1. Introduction
Fascioliasis is a zoonotic infection caused by Fasciola Hepatica. Water or raw green vegetables contaminated with encysted metacercariae are the major sources of human infections. Penetrating the intestinal wall, the larva enters the peritoneal cavity, passing through the liver and hepatic tissue. They finally become mature in the biliary tract and start producing eggs (1). During the past decade the incidence of Fasciola hepatica infection has increased worldwide, and even in developed countries this infection is not rare (2). Bolivia, China, Ecuador, Egypt, France, Iran, Peru and Portugal host the greatest number of infected patients (3). Here we report a rare case of Fasciola hepatica infection that was primarily diagnosed as cholangitis when later the CT-scan revealed chronic cholecystitis.
2. Case Presentation
During April 2010, a 76-year-old woman from North West of Iran (Ardabil) was admitted to the internal medicine ward with prolonged fever. She had fever, abdominal pain, weight loss, and anorexia for six months. Before being admitted to Baharloo Hospital, she had undergone diagnostic and treatment processes for three months. Her symptoms included fever, severe malaise and anemia; magnetic resonance cholangiopancreatography (MRCP) was carried out, and showed no abnormality in the common bile duct and intrahepatic ducts, however, it revealed multiple lesions in the liver. The patient had undergone liver wedge biopsy and the pathological examination showed eosinophilic rich inflammation with necrosis that was suggestive of parasitic infection. Finally, before her new admission she was treated for visceral larva migrans.
In our physical examination, she was found ill and pale. Her vital signs were temperature 39°C, pulse rate 98 per minute, respiratory rate 24 per minute, blood pressure 110/80. No liver and spleen enlargement were noted on palpation. With intra-abdominal infection diagnosis, ceftriaxone and metronidazole were empirically started on the first hospital day and continued for ten days. After 48 hours she was feeling better but her abdominal pain, anorexia and sweating continued.
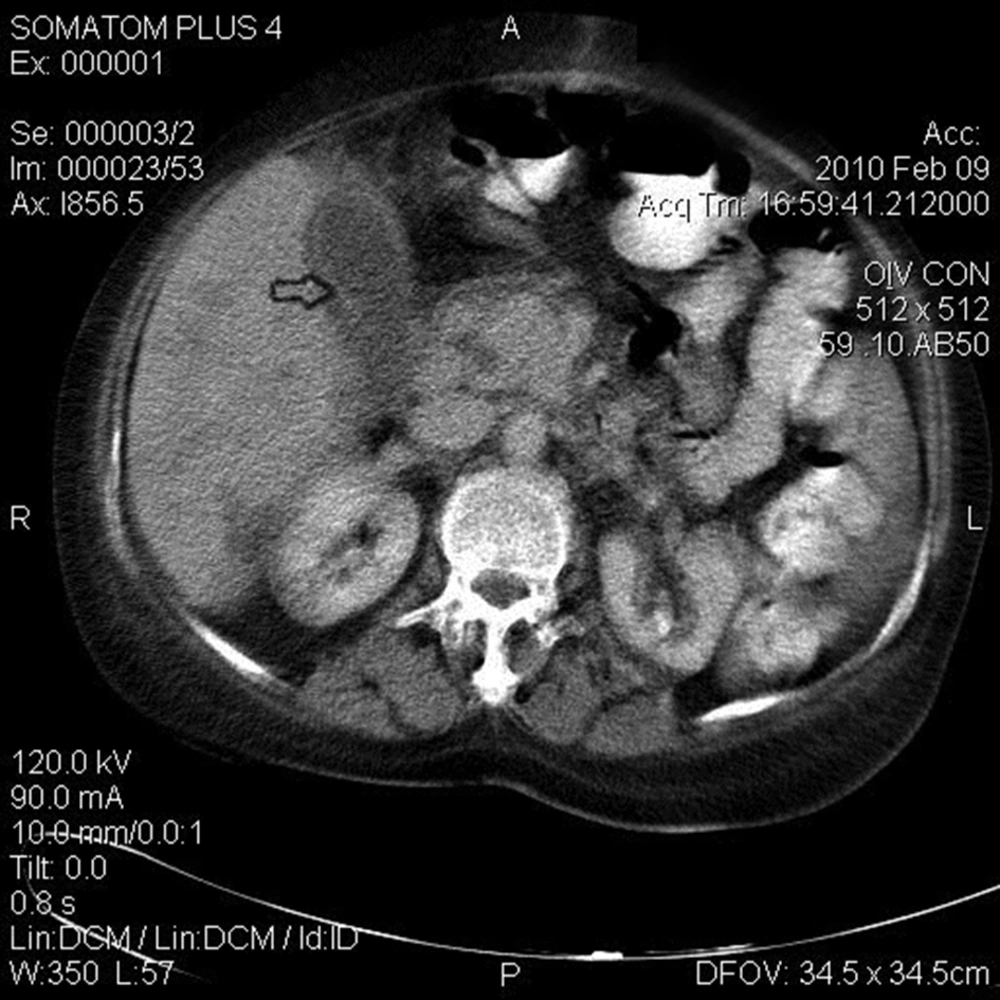
