7%, 21.8%, and 15.4% of women with OH, SCH, and euthyroidism, respectively (Table 3).
| Symptoms and Signs | Euthyroidism (n, 1264) | Subclinical Hypothyroidism (n, 541) | Overt Hypothyroidism (n, 38) | P Value |
|---|---|---|---|---|
| Symptoms | ||||
| Diminished sweating | 22 (1.7) | 4 (0.7) | - | 0.193 |
| Dry skin | 85 (6.7) | 48 (8.9) | 3 (7.9) | 0.276 |
| Cold intolerance | 130 (10.3) | 62 (11.5) | 8 (21.1) | 0.095 |
| Weight gain | 34 (2.7) | 23 (4.3) | 2 (5.3) | 0.173 |
| Constipation | 159 (12.6) | 91 (16.80)b | 8 (21.1)c | 0.026 |
| Hoarseness | 12 (3) | 4 (2.7) | - | 0.849 |
| Hearing impairment | 8 (2) | 10 (6.8)b | - | 0.015 |
| Weakness/fatigue | 407 (32.2) | 161 (29.8) | 14 (36.8) | 0.463 |
| Laziness/sleepiness | 264 (20.9) | 156 (28.8)b | 14 (36.8)c | < 0.001 |
| Signs | ||||
| Slow movement | 36 (9.0) | 13 (8.9) | 3 (30) | 0.077 |
| Coarse skin | 85 (6.7) | 48 (8.9) | 3 (7.9) | 0.276 |
| Periorbital puffiness | 53 (12.7) | 26 (16.3) | 4 (30.8) | 0.121 |
| Cold skin | 85 (6.7) | 48 (8.9) | 3 (7.9) | 0.276 |
| Pulse rate (< 75/min) | 195 (15.4) | 118 (21.8)b | 9 (23.7)c | 0.003 |
| Delayed ankle reflex | 10 (0.8) | 6 (1.1) | 34 (89.5)c | < 0.001 |
Signs and Symptoms of the Patients According to the Billewicz Index Based on Their Thyroid Statusa
Using the cut-off point suggested by Billewicz et al. (score ≥ +25 for OH; -30 to +25 [> -30 and < +25] for SCH; and ≤ -30 for excluding hypothyroidism), we observed a significant difference in the TSH level of OH patients and those with SCH and euthyroidism. Based on these cut-off values, 474 (29.4%) out of 1613 participants were categorized as the normal group, while based on the TSH levels, they were categorized as SCH. Also, 125 (54.3%) and 38 (16.5%) out of 230 women, who were categorized as euthyroid and OH based on the TSH values, respectively, were diagnosed with SCH. On the other hand, no subject was diagnosed with OH based on the Billewicz score, as the maximum score of Billewicz index is +24.
The mentioned values corresponded to sensitivities of 12.38%, 100%, and 100% and specificities of 90.11%, 90.11%, and 87.61% for distinguishing euthyroidism from SCH, euthyroidism from OH, and SCH from OH, respectively. These values yielded positive predictive values of 34.89%, 23.31%, and 36.19% and negative predictive values of 70.61%, 100%, and 100%, respectively in discrimination between euthyroidism and SCH, euthyroidism and OH, and lastly SCH and OH.
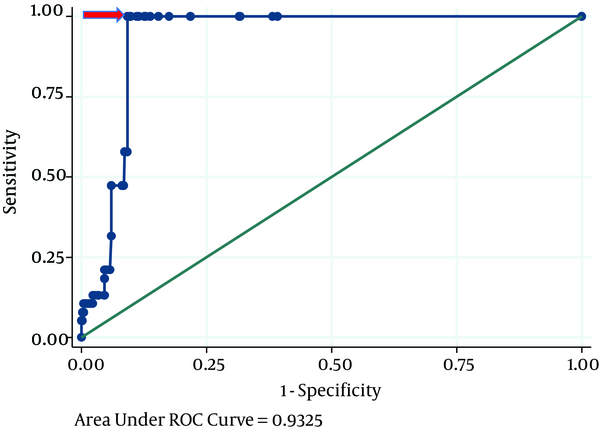
By excluding the data of SCH patients and maximizing sensitivity + specificity-1 across various cut-off points of the Billewicz index, an optimal cut-off point of -26.5 was obtained for distinguishing between normal individuals and patients with OH. The AUC of the Billewicz index for predicting the absence of risk for OH was approximately 0.93 (95% CI, 0.92 - 0.95; P < 0.001). The Billewicz score ≤ -26.5 corresponded to a sensitivity of 100% and specificity of 90.82% and yielded a positive predictive value of 24.67% for distinguishing euthyroidism from OH (this cut-off point corresponded to a positive likelihood ratio of 10.89 and a negative likelihood ratio of 0) (Figure 1).
