1. Background
The outbreak of pneumonia that emerged early in 2020 in Wuhan, Hubei province, China was identified as a great public health challenge worldwide on January 7th (1). The cause of the pneumonia outbreak was subsequently detected to be a novel coronavirus (CoV) that belongs to the beta-coronavirus lineage B (2). Due to the rapid spread of the virus in China and other countries, the World Health Organization (WHO) announced that the outbreak of Coronavirus Disease 2019 (COVID-19) constitutes a Public Health Emergency of International Concern (3). Several reports proposed that children are just as likely as adults to suffer from COVID-19, but have a lower fatality rate and are less likely to experience severe forms of disease. For various reasons, such as an underestimation of the true rate of infection and the lack of examination of children with mild symptoms, the knowledge regarding COVID-19 in children and adolescents is limited (4). Similarly to adults with COVID-19, respiratory tract manifestations such as cough and fever are the most common symptoms of the disease in children and adolescents (5, 6). Based on recently published evidence, an alarmingly high prevalence of gastrointestinal symptoms of COVID-19 has been reported among children. In this regard, a study by Fang et al estimated that the prevalence of diarrhea and/or vomiting in children with COVID-19 was much higher than that in adults (57.1% vs. 17.6%) (7).
It was subsequently documented that this virus can bind to the angiotensin-converting enzyme 2 (ACE2) receptors located in the intestinal and respiratory tracts and, via these receptors, the virus can penetrate into epithelial cells (8). However, an elevation of liver enzymes has been found in children, as well as adults, but a reason for this change in laboratory findings is still unclear (3, 9). Although COVID-19 is thought to be less severe in children than in adults, it should be noted that, in some instances, children and adolescents are more vulnerable than adults. Also, gastrointestinal and liver disorders can prolong the hospital stay and lead to treatment complications (8, 10-12).
In this systematic review and meta-analysis, we have analyzed the prevalence of gastrointestinal symptoms and the dynamics of liver enzymes in children with COVID-19, based on the reported cases up to date. Comprehending the gastrointestinal manifestations of COVID-19 in this age group is important for a better identification of children and adolescents with possible COVID-19 infection and for a better therapeutic management of these cases, thus limiting the spread of this virus worldwide.
2. Methods
2.1. Search Strategy
The present study was conducted in accordance with the PRISMA [preferred reporting items for systematic review and meta-analysis] guidelines (13). We carried out a comprehensive systematic search in PubMed/MEDLINE, Web of Science, SCOPUS and Embase from inception until May 2020 without using time or language restrictions. The following keywords were used in combination with the wild-card ‘*’ and Medical Subject Heading (MeSH) terms: (“2019-nCoV- 2” OR “coronavirus” OR “COVID-19” OR “SARS-CoV-2” OR “nCoV” OR “novel coronavirus” OR “coronavirus 2019”) and (“child*” OR “Adolescent*” OR “Pediatrics*”) AND (“abdominal pain” OR “Nausea” OR Vomiting” OR “Fecal incontinence” OR “Gastrointestinal Diseases” OR “Digestive System Diseases” OR “Esophageal Diseases” OR “Gastroesophageal Reflux” OR “GERD” OR “Constipation”). Additionally, the reference lists of the articles retrieved and related review studies were also hand-screened to identify eligible publications that our search might have omitted.
2.2. Study Selection
After excluding duplicate articles, two authors independently reviewed the titles, the abstracts or the full-text of the retrieved studies to detect eligible publications. Finally, original studies were included in the present meta-analysis if they met the following criteria: (1) the articles were case reports/case series, prospective/retrospective cohort studies, case-control studies, or randomized controlled trials; (2) the studies enrolled pediatric/children/adolescents (aged < 18 years); and (3) the articles reported digestive symptoms or dynamics of liver enzymes (ALT, AST and LDH). Duplicate data, studies with unclear information, studies involving animals, reviews and studies whose corresponding author did not offer any feedback after several emails were excluded.
