1. Background
Decrease in functional residual capacity (FRC) and atelectasis formation are well documented consequences of general anaesthesia. Longer duration and deeper levels of anaesthesia, high inspired oxygen fraction, lack of PEEP and muscle paralysis potentiate FRC reduction (1-5). Positive consequences of applied PEEP during general anaesthesia and mechanical ventilation refer as slowdown of alveolar de-recruitment and improvement of lung compliance (6-9). On the other hand, application of excessive PEEP can produce pneumothorax as well as hemodynamic instabillity. Current recommendations advocate use of PEEP 3 - 5 cmH2 in children with healthy lungs and also PEEP titration to prevent alveolar collapse (10). However, it is not stated how much PEEP we should titrate in order to improve lung function. This is due to lack of studies in children both healthy and sick (10). This research is a substudy of clinical trial (no.: NCT03745443) that investigated PEEP effect on lung function: oxygenation, ventilation, intrapulmonary shunting, lung mechanics and hemodynamics in preschool children. In previous paper we presented that PEEP titration in preschool children with healthy lungs during general anaesthesia up to 11 cmH2O improves oxygenation, reduces alveolar-arterial gradient without compromising hemodynamic stability (11).
2. Objectives
In this paper we will address to effects of the same slow PEEP titration on lung mechanics.
3. Methods
Methodology as well as study protocol were described in previous published paper but we will repeat it in the following text (1). We conducted a prospective, open label, randomized clinical trial in 70 children age 3 - 7 scheduled general anaesthesia for elective non-cardiothoracic surgery. Following ethical approval of our National Tertiary Health Care Centre (Institute for Mother and Child Health Care Ethic Committee, Institute for Mother and Child Health Care, Serbia, Chairperson K. Sedlecky, registration number No 8/30, November 2017), and after obtaining informed consent from parents/legal guardians, the subjects were recruited between January-June 2017. The study was performed in compliance with Declaration of Helsinki. Inclusion criteria were: age 3 - 7 years, ASA I and II, children undergoing orthopaedic, reconstructive, abdominal or urological surgery. Exclusion criteria were: present cardiac comorbidity and respiratory comorbidity, current or recent (up to 4 weeks) upper airway infection, children scheduled for cardiothoracic surgery and contraindication to anaesthetics used in study protocol. After triage recruits were randomly assigned in a 1:1 ratio, using “sealed envelope” technique into two groups; interventional (n = 35) and control (n = 35). The box containing 70 envelopes with allocation instruction (paper with group; I, interventional group; C, control group) was used to allocate patients randomly. Anaesthetist in charge pulled the envelope from the box and allocated patient to interventional or control group. General anaesthesia and intraoperative monitoring were the same in both groups. Midazolam 0.1 mg/kg i.v. was used for premedication 30 minutes before induction. We combined inhalation and intravenous induction using thiopental 5 mg/kg, fentanyl 3 mcg/kg, sevoflurane 1%, O2/air (35:65) and rocuronium 1 mg/kg. For Anaesthesia maintenance air/oxygen mixture (65%:35%), sevoflurane 1.5% - 2%, fentanyl 2 mcg/kg and rocuronium were used. Ventilator settings were the same in both groups (Datex Ohmeda, Avance CS2, GE anaesthesia machine) except in interventional group when trial intervention was performed: pressure control mode (PCV), inspiratory pressure (Pinsp) to achieve tidal volume of (Vt) 6 - 8 mL/kg, respiratory rate (RR) adjusted to keep end-tidal CO2 (EtCO2) 35 - 45 mmHg, PEEP 3 cmH2O. Intraoperative respiratory monitoring included: peripheral oxygen saturation (SpO2), capnography, EtCO2, Pinsp, peak airway pressure (PIP), mean airway pressure (Paw) and dynamic lung compliance (Cdyn), partial pressures of CO2 and O2. Due to goals of our clinical trial mentioned in Introduction and well known effect of PEEP on filling pressures we monitored invasive blood pressure (IBP) and EKG. All investigated variables were continuously monitored during anaesthesia in both groups using monitoring incorporated in anaesthesia machine. Twenty minutes before the end of surgery the trial intervention was performed in interventional group (11).
