1. Background
In early December 2019, the first case of severe acute respiratory syndrome-coronavirus 2 (SARS-CoV-2) was discovered in Wuhan, China. Soon, the coronavirus disease 2019 (COVID-19) pandemic was declared by the World Health Organization (WHO) throughout the world. Until October 2021, the WHO reported about 233 million confirmed cases of COVID-19 and 4.7 million deaths, with a mortality rate of 2.1% (1). Mainly, SARS-CoV-2 is transmitted by inhalation of droplets. The average incubation period of this disease is two weeks (2, 3).
The pulmonary involvement of COVID-19 patients ranges from a normal pattern to complete pulmonary involvement (4). Nonetheless, the most common CT scan finding in COVID-19 patients is bilateral subpleural ground-glass opacity (GGO) (5). The clinical pattern of SARS-CoV-2 infection may range from asymptomatic infection to acute respiratory distress syndrome (ARDS) and multi-organ dysfunction (6, 7). The therapeutic options for this disease include antivirals, immunomodulators, and corticosteroids; however, no specific drug has been yet approved for COVID-19 treatment (8).
There are several studies reporting the role of vitamin D in regulating the immune system and reducing the respiratory tract infection severity (9-12). Generally, the primary source of vitamin D for humans is sunlight. The ultraviolet-B (290 - 315 nm) portion of solar radiation converts 7-dehydrocholesterol to pre-vitamin D3 in the skin, which is then transformed to vitamin D3 (13). Vitamin D receptors are found not only in the classic target tissues of vitamin D, such as the bone, gut, and kidney tissues, but also in other organs, including the lungs (14).
1,25-Dihydroxyvitamin D (1,25(OH)2D) is the active metabolite of vitamin D, affecting both adaptive and innate immune systems (15). As an active hormone, it participates in the regulation of genes, which encode some proteins required for the first wall barrier (16). In innate immunity, a sufficient serum circulation of 1,25(OH)2D can produce cathelicidins and defensins with antimicrobial activities (17). Vitamin D also has direct antiviral effects, especially against enveloped viruses. Besides, cell culture studies suggest the antiviral role of vitamin D against enveloped viruses by upregulating antimicrobial peptides, including LL-37 (18).
The coronavirus (CoV) envelope protein is a small, integral membrane protein that participates in several parts of the virus life cycle, such as production, maturation, and pathogenesis. It has been shown that recombinant CoVs without envelope-producing genes can significantly reduce the viral titers (19). According to reports, vitamin D status varies around the world. In a narrative review by Lips in 2007, vitamin D deficiency was common in the Middle East, Southern Europe, India, China, and Japan, while it was less common in Northern Europe and Southeast Asia (20). In 2016, Cashman et al. found that 13.1% of Europeans (55,844) had serum 25(OH)D levels below 30 nmol/L (21).
From 1988 to 2006, the prevalence of serum 25-Hydroxy vitamin D3 (25(OH)D) below 30 nmol/L increased from 5% to 10% in North America. The highest prevalence of serum 25(OH)D level < 50 nmol/L was reported in Argentina and Chile (22). Based on the WHO reports, Argentina and Chile are among the first 20 countries with the highest number of confirmed COVID-19 cases.
Although there is not enough evidence regarding the relationship between COVID-19 severity and vitamin D deficiency (23-25), some researchers have reported a relationship (26-28). In this regard, Hastie et al. examined the vitamin D status and COVID-19 in the UK Biobank. However, they could not provide any evidence to support the association of 25(OH)D status with COVID-19 susceptibility (29). In another study by Baktash et al. the outcomes of COVID-19 were studied in older adults with vitamin D deficiency, and the results confirmed the correlation between COVID-19 severity and vitamin D deficiency (30). Overall, there are some contradictory findings on whether COVID-19 susceptibility and severity depend on vitamin D status. In independent studies by Moradzadeh et al. and Sadinia et al. the prevalence of vitamin D deficiency was high in most parts of Iran and the Middle East (31, 32).
