1. Introduction
Systemic lupus erythematosus (SLE) is a chronic systemic autoimmune disease, which affects many parts of the body. Based on the 2012 Systemic Lupus International Collaborating Clinics (SLICC) (1), at least four criteria with minimum of one clinical criterion and evidence of immunological activity are necessary to diagnose this condition. Childhood SLE (cSLE) is a rare disease in pediatrics but carries a high disease burden. This condition has been reported with a much higher prevalence in Asia than data from the western cohort; 3.3 - 8.8 per 100,000 children (2).
Lupus nephritis (LN), a subgroup of the disease, is confirmed by classical renal histology features on basic histopathology and immunofluorescence examination. This is a common involvement in childhood SLE, which can be the only solid organ involved at diagnosis or present later as the disease evolves. Similarly, in Asia, this is a significant risk factor for End-Stage Kidney Disease (ESKD) among children. Renal survival data suggested progression to chronic kidney disease stage 5 or ESKD could be as high as 15% in children with SLE and LN (3). Renal injury to the kidney manifests as proteinuria and hematuria. Often, renal biopsy is required for disease classification and prognostication. Although the histopathological findings in LN may vary, glomerular immune deposits described as the full-house staining pattern (4), reflect positivity for immunoglobulin G (IgG), IgA, IgM, complement (C3), and C1q, are pathognomonic.
Non-lupus full-house nephropathy (FHN) is a diagnostic challenge and needs further studies to be fully clarified. It is unclear whether this can be the initial manifestation of SLE, an isolated manifestation of SLE or a different entity that deserves recognition by itself, unrelated to SLE. Herein, we report two patients with FHN without the presence of immunological markers, i.e., antinuclear antibody (ANA) positivity, to suggest SLE.
2. Case Presentation
2.1. Case 1
A 9-year-old girl presented with a 5-day history of rashes over bilateral lower limbs associated with facial puffiness, tea-colored urine, and oliguria. On examination, she was normotensive. Her other systemic examinations were unremarkable apart from the non-raised, faint-pink erythema over the distal limbs. At admission, her serum urea was 27.2 mmol/L and her serum creatinine was 460 µmol/L, with a clinical impression of rapidly progressive glomerulonephritis. Other parameters were as follows: hemoglobin 12.5 g/dL, white blood cell 8.9/µL, platelet 227,000/µL, calcium 2.14mmol/L, phosphate 2.49 mmol/L, and serum albumin 28 g/L. Complement C3 was low 0.55 g/L (0.9 - 1.8 g/L) but C4 normal 0.42 g/L (0.1 - 0.4 g/L). Her erythrocyte sedimentation rate (ESR) was 75 mm/hr. Antinuclear antibody (ANA), anti-dsDNA, p-antinuclear antibody (p-ANCA), c-ANCA and anti-GBM antibody were all not detected. Other blood results included alanine transaminase (ALT) 6 IU/L, aspartate transaminase (AST) 26 IU/L, total bilirubin seven µmmol/L, fasting blood sugar (FBS) 5.2 mmol/L, total cholesterol 3.9 mmol/L, triglyceride (TG) 1.27 mmol/L, high-density lipoprotein (HDL) 1.47 mmol/L, low-density lipoprotein (LDL) 1.87 mmol/L and Antistreptolysin O Titer (ASOT) 400 IU/ml. Hepatitis B surface antigen and anti-HCV antibody were non-reactive. Urinalysis showed proteinuria (4+) and hematuria (5+). Kidney biopsy was done and histopathological examination showed diffuse global proliferative pattern of all 12 glomeruli, with podocyte hyperplasia observed in four glomeruli and one glomerulus with cellular crescent. Immunofluorescence microscopy revealed a full-house pattern with deposition of IgG (1+), IgA (2+), IgM (2+), C3 (1+), C1q (1+) along with glomerular capillary loop and mesangium. She received an intravenous methylprednisolone pulse before initiating high dose oral prednisolone (2 mg/kg), which was gradually tapered in addition to oral cyclophosphamide for ten weeks. The child required a week of hemodialysis support and kidney function gradually recovered and was normal after six weeks. Her ESR and C3 were within normal range as well by the 6th week. Proteinuria completely resolved by the 7th week, but microscopic hematuria only disappeared after 11 months from the onset. She remained well without resurgence in proteinuria. Her complement titers (C3 and C4) were within normal range too. Worth to mention that repeated ANA remained negative even after four years at the time of writing.