2.4. Data Extraction
Two independent researchers reviewed the data and an additional reviewer resolved any disagreements between the two. The following information was collected: author, year of publication, country, study date, age, disease severity, participants’ gender, presence of fever, respiratory symptoms, digestive symptoms (diarrhea, vomiting or nausea and abdominal pain) and elevation of ALT, AST and LDH.
2.5. Data Synthesis and Statistical Analysis
The statistical analysis was conducted using RevMan V.5.3 software and STATA version 12.0 (Stata Corp, College Station, TX, USA). The prevalences of gastrointestinal symptoms and elevated liver enzymes were expressed as proportions and 95% confidence intervals (95% CI) using the random effects model, and presented as Forest plot. .Heterogeneity was examined using the I-squared (I2) statistic, in which the source of heterogeneity was determined if the I2 value was > 50% (4). We assessed the presence of publication bias using the formal Egger’s test (14).
3. Results
Appendix 1 in Supplementary File shows a flowchart of the study selection process and reasons for excluding articles. Following the systematic search, 574 publications from the aforementioned electronic databases were retrieved. After excluding duplicate studies, a total of 469 publications remained. Then, we reviewed the title/abstract of the remaining studies, and excluded 417 articles which did not meet the inclusion criteria. A total of 52 articles was retrieved during the secondary screening (by full-text). Of these, 33 studies were discarded since they did not include data of interest. Finally, 19 studies (1, 10-12, 15-29) met the eligibility criteria and were included in the quantitative meta-analysis.
3.1. Study Characteristics
The characteristics of the pooled studies are presented in Table 1. All studies were conducted in China except for one publication (11) whose authors were from Malaysia. All articles were published between January 1st and March 10th, 2020. In total, 415 participants were included in our pooled study. The median age of the patients was 6.2 years, and 53.7% of them were males. A number of thirteen (10-12, 15-17, 19, 21, 24-28) studies reported mild to moderate manifestations of the disease, while four studies (18, 20, 22, 23) reported evidence of severe symptoms. Two papers did not provide enough information regarding the severity of COVID-19 (1, 29). Among the studies that reported disease severity, severe disease accounted for 1.8% - 100% in Lu et al. (20) and Qiu et al. (10) studies , respectively. We defined the clinical types of COVID-19 in pediatric patients based on Qiu’s et al. (10) study. Other variables include treatment schedule and medications and duration of hospital stay are completely presented in Table 1.
| Author (year) | Country | Study Date | No. | Age (Mean or Median [± 1 SD or Range) | Male (%) | Disease Severity a , No. (%) | Treatment Schedule and Medications | Duration of Admission Time | Fever, No. (%) | Respiratory Symptoms, No. (%) | All GI Symptoms b , No. (%) | Diarrhea, No. (%) | Nausea/ Vomiting, No. (%) | Abdominal Pain/ Discomfort, No. (%) | ALT, U/L Elevated, No. (%) | AST, U/L Elevated, No. (%) | LDH, U/L Elevated, No. (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lu | China | January 28 to February 26, 2020 | 171 | 7 (1d-15 years) | 104 (61) | Severe type :3 (1.8); | NA | NA | 55 (32) | Cough:83 (49); Pharyngitis: 79 (46); Nasal congestion:9 (5); Rhinorrhea: 13 (8); Tachypnoea: 49 (29); Pneumonia 113 (64.9) | 15 (9) | 15 (9) | NA | NA | 21 (12) | 25 (150 | NA |