2. Objectives
Considering the high prevalence of vitamin D deficiency in Iran and the existing evidence of its possible association with the current critical status of COVID-19, this study aimed to examine the level of vitamin D and determine its association with the severity of infection.
3. Methods
This retrospective, cross-sectional study was performed on 122 patients with symptomatic COVID-19 and 49 COVID-19 positive patients without an underlying disease, hospitalized in Amir-al-Momenin Hospital, affiliated to Islamic Azad University of Tehran, Iran, from April 2020 to May 2020, as well as 122 non-COVID-19 individuals as a control group, who were examined for vitamin D status. The patients were positive for COVID-19, based on reverse transcriptase-polymerase chain reaction (RT-PCR) of nasopharyngeal and pharyngeal swab specimens. The CT scans, laboratory tests, and clinical examinations were performed for each patient.
The vitamin D status was measured based on the serum 25-Hydroxy vitamin D3 (25(OH)D) level in each patient by electrochemiluminescence (ECL) method. Vitamin D deficiency was defined as a 25(OH)D level below 30 ng/mL in this study. The vitamin D level was measured on the first day of receiving inpatient services and before medication use. None of the patients used vitamin D supplements. The CT scans of all patients were acquired in the supine position without an intravenous contrast; all images were acquired based on standard scanning protocols. All CT scans were reviewed by the hospital radiologists. The pulmonary involvement was classified as follows: 0 (0% involvement), 1 (1 - 24% involvement), 2 (25 - 49% involvement), 3 (50 - 74% involvement), and 4 (75 - 100% involvement). This study was approved by the ethics committee of Islamic Azad University of Medical Sciences (IR.IAU.PS.REC.1399.036).
The severity of COVID-19 was categorized into four groups: Mild, moderate, severe, and critical. Mild infection was defined as a respiratory rate of 24 beats per minute (bpm) or less, O2 saturation of 93% or higher on room air, and a normal chest CT scan (or < 25% pulmonary involvement). Moderate infection was defined as O2 saturation of 90 - 93% on room air, a respiratory rate of 24 - 30 bpm, and chest CT scan involvement of 25 - 50%. Severe infection was defined as O2 saturation of 90% or less on room air, a respiratory rate of 30 bpm or higher, and pulmonary involvement > 50%. Finally, critical cases were under ventilation or experienced septic shock, along with COVID-19 complication.
The demographic information, clinical characteristics, CT findings, and laboratory results of all patients were gathered in questionnaires from the patients’ records. First, the vitamin D levels of all patients were compared with the controls, and then, the association of vitamin D level with the severity of disease was examined. Patients with underlying conditions, such as autoimmune disease, cancer, hypertension, diabetes, pulmonary disease, renal diseases, and cardiovascular disease, were excluded. A total of 49 patients remained in the study. The vitamin D status of 49 patients was examined in relation to COVID-19 severity.
The collected data were entered in IBM® SPSS® Version 26.0. The frequencies and mean values of all variables were calculated and compared to obtain reliable results. SPSS was used to calculate the probability value (P-value). For comparing the means of each O2 saturation, respiratory rate, and lung involvement percentage with vitamin D status, Kolomogrov-Smirnov test and independent t-test were used, and Spearman correlation of the amount of each variable and vitamin D status was also calculated. For comparing the severity of the disease with vitamin D status chi-square test was used and for comparing vitamin D status in the two groups, Mann-Whittney test was used. The Mann-Whittney test was also used for analyzing each O2 saturation, respiratory rate, and lung involvement percentage with vitamin D status of 49 patients without underlying diseases. P-value less than 0.05 was considered statistically significant.
4. Results
The mean ± SD age of 122 COVID-19 patients was 59 ± 16 years, and 71 (58.2%) patients were male. Overall, 77 (63%) patients were positive for COVID-19, based on RT-PCR using nasopharyngeal and pharyngeal swab specimens upon arrival, and the rest of the patients were positive in the second and third examinations. Also, the mean age of the controls, including 61 males, was 48 ± 13 years; the mean age of the patients was higher than that of the controls (P < 0.05). Based on the findings, the mean inpatient duration was 7 ± 4 days, and the mean duration of medication use was 9 ± 3 days. The mean vital signs were as follows: Pulse rate, 89.7 ± 15.3 bpm; O2 saturation percentage, 89.4 ± 8.9%; systolic blood pressure, 118.6 ± 16.65 mmHg; diastolic blood pressure, 73.2 ± 12.9 mmHg; respiratory rate, 19.4 ± 8.3 bpm, and temperature, 37.4 ± 0.8°C upon arrival.