2.2. Case 2
A 3-year-old girl presented with sudden onset of bilateral ankle joint arthritis, bilateral lower limb swelling, multiple palpable purpuric rashes and kidney function impairment. She was initially treated as Henoch-Schonlein purpura with nephritis. However, after a few months, she presented again with gross hematuria, hypertension, and nephrotic range proteinuria. Unlike her previous admission, there was the absence of purpuric rash, joint swelling, or abdominal pain. Her parents did not notice any presence of oral ulcer, unexplained fever, hair loss, loss of appetite, or loss of weight. Investigations performed during the second presentation showed bicytopenia (hemoglobin nine g/dL, white blood cells
12.7/µL, platelet 96,000/µL), normal kidney function (sodium 140 mmol/L, potassium 4.3 mmol/L, urea 4.4 mmol/L, creatinine 38 µmol/L), and hypoalbuminemia (serum albumin 19 g/L). She had nephrotic range proteinuria (24-hour urine protein 3,825 mg/day) and microscopic hematuria. Her immunological screens revealed hypocomplementemia (C3 0.65 g/L (0.9 - 1.8), C4 0.08 g/L (0.1 - 0.4)). ANA was negative. Autoantibodies to extractable nuclear antigens were also undetected. Her cholesterol level was raised due to nephrosis (8.13 mmol/L) and her TG level of 2.65 mmol/L. Liver enzymes were within normal range. Histopathological examination of her renal biopsy showed diffuse mesangial proliferative with features of membranous pattern. Immunofluorescence studies revealed a full-house pattern of IgG (1+), IgA (2+), IgM (2+), C3 (1+), C1q (1+). Immunohistochemistry for C4d showed positive granular deposition along capillary wall. In light of the full-house pattern, clinical nephritis and hypocomplementemia despite the absence of ANA positivity, a diagnosis of immune-mediated glomerulonephritis, possible SLE was made. She received an intravenous methylprednisolone pulse and thereafter prednisolone, both of which were gradually tapered and kept at a minimum. She went into remission weeks after the corticosteroid therapy. She was not subjected to maintenance immunosuppression and her prednisolone was weaned off after four years. At the time of writing, her kidney function remained normal after nine years of follow-up with persistently negative ANA.
3. Discussion
We reported two cases of ANA negative full-house nephropathy. Both patients presented with nephrotic-nephritic features. Case 1 presented with non-specific rashes and rapidly progressive glomerulonephritis with only low C3 complement level and positive ASOT. Her immunofluorescence staining showed IgG (1+) and C3 (1+), but they were not strongly positive like cases seen with post-infectious glomerulonephritis. Instead, stronger IgA and IgM signals were observed. There were no subepithelial deposits that may be expected in post-infectious glomerulonephritis. Case 2 presented with clinical features similar to IgA vasculitis but with pan hypocomplementemia. Although IgA was more prominent on Immunofluorescent staining, the presence of an equally intense IgM and C4d staining on the capillary wall instead, were uncommon observations. In both cases, kidney histopathology examination showed active proliferative features with pan-immunoglobulin (IgG, IgM, and IgA), C3, and C1q deposition on immunofluorescent study; the latter better known as a full-house phenomenon. Interestingly, ANA was not present in both cases.
The hallmark of pathogenesis in SLE is detection of autoantibodies against nuclear antigens. By far, this remains the most sensitive and specific test for SLE, estimated to be 100% and 86%, respectively. In 2019, the European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) published their new criteria for SLE in which ANA is an entry criteria (5). The criteria require at least one positive ANA with a titer of 1:80. In comparison to previous classification, the 2019 recommendation cited better sensitivity and specificity than the previous 1997 American College of Rheumatology (6) as well as the Systemic Lupus International Collaborating Clinic (SLICC) classification criteria (1). Hence, with present knowledge, ANA positivity is necessary for diagnosis of SLE.
ANA could be measured using several laboratory techniques, including indirect immunofluorescent (IIF), enzyme immunoassay (EIA), and multiplex immunoassay (MIA) (7). Indirect immunofluorescent with human epithelial (HEp-2) cells have been regarded as the gold standard (8). HEp-2 cells are a substrate that offers a wide array of antigens, making it ideal for detection of ANA. Moreover, IIF is highly sensitive but limited by its specificity. This method is labor-intensive and subjective. The interpretation of patterns on the immunofluorescent requires experienced technicians who can read even the least common pattern. Many modern laboratories have moved toward automated testing EIA. The latter has been quoted to have fairly comparable sensitivity and specificity (94.6% and 86.0%, respectively). Worth to mention that EIA has a lower positive predictive value (9) 9, (36.4%) but much higher negative predictive value 99.5% compared to IIF method. Hence, negative results reflect true ANA-negative samples.
