1. Background
Silver Nanoparticles (AgNPs) are between 1 nm and 100 nm in size (1). Until the nineteenth century silver was not considered as an antibacterial agent (2, 3). Since then, antimicrobial properties of silver have been studied and used more than any other inorganic antibacterial (4).
Micromolar concentrations of silver have no harmful effects on humans (5). Therefore, silver has been used for many biological processes and pharmaceutical development of dental materials, grafts (6) and coating materials for medical devices (7). Fungal strains of Fusarium oxysporum soil inhabitants have the ability to live as saprophytes and degrade lignin (8). Many disease-causing strains within complex F. oxysporum to plants, especially in agricultural settings. The use of AgNPs has become more common as technological advances have made it more affordable. It is possible for AgNPs to have serious consequences for the environment (9). One of potential applications is its use in plant disease management. Up to now, limited research evidence on the use of AgNPs for the control of fungal pathogens, has been published (10).
2. Objectives
The aim of this research was to evaluate the antifungal effect of AgNPs on F. oxysporum.
3. Methods
3.1. Fungal Isolate
Fusarium oxysporum, which is a plant pathogenic fungus, was used in this research. Single-spore isolates of F. oxysporum from infected melon plants were collected from the Sistan region.
3.2. Silver Preparation
The seeds of Satureja hortensis were collection from the southern region of Iran (Kerman) and silver preparation was conducted, as described by Mohassel and Pourseyedi (11).
3.3. Inhibition of Colony Formation by Different Concentrations of Silver Nanoparticles
Antifungal activity of AgNPs on basis of colony formation was evaluated by in vitro Petri dish measurement. Fusarium oxysporum was grown in CMA. Conidium cultures were incubated at 28°C for 10 days and 106 spores per mL-1 were diluted with distilled water. Same amount of spore suspension of AgNPs preparation series (1000, 2500 and 5000 ppm) were mixed to a final volume of 1 mL, and water was mixed with distilled water as a control. Ten-milliliter-sample of stock conidia and silver was mixed at 0, 1, 3 and 5 hours after treatment were silver and diluted 100 times with sterile water. Ten microliters of diluted sample in PDA released spores. Four PDA plates were tested at any combination of exposure time and concentration of AgNPs. The number of colonies that had formed on the plates were counted after four days at 28°C. The numbers of colonies of AgNPs treated with a spore suspension on controller were compared to calculate concentration of each AgNPs preparation.
4. Results
4.1. Inhibition of Colony Formation by Different Concentrations of Silver Nanoparticles
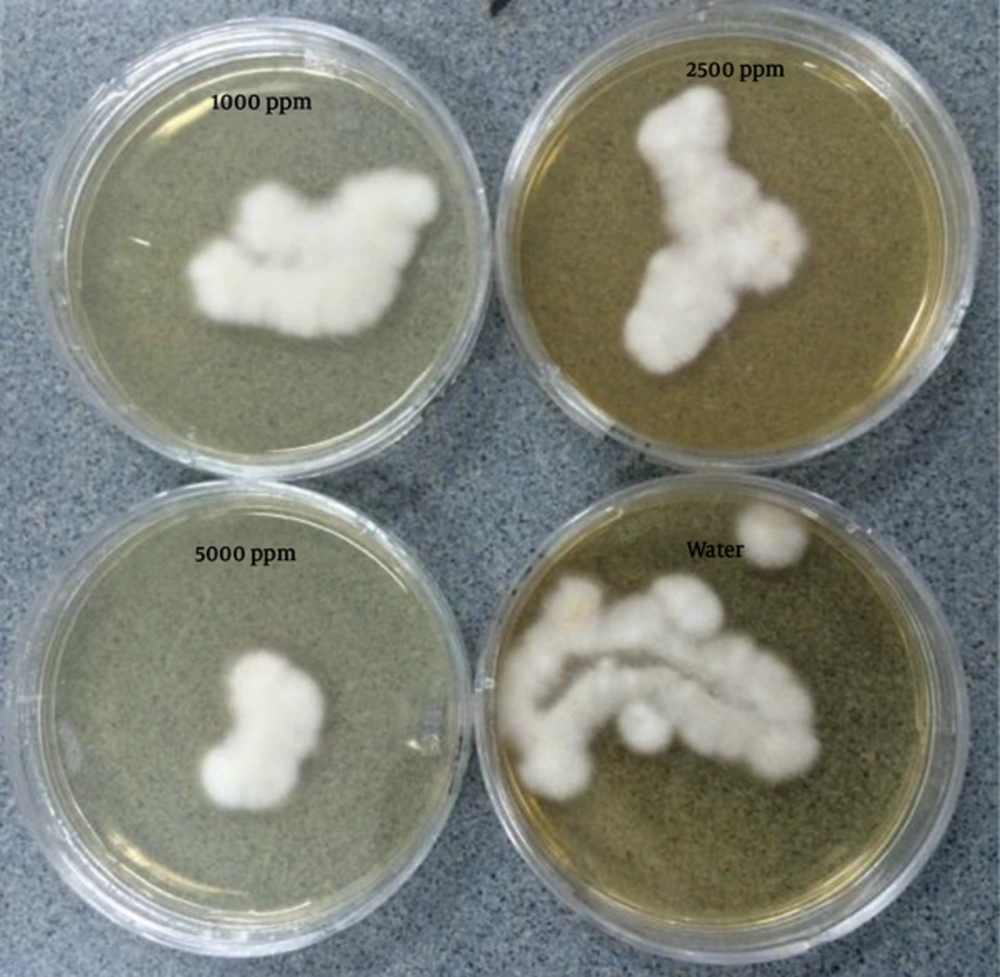
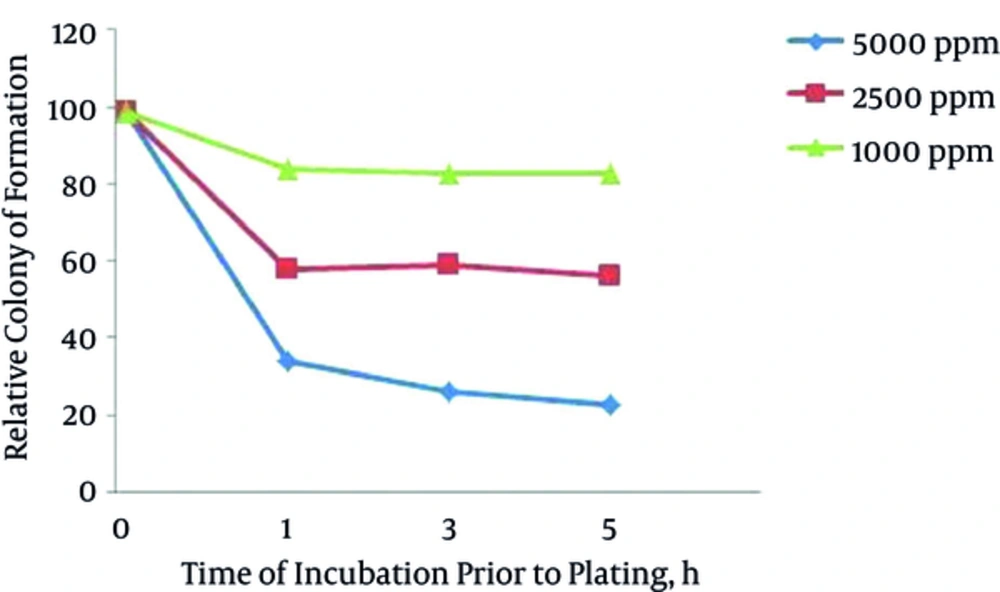
