1. Background
Hepatitis is defined as the inflammation of the liver. Many factors (such as drugs) can cause hepatitis, but globally, the most common cause of this inflammation is “viruses”. If hepatitis lasts for less than 6 months, it is referred to as “acute hepatitis”, and if it lasts longer, it is called “chronic hepatitis” (1). Acute viral hepatitis usually begins with discomfort, fatigue, nausea, vomiting, muscle aches, headaches, and mild fevers. Sometimes, the disease initially presents itself with signs similar to that of the cold. Smokers with acute hepatitis often begin to hate smoking. After that, an “Icter” or “Jaundice” appears.
Usually, as jaundice begins, the general condition of the patient improves, and even their fever might wane; these do not indicate an improvement of the symptoms of the disease, but the opposite. In fact, this is just the beginning. Measuring liver enzymes is a method which physicians use to diagnose patients with hepatitis, Jaundice, or any other disease with similar hallmarks. In hepatitis, the liver cells are destroyed, which causes a large number of liver enzymes, including Alanine Transaminase (ALT) and Aspartate Transaminase (AST) to enter the blood. The elevation of these enzymes in the patients’ serum represents hepatitis (2).
Acute viral hepatitis is a major cause of death and disability in the world. Acute classical viral hepatitis includes hepatitis A, B, C, D and E. Hepatitis B, and C and D can lead to chronic infection, liver cirrhosis, liver cancer, liver failure, and death (1). Acute viral hepatitis (AVH) caused by alimentary transmitted hepatitis A virus (HAV) and hepatitis E virus (HEV) poses a major health problem in developing countries. Both viruses are transmitted primarily by the fecal-oral route and cause acute viral hepatitis. Children with HAV tend to present with nonspecific gastrointestinal symptoms, and jaundice with cholestasis is common (3, 4). Acute viral hepatitis in children is one of the most important diseases and a major public health problem in both developing and developed countries (5-12). Despite the availability of vaccines, prophylactic measures, and improved sanitation, it is affecting millions of children every year (8).
To determine the prevalence of sonographic signs in patients with viral and acute hepatitis, ultrasound and computerized tomography scan (CT scan) studies have been investigated by some researchers (2, 13-15). Also, some other researchers have studied the efficacy of conventional ultrasound and Doppler in the diagnosis of acute hepatitis (16). The results of these studies have indicated that there is no general agreement on the prevalence and importance of sonographic signs in acute hepatitis.
In a study by Salehi and Sharifi in Zahedan city, southeast of Iran, acute hepatitis A with 71.5% and acute hepatitis B with 17.9% were the most common types of acute hepatitis (17). Ultrasound symptoms reported in children with acute hepatitis include: Hepatomegaly, increased liver echogenicity, increased echogenicity of portal vein walls, and increased the gallbladder wall thickness. These symptoms are not specific to acute hepatitis by themselves, but their presence suggests acute hepatitis (13-15). The ultrasound symptoms of acute hepatitis in pediatrics include hepatomegaly, periportal lymphadenopathy, splenomegaly, increased thickness of the gallbladder wall, decreased or increased echogenicity of the liver and increased echogenicity of the portal walls (2), but there is no consensus on the prevalence or the importance of the mentioned findings among researches (18). Although hepatitis is a long-known disease and its diagnosis usually is made clinically and Paraclinically, but there are many children with vague symptoms wondering around without diagnosis (19). There are few studies in which the sonographic features for the patients with acute hepatitis are investigated.
2. Objectives
In this paper, we now address this unique aspect for evaluation of sonographic symptoms in children with acute hepatitis all at once and we have presented the results showing that the most common sonographic signs of patients with hepatitis were increased liver echogenicity and periportal hyperechogenicity.
3. Methods
3.1. Sample Collection
At first, patients who were between 6 months and 15 years of age referred to the pediatric clinic and were visited by a pediatrician. Those who were suspected of acute hepatitis were sent to the laboratory for testing. After evaluation of ALT, AST, bilirubin, and anti-hepatitis Immunoglobulin G (IgG) antibodies, a group of patients diagnosed for hepatitis were sent for sonography. Each patient underwent ultrasound with a General Electric (GE) ultrasonographic instrument equipped with a curved probe at a frequency of 3.5 to 5 MHz, and the observed symptoms in his/her ultrasound were determined and recorded. At the end of the research, the prevalence of ultrasonographic signs was calculated in the case study group.