The most common symptoms were dyspnea (78.7%), cough (77%), body temperature > 38°C (69.7%), malaise (65%), loss of appetite (61.5%), myalgia (62.3%), and chest pain (50.8%). The analysis of pulmonary involvement indicated the following results: Normal lungs in 2 (1.6%) cases; < 25% involvement in 22 (18.0%) cases; 25 - 49% involvement in 70 (57.4%) cases; 50 - 74% involvement in 25 (20.5%) cases; and ≥ 75% lung involvement in 3 (2.5%) cases.
The mean vitamin D level was 34.14 ± 1 ng/mL (95% CI: 30.43 - 37.85) in the patients and 32.94 ± 1 ng/mL (95% CI: 30.13 to 35.74) in the controls. The patients had higher vitamin D levels compared to the controls; however, the difference was not significant (P = 0.872). The vitamin D level of 60 (49.2%) patients was deficient, and half of the controls (50%) were deficient in vitamin D. Also, 58 (47.5%) patients were mildly infected, 41 (33.6%) patients were moderately infected, and 23 (18.9%) patients were severely infected. In 49 patients without an underlying disease, the mean vitamin D level was 32.85 ± 19 ng/mL (95% CI: 27.29 - 38.41). Overall, 27 (55.1%) patients were mildly infected, 16 (32.7%) patients were moderately infected, and 6 (12.2%) patients were severely infected.
Comparison of vitamin D level and O2 saturation showed that patients with O2 saturation > 93% were mostly vitamin D sufficient (55.2%); these cases were considered mild. Also, 50% of patients with O2 saturation < 90% were vitamin D deficient, with a two-tailed P-value of 0.324; therefore, the correlation was not significant. Moreover, comparison of vitamin D status and respiratory rate indicated that 50% of patients with a respiratory rate > 30 bpm had vitamin D deficiency; this correlation was not significant, with a two-tailed P-value of 0.162. Moreover, comparison of vitamin D status and percentage of pulmonary involvement revealed that patients with 25% pulmonary involvement or more had vitamin D levels below 30 ng/mL, however, the result was not significant (two-tailed P-value = 0.074). Overall, the results showed that the severity of disease was not related to the vitamin D status of the patients; in other words, a higher severity of COVID-19 was not associated with a lower vitamin D level; the P-value was 0.125, and the correlation was not significant (Table 1).
| Status | Vitamin D Level | P Value | Spearman’s Rho | ||
|---|---|---|---|---|---|
| 25(OH)D < 30 (% Within Vit D) | 30 ≤ 25(OH)D (% Within Vit D) | Total (% Within Total) | |||
| O2 saturation (%) | 0.324 | 0.051 | |||
| < 90 | 21 (50) | 21 (50) | 42 (34.4) | ||
| 90 - 93 | 17 (54.8) | 14 (45.2) | 31 (25.4) | ||
| 93 < | 22 (44.8) | 27 (55.2) | 49 (40.2) | ||
| Total | 60 | 62 | 122 | ||
| Respiratory rate (times/min) | 0.162 | -0.140 | |||
| < 24 | 52 (48.1) | 56 (51.9) | 108 (89.3) | ||
| 24 - 30 | 5 (55.6) | 4 (44.4) | 9 (7.4) | ||
| 30 < | 2 (50) | 2 (50) | 4 (3.3) | ||
| Total | 59 | 62 | 121 | ||
| Lung involvement percentage (%) | 0.074 | -0.181 | |||
| 0 | 1 (50) | 1 (50) | 2 (1.6) | ||
| 1 - 24 | 7 (31.8) | 15 (68.2) | 22 (18.0) | ||
| 25 - 49 | 35 (50) | 35 (50) | 70 (57.4) | ||
| 50 - 74 | 15 (60) | 10 (40) | 25 (20.5) | ||
| 75 - 100 | 2 (66.6) | 1 (43.4) | 3 (2.5) | ||
| Total | 60 | 62 | 122 | ||
| Severity | 0.125 | - | |||
| Mild | 23 (39.7) | 35 (60.3) | 58 (47.5) | ||
| Moderate | 23 (56) | 18 (44) | 41 (33.6) | ||
| Severe | 14 (60.8) | 9 (39.2) | 23 (18.9) | ||
| Total | 60 | 62 | 122 | ||
Abbreviation: Vit D, vitamin D.