| Qiu | China | January 17 to March 1, 2020 | 36 | 8 years | 13 (36) | Mild type:17 (47); Moderate type:19 (52); | General; support therapy; monitoring of lung, liver, kidney, and; myocardial functions; active control over high fever; oxygen uptake if necessary; interferon alfa and lopinavir-ritonavir | 14 (3, 10-20) | 4 (11) | Cough:7 (19); Pharyngitis: 4 (11); Tachypnoea: 1 (3); Sore throat:2 (6); | 4 (12) | 2 (6) | 2 (6) | NA | 2 (6) | 3 (8) | NA |
| Zhang | China | January 1 to February 25, 2020 | 34 | 5 (1-6) years | 14 (41) | NA; | antibiotic therapy antiviral therapy interferon-α nebulization corticosteroid therapy; Oxygen inhalation | 3 (2-4); between admission and recover from fever | 26 (76) | Cough:20 (59); Tachypnoea: 3 (9); | 8 (24) | 4 (12) | 4 (12) | NA | NA | NA | 28 (82) |
| Wang | China | January 25 to February 21, 2020 | 31 | 6 months-17 years | 15 (50) | Asymptomatic type in 4 cases (13%), Mild type in 13 cases (42%), and Common type in 14 cases (45%) | Interferon; Oseltamivir; Ribavirin; Arbidol; Lopinavir | NA | 20 (64.5) | Cough: 14 (45); Sore throat: 2 (6.4) | 5 (16) | 3 (10) | 2 (6.4) | NA | Both 6 (22) | 2 (8) | |
| Tang | China | January 16 to February 8, 2020 | 26 | 7 years | 17 (65) | Mild type:8 (31); Ordinary:18 (69); | Oseltamivir, ribavirin, interferon, kaletra and traditional Chinese medicine | 13.6±1.03 | 11 (42) | Cough:12 (46); Rhinorrhea: 2 (8); | 4 (16) | 2 (8) | 2 (8) | NA | 3 (12) | 3 (12) | 12 (46) |
| Xia | China | January 23 to February 8, 2020 | 20 | 2 years (1.5 mon) | 13 (65) | Mild type:100 (20); | Based on Protocol for COVID‐19 (Fifth Revised Edition)” distributed by the; National Health Commission | 12.9 days (8‐20) | 12 (60) | Cough: 13 (65); Nasal discharge: 3 (15); Sore throat: 1 (5); Tachypnea: 2 (10) | 5 (25) | 3 (15) | 2 (10) | NA | ; 5 (25) | ; NA | NA |
| Xin | China | January 17 to February 29, 2020 | 13 | 7.9 years | 4 (30) | Mild type:7 (53); Common type:5 (38); Severe type:1 (7) | Interferon; Ribavirin; Arbidol; Lopinavir | 13 | 6 (46) | Cough: 5 (38); Nasal discharge: 3 (15); Throat discomfort: 2 (15); Tachypnea: 2 (10) | 4 (30) | 2 (15) | 1 (7) | 1 (7) | NA | NA | NA |
| Chen | China | 11-Feb-20 | 12 | 14.50 (9.25-15.75) years | 6 (50) | Mild pneumonia:10 case; asymptomatic infection: 2 cases | Interferon; Ribavirin; Lopinavir | NA | 7 (58.3) | Cough: 9 (75); Upper airway symptom: 2 (16.7) | 4 (33.3) | 4 (33.3) | NA | NA | NA | NA | NA |
| Tan | China | January 27 to; March 10 | 10 | 7 years | 3 (30) | Mild type:10 (100) | Empirical antibiotic therapy | 2 | 4 (40) | Cough: 3 (30); Convulsion:1 (10) | 2 (20) | NA | 1 (10) | 1 (10) | NA | 2 (20) | 1 (10) |
| Cai | China | January 19 to February 3, 2020 | 10 | 6 (3 mon-11 years | 4 (40) | NA; | Empirical antibiotic therapy | 14 | 7 (70) | Cough:6 (60); Pharyngitis: 4 (40); Nasal congestion:3 (30); Rhinorrhea: 2 (20) | NA | NA | NA | NA | 1 (10) | 2 (20) | 3 (30) |
