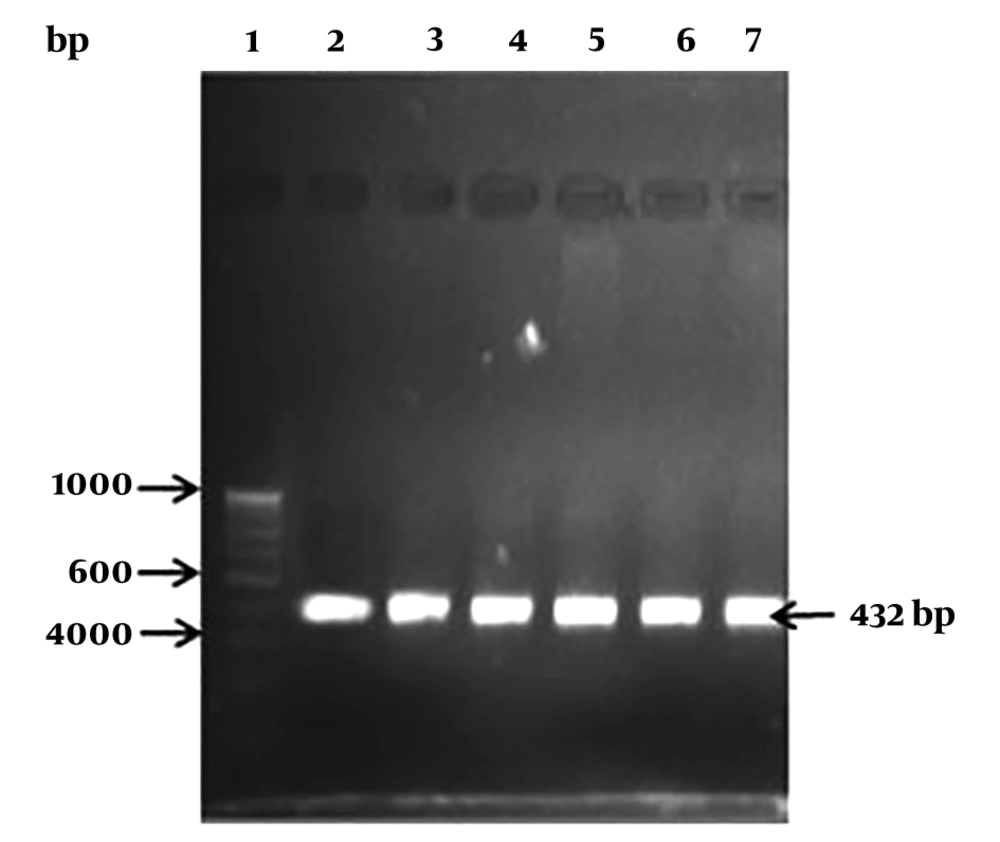
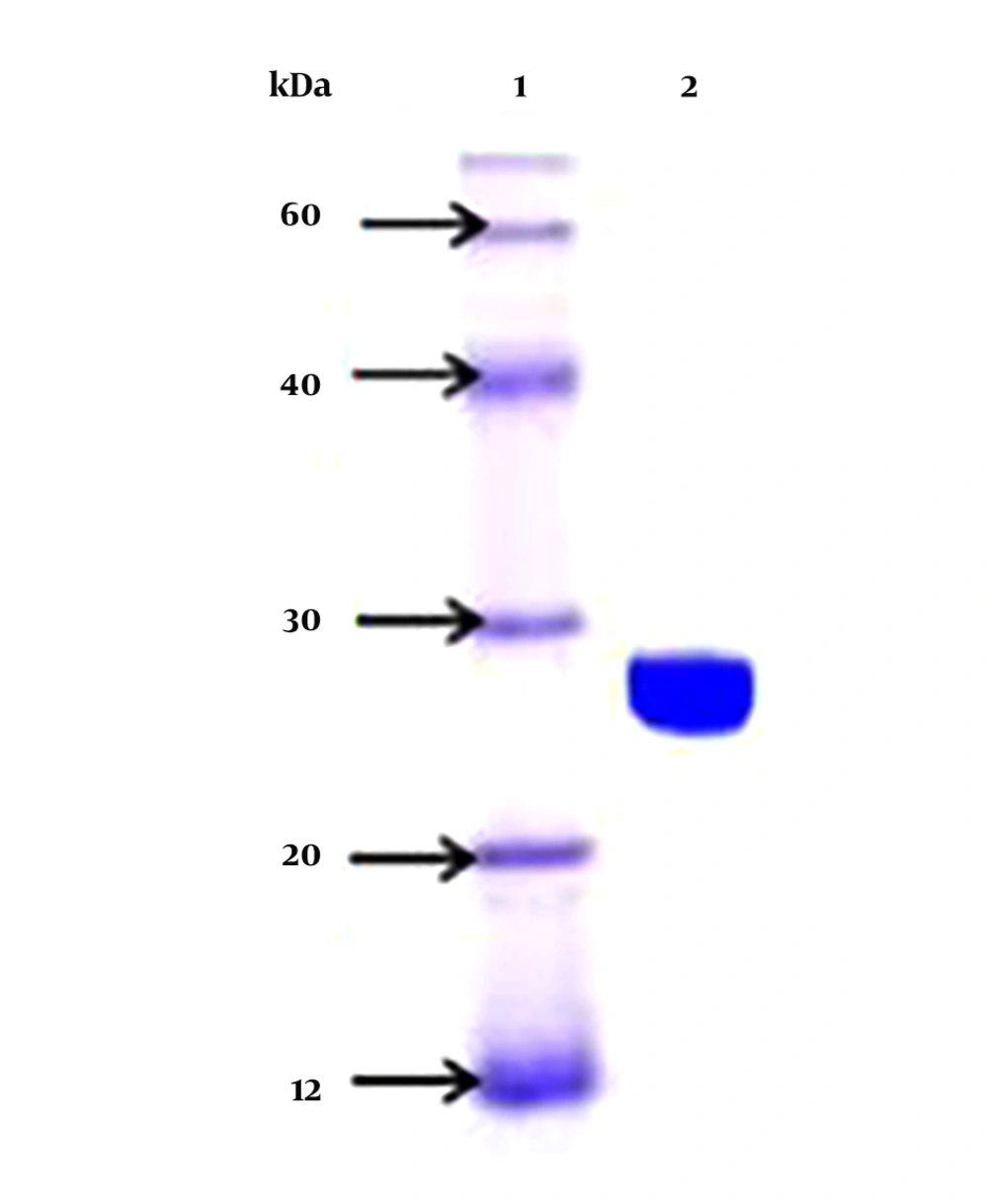
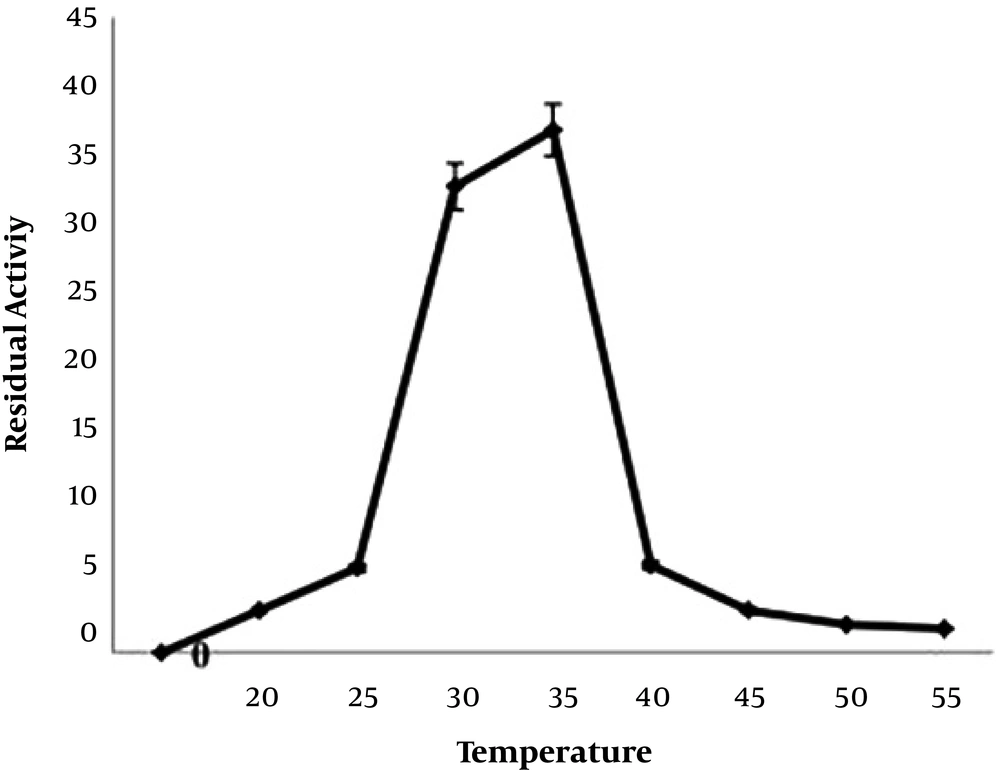
The selected isolates were microscopically examined and subjected to conventional biochemical tests, which include, gram-staining, catalase, oxidase, and β-hemolysis. Furthermore, carbohydrate fermentation test was performed with different sugars, such as