a Vitamin D status is in ng/mL.
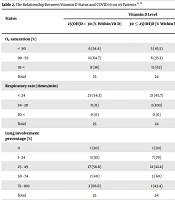
Moreover, 49 patients without an underlying disease were examined in this study. The results indicated that patients with lower percentages of O2 saturation were mostly vitamin D deficient; however, the distribution was nonparametric, and the P-value (0.127) was not significant. Analysis of respiratory rate and pulmonary involvement percentage in relation to the vitamin D status showed no significant relationship (P > 0.05). Finally, analysis of the relationship between the severity of disease and vitamin D status showed no significant correlation in patients without an underlying disease (P > 0.05) (Table 2).
| Status | Vitamin D Level | P Value | Spearman’s Rho | ||
|---|---|---|---|---|---|
| 25(OH)D < 30 (% Within Vit D) | 30 ≤ 25(OH)D (% Within Vit D) | Total (% Within Total) | |||
| O2 saturation (%) | 0.127 | 0.183 | |||
| < 90 | 6 (54.4) | 5 (45.5) | 11 (22.5) | ||
| 90 - 93 | 11 (64.7) | 6 (35.3) | 17 (34.7) | ||
| 93 < | 8 (38) | 13 (62) | 21 (42.8) | ||
| Total | 25 | 24 | 49 | ||
| Respiratory rate (times/min) | 0.408 | -0.114 | |||
| < 24 | 25 (54.3) | 21 (45.7) | 46 (0) | ||
| 24 - 30 | 0 (0) | 3 (100) | 3 (7.4) | ||
| 30 < | 0 (0) | 0 (0) | 0 (0) | ||
| Total | 25 | 24 | 49 | ||
| Lung involvement percentage (%) | 0.714 | -0.109 | |||
| 0 | 1 (50) | 1 (50) | 2 (4) | ||
| 1 - 24 | 3 (30) | 7 (70) | 10 (20.4) | ||
| 25 - 49 | 17 (58.6) | 12 (41.4) | 29 (59.2) | ||
| 50 - 74 | 2 (40) | 3 (60) | 5 (10.2) | ||
| 75 - 100 | 2 (66.6) | 1 (43.4) | 3 (6.2) | ||
| Total | 25 | 24 | 49 | ||
| Severity | 0.261 | - | |||
| Mild | 11 (40.7) | 16 (59.3) | 27 (55.2) | ||
| Moderate | 11 (68.7) | 5 (31.3) | 16 (32.6) | ||
| Severe | 3 (50) | 3 (50) | 6 (12.2) | ||
| Total | 25 | 24 | 49 | ||
Abbreviation: Vit D, vitamin D.
a Values are expressed as No. (%).
b Vitamin D status is in ng/mL.