Laboratory investigations on the first hospital day revealed the following results: hematocrit 22.6% (normal, 33-45), mean corpuscular volume 84 fL (normal, 80-96 fL), white blood cell count 18500/mm3 (normal, 4000-11000) with 4% eosinophil on peripheral smear, platelet 562000/mm3 (normal, 150000-45000), erythrocyte sedimentation rate (ESR) 95 mm/hour, alanine aminotransferase (ALT) 27 IU/L (normal,5-37), aspartate aminotransferase (AST) 26 IU/L (normal, 5-37), alkaline phosphatase (ALP) 1125 IU/L (normal, 80-306), total bilirubin 1.2 mg/dL (normal ≤ 1.2mg/dL). In sonography, liver size and its parenchyma were normal, while gallbladder wall was thick without stone or sludge, common bile duct and intra hepatic ducts were also normal. Abdominal CT scan with contrast revealed a thick gall bladder wall without any stricture or stone in the distal common bile duct that suggested cholecystitis (Figure 1). Stool examination for ova (Fasciola hepatica) was negative, but ELISA for Fasciola hepatica was positive (nearly three times the normal value that is 11).
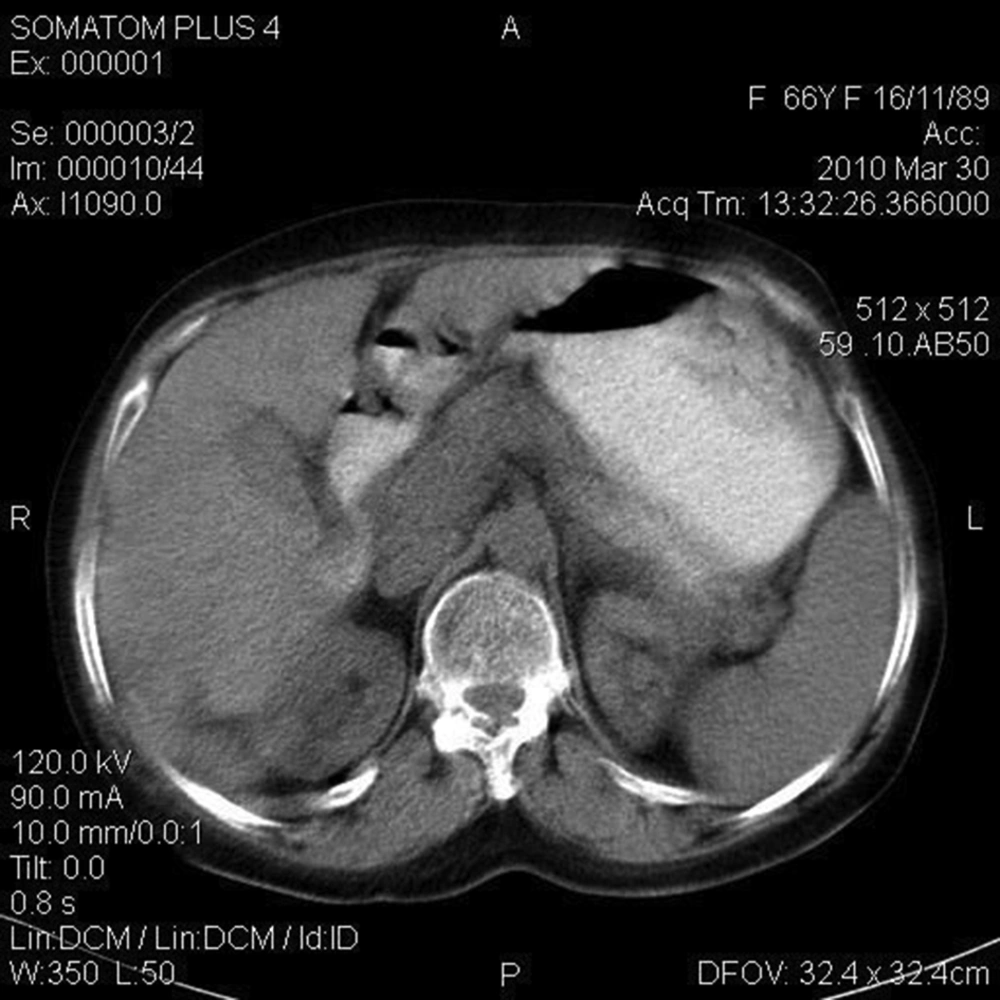
Considering the biopsy results (see above) that indicated eosinophilic rich inflammation with necrosis, on the 18th hospital day, the patient was given two individual doses of 10 mg/kg triclabendazole, for each 24 hours. Her symptoms completely disappeared after one week, on the 25th hospital day. During the one-year follow up, she had no symptoms, and abdominal CT scan became normal (Figure 2).
3. Discussion
As common diseases of livestock animals (e.g. sheep and cattle), fascioliasis targets humans only accidentally (4). Iran is frequently cited as one of the major countries with the greatest number of people infected by Fasciola hepatica (3, 5). Considering the popularity of consumption of vegetables such as lettuce, spinach and undercooked infected liver in Iran (6, 7), which are major sources of infection, the risk is significant. After human consumption of contaminated food products, two phases have been described in the evolution of the disease. 