3.1. Trial Intervention
In the interventional group, 20 minutes before the end of surgery the PEEP was increased by 2 cmH2O on every 5th breath up to 11 cmH2O. Ventilation was maintained with a PEEP of 11 cm H2O for 2 minutes. The PEEP was then reduced by 2 cm of H2O at every 5th breath until a plateau of 5 cmH2O was reached. Ventilation with a PEEP of 5 cmH2O was maintained until extubation. The total time required for this intervention was approximately 5 minutes. The control group was ventilated with a baseline PEEP setting (3 cmH2O) until extubation. The criteria for aborting the experiment were: hypotension (defined as SBP < 70 mmHg and MAP < 55 mmHg.), bradycardia (defined as heart rate ≤ 60/min), desaturation (defined as SpO2 < 94%) and a sudden rise in PIP and/or PIP above 30 cmH2.
3.2. Study Outcomes
Our primary outcome was change in Cdyn. Secondary outcomes were changes in PIP, Paw and cases of desaturation in interventional group during PEEP titration. We obtained measurements for analysis in 2 time points defined as: TI, 20 minutes before the end of surgery; and TII, before extubation. We monitored Cdyn, Paw, PIP, and desaturation during PEEP titration in interventional group.
3.3. Statistical Analysis
Since this is a substudy, as stated in Introduction section, the sample used here is the same in previously published paper (11). In brief, calculation of the sample size was based on expected effect of PEEP on PaO2/FiO2 with α = 0.05 and study power of 80%. This parameter was discussed in previously published paper (11). The number needed was read from statistical tables and there were 30 participants per group (12). A further 5 were added in each group to compensate for dropouts. We needed 35 participants per group. All data were analysed using IBM-SPSS 20.0 (IBM Corp, New York, USA) software. The results were presented as mean and standard deviation with CI 95% or proportions. To evaluate size effects that PEEP titration caused, delta value (Δ) was used and compared between groups. The differences between groups were tested using independent t-test at given endpoints. Within the interventional group variation of variables were explored using ANOVA. All tests were 2-tailed and a P < 0.05 was considered statistically significant.
4. Results
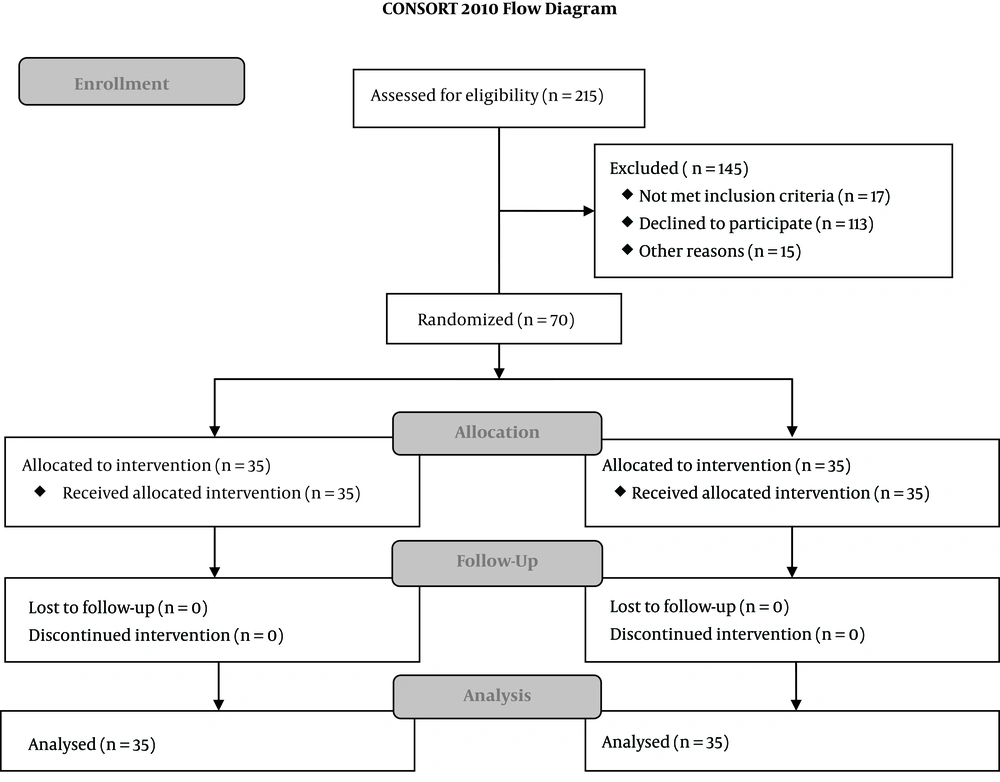
The demographic data of the study groups are presented in Table 1. There were no differences between groups regarding age, gender, body weight and type of surgery. The participant flow is shown in the CONSORT flow diagram (Figure 1). Of 486 children scheduled for surgery between January 2017-June 2017, 271 were younger than 3 or older than 7 years of age, 113 refused participation, 10 had upper respiratory tract infections, 7 had a chronic heart or lung condition and 15 didn’t show up for surgery. Recruited participants (n = 70) finished the study and were analysed, there were no dropouts.