sucrose, mannitol, rhamnose, maltose, inositol, xylose, fructose, and lactose, as described previously by Janzten et al. (
13). The molecular detection of
L. monocytogenes was performed by using hemolysin gene (
hlyA) species-specific primers. The bacterial genomic DNA from the native
L. monocytogenes and ATCC 19155 was extracted using the Quick-g DNATM miniprep kit (Zymo Research, SA). The protocol for the isolation of the genomic DNA was performed as described by the manufacturer. The
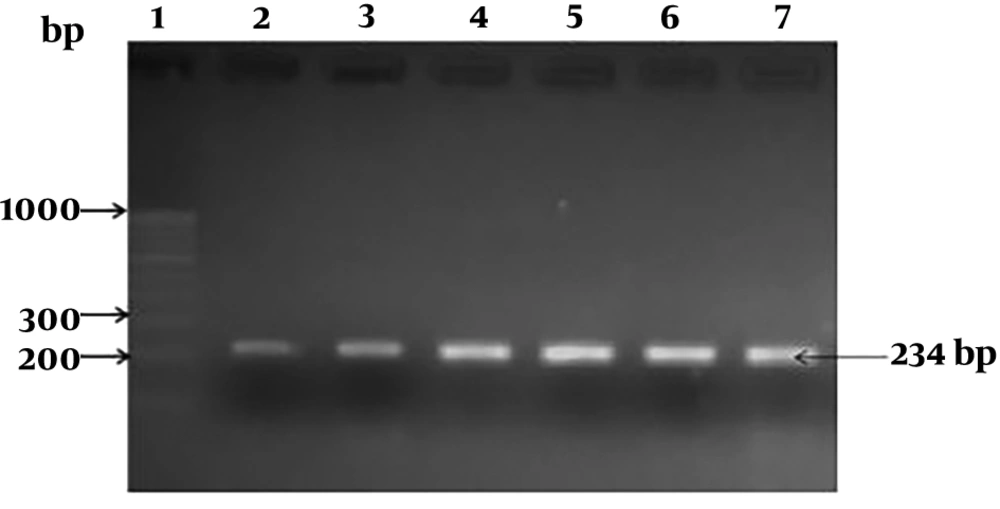
hlyA gene was amplified by using the primers F-5’CGGAGGTTCCGCAAAAGATG3’ and R-5’CCTCCAGAGTGATCGATGTT3’. The PCR was performed as described by Khan et al. (
14), and an amplicon size of 234 bp was considered as a positive result.