On the other hand, full-house phenomenon illustrated in kidney biopsy samples is invariably present in SLE patients with LN. Nevertheless, this is not exclusive to SLE. In the absence of ANA positivity, other conditions that may have full-house staining are IgA nephropathy, C1q nephropathy, infection-related glomerulonephritis, membranous nephropathy, membranoproliferative glomerulonephritis, and unclassified mesangial glomerulonephritis. In a large Italian cohort, involving 203 children and adolescents with LN, 53 children (26.1%) demonstrated only FHN at the onset. Out of which, 11 of the 53 children (20.8%) did not show ANA positivity at presentation and only developed the rest of SLE features on average after five years (10).
Full house nephropathy (FHN) has been reported in both children and adult cohorts. The cases were treated similarly to establish LN with or without maintenance therapy with variable kidney outcomes (Tables 1 and 2).
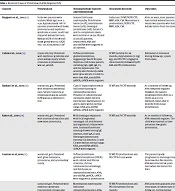
| Citation | Presentation | Histopathology Features and Immunology | Treatment Received | Outcomes |
|---|---|---|---|---|
| Ruggiero et al. (11) | Forty-two patients were studied. Mean age: 9.64 ± 4.28. Approximately 79% of patients presented with heavy proteinuria and nephrotic syndrome at onset. 30.9% had impaired kidney function. Median eGFR of the overall study group was 113.4 (IQR 78.8 - 127.4) mL/min/1.73 m2 | Isolated full-house nephropathy. Proliferative lesion (54.7%), membranous lesion (26%), mesangial proliferative (19%). Serum C3 and C4 complement levels were normal in 28 (66.7%) and 25 (59.5%) patients, respectively. ANA and anti-dsDNA were negative in all patients. | Induction: IV MP, IV/PO CYP, MMF, AZA, CSA. Maintenance: prednisolone, IV/PO CYP, MMF, AZA, CSA. | After 20 years, most patients had normal kidney function except two patients who had kidney failure from the onset. |
| Caltik et al. (12) | 13-year-old, boy. Presented with nephrotic syndrome and acute kidney injury. Serum creatinine at presentation was 145 µmol/L. | Diffuse proliferative glomerulonephritis, suggesting Class IV B Lupus Nephritis. Full-house pattern, including IgG, IgM, IgA, C3, and C1q depositions. The activity and chronicity index were 12/24 and 1/12. C3 and C4 were low. ANA, antidsDNA pANCA, cANCA were negative. | IV MP monthly for six months, dipyridamole (5 mg/ kg), and PO CYP (2 mg/kg for three months) followed by PO AZA and PO prednisolone. | Remained in remission during follow-up, 3 years from onset. |
| Baskin et al. (13) | 10-year-old, girl. Presented with recurrent abdominal pain. Serum creatinine at presentation was 88 µmol/L (GFR was 60.9 ml/min/1.73 m2) | Class III lupus nephritis with full-house staining on immunofluorescence. Presence of tubuloreticular inclusions under electron microscopic examinations. C3 and C4 were within normal range. ANA, anti-dsDNA pANCA, cANCA were negative. | IV MP and IV CYP monthly | At 12 months of follow-up, ANA remained negative. However, the patient progressed from CKD3 to 4 (from 60.9 to 17.9 ml/min/1.73m2) after 17 months from initial presentation. |
| Kim et al. (14) | 16-year-old, girl. Presented with interstitial vasculitis and new-onset proteinuria. | Mild mesangial expansion with focal segmental mesangial cell proliferation and segmental collapse were seen. Immunofluorescent staining showed strong IgG deposits, mild IgA, C3 and fibrinogen deposits and minimal C1q deposits. C3 and C4 were within normal range. ANA, antidsDNA pANCA, cANCA were negative. | IV MP and IV CYP monthly | At 24 months of follow-up, ANA remained negative. The child was reported to have persistent low-grade proteinuria. |
| Gianviti et al. (15) | 9-year-old, girl. Presented with gross hematuria, proteinuria, and acute kidney injury. | Diffuse proliferative glomerulonephritis (DPGN) with cellular and fibrous crescents, chronic tubulointerstitial damage, and full-house on immunofluorescence. ANA, anti-dsDNA, pANCA, cANCA were negative at presentation | IV MP, PO prednisolone and PO CYP for nine weeks | The patient rapidly progressed to end-stage renal failure within five months. ANA was detected six years later when she had an SLE flare. |