| Xu | China | December to February 20, 2020 | 10 | 6 (2 mon-15 years | 7 (70) | Mild type:10 (100) | Antiviral therapy with α-interferon oral; spray initiated from admission (8,000 U, two sprays, three times; a day) and zithromycin | 9 | 6 (60) | Cough:5 (50); Pharyngitis: 4 (40); Nasal congestion:2 (20); Rhinorrhea: 2 (20) | 3 (30) | 3 (30) | NA | NA | 1 (10) | 2 (200 | 2 (20) |
| Shen | China | January 8 to February 19,2020 | 9 | 7.5 years | 3 (33) | Mild type:9 (100) | Lopinavir/ritonavir; Azithromycin; meprednisone | 18 | 3 (33) | Cough: 1 (11); Sore throat: 1 (11) | 2 (22) | 2 (22) | NA | NA | NA | 2 (22) | 1 (11) |
| Sun | China | January 24 to February 24, 2020 | 8 | 6.81 years | 6 (75) | Severe type :5 (62); critically; ill:3 (37) | High-flow oxygen therapy; mechanical ventilation; Antiviral treatments (virazole, oseltamivir; and interferon). Antibiotics, traditional Chinese medicine; intravenous glucocorticoids and immunoglobulin | 19 | 6 (75) | Cough: 6 (75); Unilateral pneumonia:2 (25); Bilateral pneumonia:6 (75) | 6 (75) | 3 (37) | 4 (50) | NA | ; 4 (50) | ; NA | 5 (62) |
| Han | China | January 31; to February 16, 2020 | 7 | 1.3 (0.2‐13 years | 4 (57.1%) | Mild type:7 (100) | Oxygen inhalation; Empirical antibiotic treatment; Antiviral treatment; Glucocorticoids | NA | 5 (71.4) | Cough: 5 (71.4); Shortness of breath:3 (42.9); Pharyngalgia: 1 (14.3) | 4 (57.1) | 2 (28) | 2 (28) | NA | 1 (14.3) | 3 (42.9) | 2 (28) |
| Liu | China | January 7 to January 15, 2020 | 6 | 3 years | 2 (33) | Severe type :1 (16) | Pooled immune globulin from healthy donors. Antiviral agents, antibiotic agents, and supportive; therapies | 7.5 (5-13) | 6 (100) | Cough: 6 (100); | 4 (66) | NA | 4 (66) | NA | NA | NA | NA |
| see | Malaysia | until end of February, 2020 | 4 | 6.4 years | 3 (75) | Mild type: 4 (100) | None of the children required treatment with anti-virals | 10.3 | 2 (50) | Cough: 2 (50); Runny nose:1 (25); Wheeze:1. (25) | 1 (25) | 1 (25) | NA | NA | NA | NA | NA |
| Xia lu | China | 3 | 7.2 years | 1 (33) | Mild type:3 (100) | NA | 10 | 3 (100) | Cough: 1 (33); nasal congestion: 2 (66); rhinitis: 2 (66) | 2 (66) | 2 (66) | NA | NA | NA | NA | NA | |
| China | January 23 to February 5, 2020 | 3 | 5 (1-6) years | 2 (66) | Mild to moderate type:3 (100) | Oral Ribavirin and traditional Chinese | 23 | 3 (100) | NA | NA | NA | NA | NA | NA | NA | 1 (33) | |
| Ji | China | January 25 to February 3, 2020 | 2 | 12 years | 2 (100) | Mild type:2 (100) | Symptomatic treatment; Oral probiotic | NA | 0 (0) | pharyngeal congestion:1 (50) | 1 (50) | 1 (50) | NA | NA | NA | NA | NA |
Abbreviations: SD, standard deviation; IQR, interquartile range; GI, gastrointestinal; NA, not available; No, number; ALT, alanine aminotransferase (normal range 0 - 42 U/L); AST, aspartate aminotransferase (normal range 0 - 37 U/L); LDH, Lactate dehydrogenase (normal range 0 - 225 U/L)
aSevere disease was defined as the American Thoracic Society and Infectious Disease Society of America guidelines for community-acquired pneumonia, need of ICU admission, and death.
bIf all gastrointestinal symptoms were not reported and the number of events of any individual GI symptoms was less than one, it was regarded as “not available” and was excluded from the meta-analysis of all gastrointestinal symptoms. However, this study was still included in the meta-analysis of individual gastrointestinal symptom if the proportion of patients with that symptom was reported.