4.1. Main Outcomes
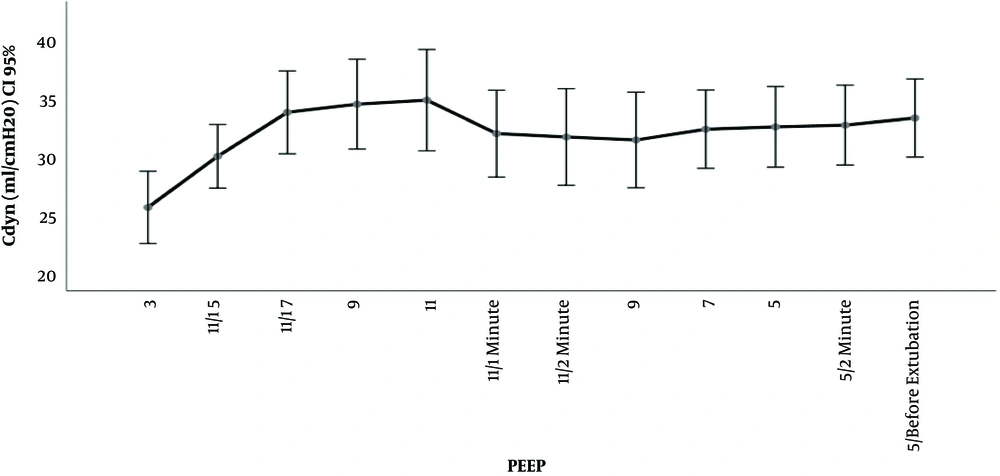
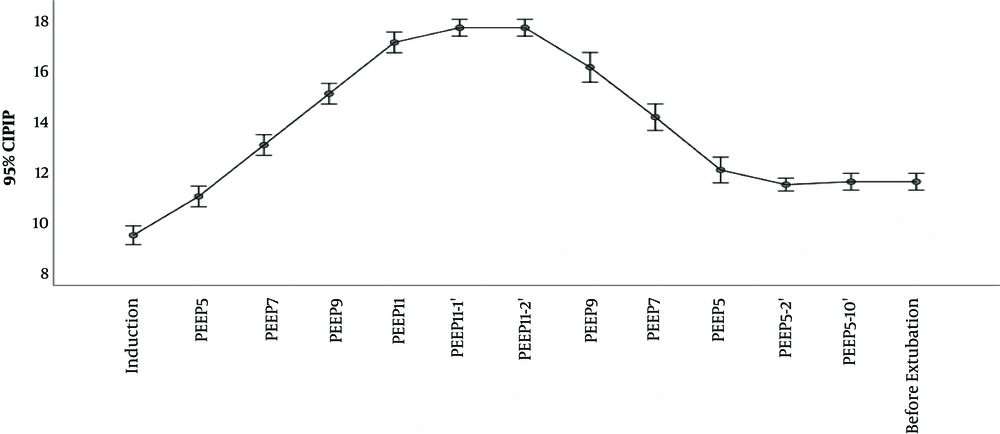
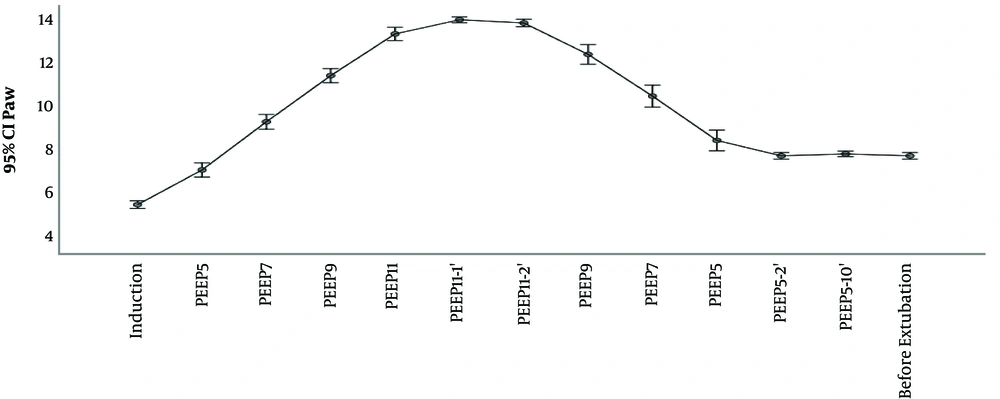
The outcomes Cdyn, PIP, Paw are presented in Table 2 and Figures 2 and 3 and 4. We used delta (Δ) to evaluate effect size PEEP titration caused and compared it between groups. Independent t-test for delta between groups revealed that there was a statistically significant difference in the magnitude of change in investigated parameters between the groups and all were P < 0,001: Cdyn (ΔTI-TII I group -3.2 vs C group 0.63); PIP (Δ TI-TII I group -0.57 vs C group 0.11) and Paw (ΔTI-TII I group -0.63 vs C group 0.0). The changes in lung compliance, PIP, Paw during PEEP titration in the Interventional group are presented in Figures 2-4.
| Interventional | Control | Pc | |
|---|---|---|---|
| Cdyn | |||
| TI | 30.1 (27.4 - 32.8) | 27.8 (24.9 - 30.6) | 0.225 |