3.2. Sample Size
Sample size and sampling method are obtained from the following equation:
According to Toppert study (13): P = 0.5, d = 0.05, and z = 1.96, based on census method for each case of acute hepatitis who referred to ultrasound clinic.
3.3. Data Analysis Method
After collecting the data, all data were analyzed using the Statistical Package for the Social Sciences (SPSS) version 20.0 (Chicago, II, USA). To describe the quantitative data, the central and dispersion indexes (mean and standard deviation), and for the qualitative data description, the distribution tables (absolute prevalence and relative prevalence) were used.
3.4. Moral Considerations
In this research, all ethical considerations have been reviewed and approved by the Ethics Committee of the Zahedan University of Medical Sciences (IR.ZAUMS.REC.1393.6714).
4. Results
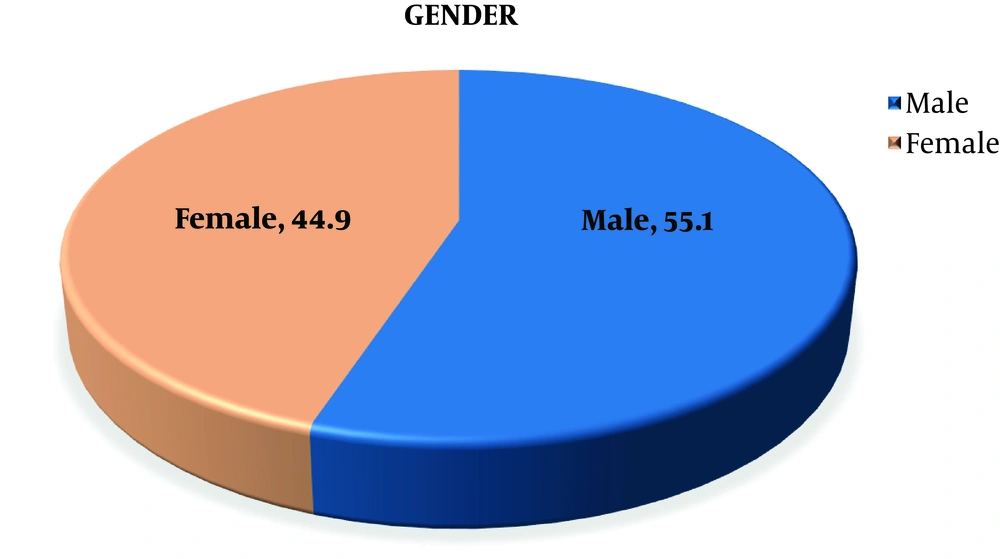
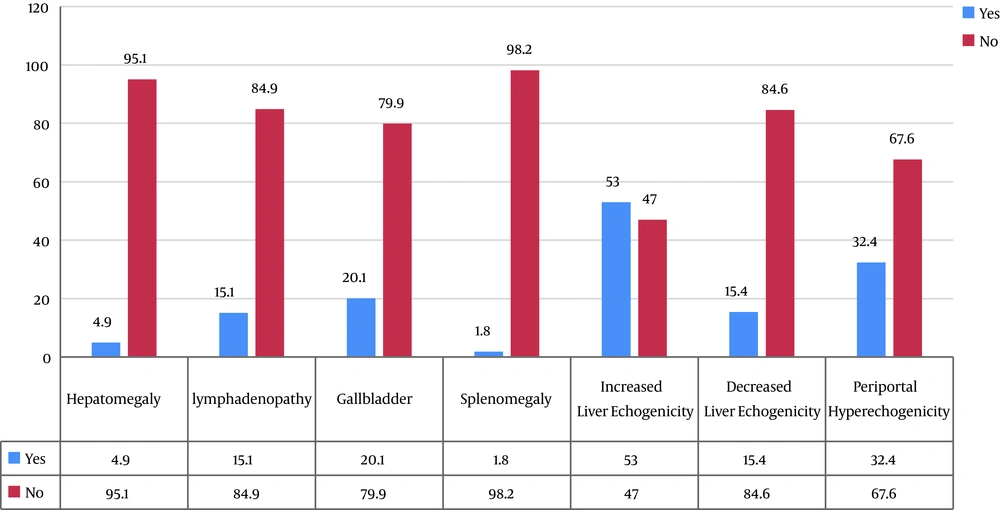
In this study, 383 children with acute hepatitis referring to the Ultrasound Clinic of Imam Ali Hospital in Zahedan city were studied. Given the gender distribution, 211 (55.1%) were male, and 172 (44.9%) were female and the mean age of children was 6.1 ± 2.8 years (Figure 1). The prevalence of sonographic signs among all 383 children with acute hepatitis was 77 children (20.1%) with gallbladder wall edema, and 306 children (79.9%) lacked the sign. Also, 58 children (15.1%) showed periportal lymphadenopathy, while 325 children (84.9%) did not show this sign. In addition, hepatomegaly was seen in 19 children (4.9%), whereas 364 cases (95.1%) lacked the sign of it. Additionally, 203 children (53.0%) were classified among those showing liver hyperechogenicity, and 180 children (47.0%) lacked it. Moreover, hypoechogenicity was one of the signs that 59 patients (15.4%) showed, in turn, 326 patients (84.6%) lacked the liver hypoechogenicity. Seven children (1.8%) also showed splenomegaly, and 376 children (98.2%) lacked it. Finally, 124 children (32.4%) had hyperechogenicity and 259 children (67.6%) lacked it (Table 1 and Figure 2).
| Sonographic Sign | Frequency, No. (%) | |
|---|---|---|
| Yes | No | |
| Hepatomegaly | 4.9 | 95.1 |
| lymphadenopathy | 15.1 | 84.9 |
| Gallbladder | 20.1 | 79.9 |
| Splenomegaly | 1.8 | 98.2 |
| Increased liver echogenicity | 53.0 | 47.0 |
| Decreased liver echogenicity | 15.4 | 84.6 |
| Periportal hyperechogenicity | 32.4 | 67.6 |