3.2. Meta-Analysis
Based on data from 17 studies with a total of 407 participants, we detected an overall prevalence of gastrointestinal symptoms (diarrhea, nausea/vomiting, abdominal pain and constipation) in children with COVID-19 of 26% [95% CI: 0.18 - 0.35 with significant heterogeneity noted among the included studies (P < 0.001; I2 = 67.66%)]. For the individual digestive symptoms, 15 studies with 391 participants reported the prevalence of diarrhea, 10 with 191 participants reported the prevalence of nausea/vomiting, and 2 reported the prevalence of abdominal pain. The pooled prevalence was: diarrhea = 12% (95% CI: 0.08 - 0.16) with no heterogeneity (P = 0.19; I2 = 23.53%) among the included studies (Figure 1A) and nausea/vomiting = 11% (95% CI: 0.05 -0.17) with no heterogeneity among the included studies (P = 0.06; I2 = 47.65%) (Figure 1B).
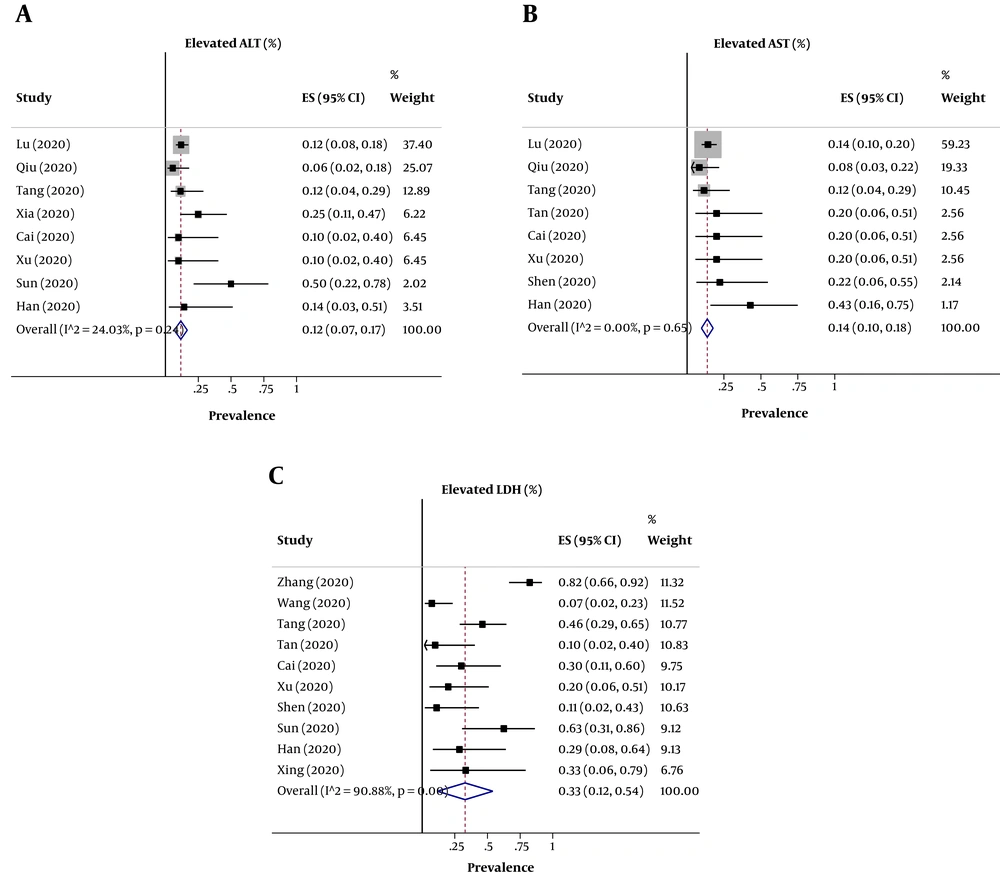
Regarding the individual elevation in liver enzymes, the pooled prevalence of elevated ALT levels in 293 participants was 12% (95% CI: 0.07 - 0.17) (Figure 2A) with no heterogeneity (P = 0.24; I2 = 24.03%) among the included studies, of elevated AST levels in 284 participants was 14% (95% CI: 0.10 - 0.18) with no significant heterogeneity among the included studies (P = 0.65; I2 = 0.0%) (Figure 2B), and of elevated LDH levels in 144 participants was 33% (95% CI: 0.12 - 0.54) (Figure 2C) with a high heterogeneity among the included studies (P < 0.001; I2 = 90.88%). The results, including data regarding the heterogeneity of the variables, are shown in Table 2.