4. Discussion
The results of this study suggest that despite confusions in identifying thyroid disorders during pregnancy due to physiological changes, the Billewicz scoring index is a valid scale for diagnosis of gravid women with OH. The optimal cut-off point was -26.5, with sensitivity of 100%, specificity of 90.82%, and positive predictive value of 24.67%. Our findings, validating the accuracy of Billewicz index during pregnancy, are novel, and to the best of our knowledge, no study has been published in this field so far.
We noted that delayed ankle reflex, fatigue, weakness, sleepiness, laziness, pulse rate < 75/min, constipation, and cold intolerance were the most common signs and symptoms of OH. In accordance with our findings, in a study by Galia et al. the most frequent symptoms in hypothyroid non-pregnant patients were easy fatigue (64%), dyspnea on effort (52%), weight gain (44%), and constipation (44%), while the most common signs were rough and dry skin (36%), thyroid enlargement (32%), and sluggish movement (32%) (19). Almost similarly, Zulewski et al. reported weight gain (54%), constipation (48%), dry skin (76%), and bradycardia (58%) as the most common manifestations of hypothyroidism, which are comparable to the current study (15).
We also noticed that all signs and symptoms, except for three (diminished sweating, hoarseness, and hearing impairment), were present in both euthyroid and hypothyroid women. However, only four items, including laziness/sleepiness, constipation, ankle jerk reflex, and pulse rate < 75/m, were significantly higher in hypothyroid women, compared to their euthyroid counterparts; therefore, there is a lower risk of overlap with pregnancy-related physiological changes. Since physiological changes of pregnancy may mimic thyroid dysfunction signs, accurate diagnosis of hypothyroidism, based on the common signs and symptoms used in non-pregnant populations, is problematic (20). Meanwhile, laboratory measurements of thyroid function need to be interpreted cautiously during pregnancy, compared to non-pregnant women (17).
Screening of thyroid disorders during pregnancy has been a long-disputed issue. Universal screening and targeted high-risk case-finding have their own advantages and disadvantages (17). Overall, universal thyroid screening is a common and cost-effective approach; however, there are still controversies about its application, since the prevalence of thyroid dysfunctions varies in different racial and ethnic populations (21); therefore, the significance of universal screening may be diminished among low-risk groups. In addition, there are still areas of uncertainty and disagreement among experts about the beneficial impact of maternal SCH diagnosis and management of poor pregnancy outcomes (22, 23).
A recent guideline of the American thyroid association declared that there is insufficient information to recommend or argue against universal screening during pregnancy, and TSH tests are recommended for women with the risk factors (17). On the other hand, the selective case-finding approach fails to identify about one-third of women with OH or SCH (24), which is considered a significant disadvantage for a screening test. Furthermore, challenging limitations, such as high cost of laboratory tests and endocrinologist visits, maternal anxiety, and absence of trimester-specific reference ranges, may necessitate validation of a preexisting reliable scale.
The Billewicz scale, appraised as one of the most cited thyroid scoring indices worldwide (25), has the potential to fill the mentioned gaps. This index serves as an inexpensive and accurate tool, basically for high-risk populations from low- and middle-income countries with limited access to costly laboratory tests. We agree that despite the strong association between clinical symptoms and abnormal TSH level (26), the accuracy of clinical diagnosis is limited, and laboratory diagnosis is also needed. Yet, we emphasize that the Billewicz index is not intended to replace sensitive biochemical measurements, but is a useful tool in detecting high-risk women prior to serum testing and can be applicable for measuring the patient’s response to medical treatment.
While the consequences of OH for pregnancy outcomes and neonatal/child development are well-established (5, 6), findings regarding the relationship between maternal SCH and adverse pregnancy outcomes in both mothers and neonates are contradictory. Current evidence does not support the lower level of intellectual development in children born to mothers with SCH (27), and data on the beneficial effects of thyroxine supplementation on SCH women are inconsistent (28). Therefore, identifying OH during pregnancy screening is a principal issue. Undoubtedly, undetected OH is associated with increased maternal and neonatal complications (5, 6, 29). However, even in the event of severe maternal hypothyroidism, neurocognitive deficiencies in the child may not occur once the condition is managed immediately in the first half of pregnancy; therefore, use of the Billewicz scale can decrease undiagnosed OH during pregnancy screening.
This study has one limitation. Instead of using the newly recommended limit of 4.0 μIU/mL, we defined SCH based on the TSH cut-off point of 2.5 μIU/mL according to the available guideline during study design and conception (September 2013 to February 2016). Considering the difference between the previous (12) and new (17) guidelines, a number of our euthyroid subjects might be classified as SCH patients. Euthyroid women had a significantly lower BMI, compared to the OH and SCH groups. However, this difference is not significant enough to influence the cut-off points; accordingly, there is no need to present age-BMI specific cut-off points.
4.1. Conclusions
In summary, this is a novel study, validating the Billewicz scale and identifying the cut-off points during pregnancy. We found that the Billewicz index could be simply used as an auxiliary diagnostic tool in resource-constrained settings. However, future studies should include pregnant women from different ethnic or geographic (in terms of iodine sufficiency) populations.