| TII | 33.4 (30.1 - 36.7) | 27.1 (24.3 - 30.0) | 0.005 |
| ΔTI-TII | - 3.2 (-4.5 - -1.9) | 0.63 (0.25 - 1.01) | < 0.001 |
| PIP | |||
| TI | 10.9 (10.5 - 11.3) | 10.6 (9.9 - 11.4) | 0.530 |
| TII | 11.5 (11.2 - 11.8) | 10.5 (9.8 - 11.3) | 0.020 |
| ΔTI-TII | - 0.57 (-0.9 - -0.3) | 0.11 (0.0 - 0.2) | < 0.001 |
| Paw | |||
| TI | 7.14 (6.8 - 7.5) | 6.1 (5.8 - 6.5) | < 0.001 |
| TII | 7.8 (7.6 - 7.9) | 6.1 (5.8 - 6.5) | < 0.001 |
| ΔTI-TII | - 0.63 ( -0.9 - -0.3) | 0.0 (0.0 - 0.0) | < 0.001 |
aValues are expressed as mean (95% CI).
bTI-20 minutes before the end of surgery; T II -before extubating.
cIndependent samples t-test.
5. Discussion
In this research we found out two things: first, lung compliance improved after PEEP titration in interventional comparing to control group at the end of surgery, and second, maximal PEEP level of 11 cmH2 did not cause increase in airway pressure that could lead to pneumothorax.