| Variable | Pooled Value | 95% CI | I2 (%) | P Within Group |
|---|---|---|---|---|
| All GI symptom | 17 (26) | 0.18 - 0.35 | 67.66 | < 0.001 |
| Diarrhea | 15 (12) | 0.08 - 0.16 | 23.53 | 0.19 |
| Nausea/vomiting | 10 (11) | 0.05 - 0.17 | 47.65 | 0.06 |
| Elevated ALT | 8 (12) | 0.07 - 0.17 | 24.03 | 0.24 |
| Elevated AST | 8 (14) | 0.10 - 0.18 | 0.0 | 0.65 |
| Elevated LDH | 10 (33) | 0.12 - 0.54 | 90.88 | < 0.001 |
3.3. Publication Bias
The evaluation of publication bias by visual inspection of the funnel plot and Egger’s test demonstrated some evidence for publication bias in the meta-analysis of digestive symptoms (diarrhea and nausea/vomiting) of COVID-19 in children (P < 0.001) (Appendix 2 in Supplementary File). However, the results of the meta trim and fill analysis found no study. Egger’s linear regression test for elevated ALT (P = 0.20), AST (P = 0.11) and LDH (P = 0.82) revealed no publication bias (Appendix 3 in Supplementary File).
4. Discussion
To this date, the increasing number of articles have documented an involvement of the digestive system in children and adolescents with COVID-19. The pooled prevalence of all gastrointestinal symptoms (diarrhea, nausea/vomiting, abdominal pain and constipation) was 26%. Diarrhea was the most common gastrointestinal manifestations (12%), followed by nausea/vomiting (11%). Abdominal pain/discomfort was reported only in two studies. We also observed that some children and adolescents with COVID-19 had elevated levels of ALT (12%), AST (14%) and LDH (33%) during the course of the illness. Although diarrhea is one of the most common gastrointestinal symptoms of COVID-19, the presence of abdominal pain and constipation should not be ignored.
Emerging evidence suggests that, as the severity of the disease increases, digestive symptoms become more pronounced. Based on the evidence coming from adults, about 10% of patients with COVID-19 presented with digestive symptoms alone without respiratory manifestations. Unfortunately, this feature delays the diagnosis of COVID-19 and enables the rapid spread of the disease in the population especially in children (9). Moreover, subjects with digestive symptoms have an increased risk of critical illness, and can progress to acute respiratory distress syndrome (30, 31).
The digestive symptoms of COVID-19 in children and adolescents were various, and included diarrhea, vomiting, abdominal pain and constipation. There is little evidence regarding the histopathological changes which can affect the digestive system of the patients with COVID-19. For example, segmental dilatation and stenosis of the small intestine has been seen in an 85-year-old man diagnosed with COVID-19 (32). However, it is uncertain whether this finding was related to COVID-19 or merely incidental. Thus, further research is needed to clarify gastrointestinal involvement in COVID-19.
Remarkably, fever and cough are still the most common symptoms of the disease (16). Several reports indicate that the occurrence of gastrointestinal symptoms increases with the severity of COVID-19. However, as reported in a recent study, patients with gastrointestinal symptoms are less likely to be treated and most likely to be discharged versus patients without gastrointestinal symptoms, probably because there is insufficient knowledge regarding the possible gastrointestinal involvement in COVID-19 (33). On the other hand, Han et al. (16) stated that one of the reasons for the high prevalence of gastrointestinal symptoms in children with COVID-19 was their hospitalization for a longer period of time versus children without gastrointestinal symptoms. Therefore, it is not clear whether the high prevalence of gastrointestinal symptoms of COVID-19 children is due to long-term hospital treatment or to a higher expression of ACE2 in the gastrointestinal tract or to different functions of ACE2 between children and adults (6). Undoubtedly, further studies involving children with COVID-19 are needed to confirm this hypothesis.