5. Discussion
The results of this study showed that the prevalence of sonographic signs in children with acute hepatitis was as follows: Gallbladder wall edema 20.1% (77 children), lymphadenopathy 15.1% (58 children), hepatomegaly 4.9% (19 children), liver hyperechogenicity 53.0% (203 children), liver hypoechogenicity 15.4% (59 children), splenomegaly 1.8% (7 children), and periportal hyperechogenicity 32.4% (124 children); (Figure 3).
In a study conducted by Braden et al. (15), it was concluded that periportal lymphadenopathy is common in infectious diseases and acute autoimmune diseases, and the absence of periportal lymph nodes in acute liver injury has led to a search for toxic and metabolic causes.
Garassini et al. (14) also studied the ultrasonographic signs of acute hepatitis and reported the signs of lymphadenopathy, splenomegaly, and gallbladder wall thickening. Although in our study, these symptoms were also observed in acute hepatitis, these symptoms were different due to the low percentage of splenomegaly (1.8%), increase thickness of the gallbladder wall (20.1%) and lymphadenopathy (15.1%) in comparison with liver hyperechogenicity (53.0%) and periportal echogenicity (32.4%). Various studies have been carried out on this topic, i. e. in the Toppet et al. study on 58 children with acute hepatitis (13), the lymphadenopathy for all patients was much more common than what we observed in our study. These lymph nodes were mainly located at periportal vein and in the front of the pancreas head. The other sonographic signs were observed with the following frequencies: an increase in the liver echogenicity was observed in 53% of the patients, which is consistent with our study. Moreover, an increased thickness of the gallbladder wall between 3 and 6 mm was observed in 52% of patients, much higher than our study, which was about 20%. Hepatomegaly was observed in 43% of patients that was much more than our study with only 5% hepatomegaly. Also, periportal hyperechogenicity was seen in 24% of the patients that it is lower than our study with 32% (13).
In the study of Kumar et al. (20), some children with acute hepatitis were examined, which showed gallbladder wall thickening in 41%, pleural effusion in 14.1%, and ascites in 44.9% of patients. Furthermore, the mortality rate was reported to be 1.3 and the rate of gallbladder wall thickening in their study was more than our study.