We used dynamic lung compliance as a marker of lung mechanics (13). Dynamic lung compliance is measured in presence of gas flow and is inversely related to PIP: [(Cdyn = Vt/(PIP - PEEP)]. Therefore, changes in respiratory mechanics can be spotted immediately, at bedside. This is very important in clinical setting since it enables clinician to estimate effect of treatment immediately. As we demonstrated, lung compliance improved in interventional group comparing to control after PEEP titration at the end of surgery. Similar results exist in available literature. In experimental studies on animals, with and without induced lung injury, PEEP titration of 5, 10, 15 or 20 cmH2O caused the increase of lung compliance (14-16). Recent study in adult patients ASA I and II, without cardiorespiratory comorbidity demonstrated that application of PEEP from 4 up to 12 cmH2O improved lung compliance (17). Substudy of the PROVHILO trial also demonstrated that in adult patients without cardiorespiratory comorbidities lungs became more compliant when PEEP 12 cmH2O was applied (18). When it comes to children with healthy lungs and idea of preventive role of PEEP literature data are limited. Recently, Cruces et al. (19) demonstrated that application of PEEP 5 cmH2O in children up to 15 years (median 4 years) resulted in improvement of respiratory compliance. Similar studies, mostly using PEEP 4 - 5 cmH2O, were also published earlier. Due to experimental data in lung model, we know that improvement in lung compliance is a result of reduction in non-aerated and poorly aerated airspaces (20). Titration of PEEP up to 15 cmH2O reduced surface of collapsed and poorly aerated airspaces from 31% with 0 PEEP to 7% with initial recruitment happening at a PEEP less than 10 cmH2O (20). As much PEEP affects these lung regions, it also increases volume of already normally aerated alveoli which can lead to overdistension. Experimental studies in animals demonstrated overdistension with a PEEP of 9 cmH2O but not with a PEEP 7 - 8 cmH2O (21, 22). On the other hand, analysis from PROVHILO substudy concluded that incidence of overdistension between groups (comparing PEEP 12 and 2 cmH2O) wasn’t significant, in only one patient overdistension was observed (21). In our study, during the slow step up PEEP titration, the lung compliance increased reaching the maximum at a PEEP of 11 cmH2O. After 2-minute ventilation on PEEP 11 cmH2O there was clinically insignificant fall of compliance by 9%. It is possible that this was because of overdistension according to mentioned experimental data (20). Further investigation and measurements of lung mechanics are needed in order to draw conclusion form this observation. For now, we can state that overall effects of stepwise up and down PEEP titration from 5 - 11 cmH2O had a positive effect. The dynamic compliance in the interventional group at the end of surgery was better than that of the control group.
Awareness of potential risk of barotrauma among clinicians limits to some extent application of high PEEP. Elevated PIP and Paw have been implicated as being traumatic for lung parenchyma. High PIP is associated with pneumothorax, whereas elevated Paw is associated with pneumothorax and reduction in cardiac output (23). In the case of tension pneumothorax, insidious complication of mechanical ventilation, PIP will rise and dynamic lung compliance will fall. In clinical practice, in order to induce such trauma, besides PEEP, set inspiratory pressure has to be high too (since, in pressure control mode, PIP is sum of inspiratory pressure and PEEP). As experimental data show, distension pressures that could induce barotrauma are greater than ones applied in everyday practice. Even with PEEP of 20 cmH2, high inspiratory pressure over 60 cmH2 was the factor that caused pneumothorax among laboratory animals (24). One of the safety limitations of our study was sudden rise in PIP above 30 cmH20. In our study, maximal PIP was 17 cmH2O and maximal PEEP level used was 11 cmH2. During PEEP titration, as stated in methodology section, we didn’t change preset inspiratory pressure, which was adjusted to achieve Vt 6 - 8 mL/kg, so the only factor that could influence PIP was PEEP titration. No desaturation or pneumothorax was observed in investigated group. Therefore, we interpreted that presented PEEP titration in children with healthy lungs came out as safe.
The limitation of our study is open label design. There was no possibility for blinding because of the randomization design which means that anaesthetist in charge had to know on whom and when to perform the PEEP maneuver.
5.1. Conclusions
Administration of staggered PEEP from 5 - 11 cmH2O 20 minutes before end of anaesthesia in Anaesthetized ventilated children without cardiorespiratory comorbidity improved lung compliance without posing a risk of barotrauma and subsequent pneumothorax.