Unlike Fang et al.’s (7) study, the prevalence of gastrointestinal symptoms such as diarrhea and vomiting in children with COVID-19 did not differ much from the prevalence of these conditions in adults, as reported in a recent meta-analysis (2). This finding might be due to the greater number of pooled studies that included in their meta-analysis more digestive symptoms of COVID-19 in adults than in the present meta-analysis. Another difference in the incidence of digestive symptoms of COVID-19 among children and adults was the high prevalence of anorexia in adults. Anorexia was not reported in any of the studies included in our meta-analysis and this difference between children and adults with COVID-19 should warrant further research on the topic.
In addition to the influence of gastrointestinal involvement on COVID-19 prognosis, patients with COVID-19 are also at risk of liver function abnormalities (9). As our meta-analysis showed, a percentage of children and adolescents with COVID-19 experienced increased levels of liver enzymes, mainly ALT and AST, accompanied by a notable elevation of LDH levels especially in patients with severe forms of the disease.
Liver dysfunction was characterized by a slight increment in hepatocyte-related enzymes, including ALT, AST and LDH levels (34, 35). Total bilirubin was also reported to be slightly increased in a few patients (16, 29). At the moment, no mechanisms have been identified linking COVID-19 and liver damage. Most probably, the dysfunction of the liver is related to the viral infection of liver cells, to systemic inflammation and/or linked to drug toxicity (36).
Moreover, according to a recent report, liver biopsy specimens collected from a 50-year-old man with COVID-19 exhibited moderate microvesicular steatosis and mild lobular and portal activity (37, 38). In that study, it was assumed that the liver injury could have been caused by either SARS-CoV-2 infection or by drug toxicity. On the other hand, Fan et al. (38) reported no statistical differences between patients with normal liver function and abnormal liver function in terms of medications taken before hospitalization. Interestingly, patients with elevated liver enzymes spent more time in the hospital and received more lopinavir/ritonavir prescriptions after admission compared to patients with normal liver function. In this context, it is difficult to identify the clear cause of the elevation of liver enzymes in patients with COVID-19, and especially in children where the number of studies is limited (38).
4.1. Strength and Limitations
The primary strength of our study was that this meta-analysis provided a comprehensive overview of the prevalence of digestive symptoms and elevated liver enzymes among children and adolescents, far beyond the available evidence in 19 different studies. The evidence base available before our meta-analysis lacked a summative and consensual assessment, and thus a quantitative assessment was needed, which we have provided in this report. There are also a few limitations worth considering. We analyzed articles with different study designs, e.g. brief communications (19), or in some studies gastrointestinal symptoms of COVID-19 might have been under-reported. Thus, unfortunately, we were unable to extract sufficient information from these studies. We have also not been able to accurately extract information such as percent of patients with GI presentation who have respiratory symptoms or the mortality percentage in patient with GI from the articles, so it seems that these issues will be considered in future studies. Although our meta-analysis allowed for a large number of studies and participants to be analyzed, it is difficult to generalize our findings and to hypothesize the possible mechanisms involved in the development of gastrointestinal symptoms in COVID-19 in children.
5. Conclusion
The major finding of this review was the presence of gastrointestinal symptoms in 26% of the children and adolescents diagnosed with COVID-19. This review has provided a comprehensive overview of the prevalence of digestive symptoms of COVID-19 and elevated liver enzymes among children and adolescents, far beyond the evidence available until now from singular studies.
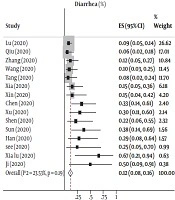
![This figure shows the pooled estimate of the prevalence of gastrointestinal symptoms [(A) diarrhea and (B) nausea/vomiting] in children and adolescents with COVID-19 This figure shows the pooled estimate of the prevalence of gastrointestinal symptoms [(A) diarrhea and (B) nausea/vomiting] in children and adolescents with COVID-19](https://services.brieflands.com/cdn/serve/3170b/22fd01d803e903a1691c0c0cadccb0a682c57a21/ijp-106935-i001-F1-preview.webp)