Kc et al. (21) from Nepal reported that of 368 patients less than the age of 15, attending the clinic with complaints related or similar to acute viral hepatitis, 312 patients were diagnosed with acute viral hepatitis. The patients were divided into 3 groups based on their age; 0 - 5, 5 - 10, and 5 - 15 years. The study results were as follows: 266 (85%) were infected by hepatitis A virus, 24 (8%) by hepatitis E virus, 15 (5%) by hepatitis B virus, and the etiology was unknown in the remaining 7 (2%) patients. Ascites was more common in the 5-10-year age group. The ultrasound findings were as follows: 244 (78%) hepatomegaly, 125 (40%) splenomegaly, 266 (85%) collapsed (partial or complete) gallbladder, 284 (91%) gallbladder wall thickening, 203 (65%) pericholecystic edema, 150 (48%) gallbladder sludge, 162 (52%) prominent hilar node, 47 (15%) ascites, 79 (25%) prominent wall of IHBD, 32 (10%) normal H-B scan. Collapsed gall bladder with pericholecystic edema was the most common finding. Ascites was present in 21% of the cases. These ultrasound findings were similar to the sonographic signs of our study.
Mahmud et al. (12) from Bangladesh also observed that, out of 100 acute hepatitis children, 9 (9.0%) were in 1-5-year age group, 52 (52.0%) in 5-10-year age group and 39 (39.0%) in 10 - 15 years age group. In ultrasound findings, hepatomegaly (93.0%) was found in most of the cases followed by splenomegaly (23.0%), gallbladder wall thickening (33.0%), gallbladder sludge (23.0%), hepatosplenomegaly (23.0%) and ascites (16.0%). Normal ultrasound was found in 3.0% of cases. These findings are similar to our ultrasound findings, but with different prevalence.
In Zivkovic and Trajer’s research (22), ultrasound findings were evaluated in 90 patients with acute viral hepatitis. An increase in the thickness of the gallbladder wall was observed in the first week of the disease, which was 4.67 mm. In the current study, an increase in the thickness of the gallbladder wall also observed in 20.1% of the patients.
In another study by Shin et al. (16), the efficacy of conventional ultrasound and Doppler in the diagnosis of acute hepatitis was studied, and it has been shown that ultrasound is consistent with the laboratory criteria for acute hepatitis and can help to diagnose it.
In a different study by Yoo et al. (23), CT scan findings in acute hepatitis were evaluated, and variations in the symptoms observed in patients have been reported as follows: gallbladder changes in 75%, priportal edema in 43%, hepatomegaly in 23%, and splenomegaly in 52% of patients. These findings are more frequent than the ultrasound findings, which is due to the quality of the instruments used.
Thus, the ultrasonography findings have a significant role in the early diagnosis of acute viral hepatitis and can help us to detect the changes in the hepatobiliary system on ultrasound in most of the acute viral hepatitis. Some very common findings related to the gallbladder include an increase in thickness of gallbladder wall and pericholecystic edema. Detection of these changes can be used as further evidence of acute viral hepatitis. This is most useful if serological diagnostic facilities are not available as they reinforce the diagnosis of suspected acute hepatitis. The detection of the mentioned changes within the gallbladder has been further verified by a more accurate testing technique called endoscopic ultrasonography (2, 24).
One of the most important subjects in previous studies is that all of the variables in acute hepatitis in children (including increased liver echogenicity, decreased liver echogenicity, hepatomegaly, splenomegaly, periportal hyperechogenicity, periportal lymphadenopathy and increased thickness of the gallbladder wall has not been studied altogether. This subject can be an important cause of some differences between the results of various studies. Our research advantage over the other researches is that we have considered all the parameters studied in previous studies thoroughly and simultaneously, and then we compared the results of our study with the results of previous studies.
The limitation of our study is that we were not able to determine any specific time for the sonographic symptoms. For instance, during different sonographic exams, patients were only diagnosed under acute, subacute, and chronic conditions with no other apparent distinctions.
5.1. Conclusions
As a general conclusion, our ultrasonography findings are present in most of the acute viral hepatitis. Our results show that the sonographic signs that are seen in children are nearly similar to most other studies, but the incidence rate and prevalence of each sign are different. It may be due to the type of sonographic instrument, the proficiency of the sonographer, and even the location of the study (for example, nutritional habits or patients’ level of health). Therefore, because of the dependence of ultrasound findings on the type of instrument used and the sonographer, these differences are reasonable. Overall, our results have shown that the most common sonographic signs among patients are liver hyperechogenicity and periportal hyperechogenicity.