5. Discussion
In the present study, the vitamin D status was not significantly different between 122 patients with COVID-19 and the controls (P > 0.05). The correlation between vitamin D level and disease severity was also not significant in 122 patients (P > 0.05), and in 49 patients without an underlying disease, the correlation was not significant (P > 0.05). The insignificant correlation in this study suggests that in small scale of individuals vitamin D deficiency was not related to neither COVID-19, nor its severity. However, in larger community the results could be different. Nonetheless, the Spearman’s Rho correlation demonstrated that respiratory rate and lung involvement percentage correlation with vitamin D status was more meaningful in 122 patients than those without underlying disease, this could show that some underlying diseases would affect the serum vitamin D level, which might increase the respiratory rate and lung involvement. In meta-analyses, several researchers have examined the antiviral role of vitamin D sufficiency in the human body and its alleviating role in acute respiratory tract infections. The serum vitamin D level can predict the severity of the recent SARS-CoV-2 infection (18, 33, 34). Vitamin D deficiency may also suppress dipeptidyl peptidase-4 (DDP4/CD26) as one of the adhesion molecules for the Middle East respiratory syndrome (MERS)-CoV and COVID-19 in the human body cells (35, 36).
In the present study, the patients’ vitamin D status and O2 saturation were compared, similar to a study by Franco et al. and another study by Choudhary and Gupta on children under five years. Some of the patients with adequate vitamin D levels had less severe infections. However, in the current study, there was no significant correlation between vitamin D status and O2 saturation, similar to the study by Choudhary and Gupta, despite the age differences of participants in these two studies. The present results did not indicate a higher respiratory rate associated with vitamin D deficiency. In the study by Franco et al. the risk and severity of ARDS and pneumonia were higher in vitamin D-deficient patients; it should be noted that Franco et al. studied patients with chronic obstructive pulmonary disease (COPD) (37, 38).
In this study, the association of vitamin D deficiency with pulmonary involvement was examined in COVID-19 patients. Based on the results, patients with 25% pulmonary involvement or more had vitamin D levels below 30 ng/mL compared to those with vitamin D levels of 30 ng/mL or higher. The current results showed a non-significant relationship between pulmonary involvement and vitamin D status, versus a study by Zosky et al. on vitamin D deficiency and its effects on the pulmonary function and changes in the lung structure in mice samples. Zosky et al. found that vitamin D deficiency decreases lung volume and function, however the whole study explains the association between obstructive lung disease and vitamin D status (39).
The results of the present study indicated the insignificant effects of vitamin D deficiency on the COVID-19 severity. We need to show that patients with moderate and severe diseases have lower vitamin D levels than mild cases. There are several studies showing that vitamin D deficiency can potentially increase the severity of SARS-CoV-2 infection. In a study by Brenner et al. in Germany, vitamin D deficiency and insufficiency contributed to a COVID-19 mortality rate of 41% (40). In another similar study by D'Avolio et al. in Switzerland, the vitamin D level of PCR-positive SARS-CoV-2 patients, aged 70 years or above, was deficient (median, 11.1 ng/mL); however, the age range of participants in their study was different from ours (41).
Although there are studies similar to the one by Panagiotou et al. rejecting a correlation between vitamin D status and mortality, our study focused on the severity of disease rather than mortality. Panagiotou et al. studied 134 hospitalized COVID-19 patients and showed that only 19% of the participants had sufficient vitamin D levels (42). Moreover, Hastie et al. examined the association between vitamin D level and COVID-19 after removing participants with potential cofounders similar to 49 patients of ours without underlying diseases, and found no significant correlation; however, the correlation between vitamin D status and COVID-19 severity was unclear in their study (29).
In conclusion, vitamin D deficiency (25(OH)D level < 30 ng/mL) was not associated with COVID-19 severity in all patients who participated in this study. As well as the patients without an underlying disease. On the other hand, the analysis of disease severity and vitamin D status of patients without an underlying disease demonstrated that vitamin D sufficiency was more common in patients with a mild disease compared to those with a moderate disease. However, among severely infected cases, the number of patients was not significantly different in relation to the vitamin D status (P > 0.05).
5.1. Conclusions
The present study did not find a significant relationship between the vitamin D status and the severity of COVID-19 in patients with/without an underlying disease. For investigating this correlation, it is important to exclude the underlying diseases. We know well that evidence is against our findings, therefore, for more accurate results, a larger sample size and further case-control studies are required. Besides, use of vitamin D supplementation for COVID-19 patients with vitamin D deficiency seems beneficial; therefore, we highly recommend this supplementation for all patients with vitamin D deficiency.