1- In the acute phase the larva enter the peritoneal cavity, the liver capsule, hepatic tissue and ultimately the bile ducts. During this phase the following symptoms might appear: urticaria, cough, shortness of the breath (8). Our patient did not present these symptoms. 2- In the chronic phase the parasite, now in bile ducts, is matured into adult flukes and produces eggs. During this phase the patient may be asymptomatic or present the following symptoms: cholangitis, biliary obstruction, cholecystitis, gallstones, pancreatitis and cirrhosis (8).
In the literature, there are several reports of acute cholecystitis due to Fasciola hepatica (9-11); extraction of viable fluke by endoscopic retrograde cholangiopancreatography (ERCP) was the diagnostic tool (10, 11). In the present case, however the ERCP was not applied. Instead, as noted earlier, the result of the liver biopsy implied parasitic infection. Also in the abdominal CT scan of the patient, thickening of the gallbladder wall was another indication (Figure 1). These along with mild eosinophilia are compatible with chronic fascioliasis. Clinical presentation of the patient, elevated alkaline phosphatase, elevated ESR, anemia, eosinophilia and fever suggested a stool examination for detection of Fasciola eggs as the first diagnostic step. The results were negative, thus immunodiagnostic test (ELISA) was carried out giving positive results. A sensitivity and specificity of 100% and 97.8%, respectively, was cited for the enzyme-linked immunosorbent assay (12, 13). This report aimed to emphasize that in chronic cholecystitis especially in endemic areas, we should have fascioliasis in our differential diagnosis.
Fascioliasis has different clinical presentations related to the hepatobiliary system; it may however be easily confused with other conditions. High index of suspicion is required to establish a correct diagnosis. Here we used imaging studies and immunological testing along with clinical presentation to achieve the diagnosis. The present report emphasizes that fascioliasis should be considered in the differential diagnosis of chronic cholecystitis, especially in endemic areas. This would help timely diagnosis of cholecystitis when facing patients with apparent abnormality.