Different concentrations of AgNPs inhibited colony formation of F. oxysporum at different levels (Figure 1). As concentration of AgNPs increased, there was decreased colony formation. Silver nanoparticles at the highest concentration (5000 ppm) did not fully inhibit colony formation of F. oxysporum. Silver nanoparticles (5000 ppm) led to a decrease in F. oxysporum colonies to 35% after one hour of exposure, 27% after three hours of exposure, and 24% after five hours of exposure (Figure 2).
Images were captured after culture plates had been incubated for four days at 28°C.
Antifungal activity of AgNPs to reduce colony formation was apparent within one hour. Extended exposure to the spores of AgNPs (Figure 2) from one to five hours significantly reduced colony formation (P > 0.05).
5. Discussion
The control of fungal diseases in crops is very important. Recently, efforts to further develop management practices and lower the risks for human safety have been raised. In addition, several studies were carried out to test antifungal activity of AgNPs against plant pathogenic fungi. Results presented in this research confirm that AgNPs has an inhibitory effect on colony of spores of F. oxysporum. Silver nanoparticles may be directly attached to penetrate cell membranes to kill spores (12).
Researchers have used AgNPs, as carriers to deliver various loads of large biological molecules to specific targets. Targeting high-cargo density and concentration of nanoparticles may provide a specific target site and has the least side effects (13).
Introduction of silver in bacterial cells results in a high degree of structural and morphological changes, which can lead to cell death. As AgNPs come in contact with bacteria holds cell wall and cell membrane them (14). Introduction of AgNPs is sufficient to prevent a repeat and to cause cell death. In addition to increasing its effectiveness, when silver comes into contact with the liquid, it leads to an increase in ionized particles’ antibacterial activity (15). This leads to suppression of enzyme and protein expression, which is linked to the ability of cells to produce ATP related to the condition (16). Although it is a different offer for each type of cell, cell membrane components differ greatly, it has been seen that in general, AgNPs with an average size of 10 nm or less indicates that electronic effects, which to increase greatly its antibacterial activity (17). It has been noted that the introduction of AgNPs has synergistic activity with common antibiotics currently used today, such as erythromycin, ampicillin and vancomycin-resistant Staphylococcus aureus (18). Medical equipment, indicate that AgNPs reduces the number of bacteria on the device used (19).
5.1. Conclusion
Antifungal activity of AgNPs has a great potential for use in production of control fungal spores.