| 8-year-old girl. Presented with nephrotic syndrome. Concurrently, she was also diagnosed with Fisher-Evans syndrome because of autoimmune thrombocytopenia and Coombs-positive hemolytic anemia. | Membranous nephropathy full-house immunofluorescence. ANA, anti-dsDNA, pANCA, cANCA were all negative at presentation. | IV MP, PO prednisolone and PO chlorambucil for six months | ANA positivity appeared after three years. At seven years of follow-up, normal kidney function | |
| 10-year-old girl. Presented with gross hematuria and moderate proteinuria. Her initial serum creatinine was 80umol/Ll. | Kidney histopathology examination showed focal glomerulonephritis (FGN). Electron microscopy showed tubuloreticular inclusions in endothelial cells. ANA, anti-dsDNA, pANCA, cANCA were negative. | PO prednisolone and PO CYP for two months | ANA positivity appeared 10 years after the onset. | |
| Bharati et al. (16) | 15-year-old boy. Presented with fever for three weeks, progressive swelling of the body for two weeks and reduced urine output for one week. No history of joint pain, oral ulcer, photo- sensitivity, malar rash, or alopecia. | C3 and C4 were both low. ANA was negative. Biopsy was suggestive of proliferative glomerulonephritis with cellular crescents and some interstitial fibrosis tubular atrophy. IgG (3+), IgA (2+), kappa (3+), lambda (3+), C3 (2+) and C1q (2+) | PO prednisolone at a dose of 1 mg/kg/day. | ANA positivity after four weeks from onset. |
| Arslansoyu Camlar et al. (17) | 7-year-old girl. with streptococcal throat infection who developed palpable rash, acute nephritis and carditis. | C3 hypocomplementemia. Kidney biopsy demonstrated endocapillary and extracapillary proliferative glomerulonephritis with crescents. IgA (2+), IgG (3+), IgM (2+), C3c (1+), C1q (2+) ANA, anti-dsDNA and ANCA were negative. | IV MP followed by PO prednisolone. | - |
Reported Cases of Children with ANA Negative FHN
| Citation | Presentation | Histopathology Features and Immunology | Treatment Received | Outcomes |
|---|---|---|---|---|
| Wen et al. (18) | 94 patients with FHN were evaluated. 24 patients with no serological and clinical evidence of SLE. | The clinicopathologic diagnoses included membranous glomerulonephritis (GN) (46%), IgA nephropathy (21%), membrano- proliferative glomerulonephritis (12.5%), post-infectious glomerulonephritis (12.5%), C1q nephropathy (4%), and unclassified mesangial glomerulonephritis (4%). | Treatment received varies as it depends on the clinical presentation and the histology diagnosis. | One patient developed anti-DNA antibody eight months and another one developed hypocomplementemia 10 months after renal biopsy. These two patients developed clinical symptoms of lupus subsequently. The other 22 patients remained well. |
| Bitencourt Dias et al. (19) | 20 patients (mean age of 40 years old) presented with full house immunofluorescence staining in renal biopsies samples with initial negative ANA were evaluated. | Membranoproliferative glomerulonephritis (45%), membranous nephropathy (30%), mesangial glomerulonephritis (10%), acute diffuse glomerulonephritis (5%), cryoglobulinemic vasculitis (5%), crescentic glomerulonephritis (5%). | Fourteen patients received immunosuppressive therapy. | A-fifth of the cohort (four out of 20) developed SLE with ANA positivity after 64.32 ± 55.26 months. |
| Wani et al. (20) | 6,244 kidney biopsies specimens were evaluated. 498 kidney biopsies were identified with full house immuno- fluorescence. 81 of the 498 (16.2%) patients had no clinical or serological evidence of SLE at the time of renal biopsy. | The major diagnoses were membranous nephropathy (25.9%), Ig A Nephropathy (22.2%), membrano- proliferative glomerulonephritis (14.8%), diffuse proliferative glomerulonephritis (12.3%), crescentic glomerulonephritis (12.3%), amyloidosis (8.6%), C1q nephropathy (3.7%). These observations concluded that FHN is not limited to lupus. | Not included in the paper. | Not included in the paper. |
| Rijnink et al. (21) | 149 patients were included in the study. 32 had non-lupus full house nephropathy. 20 non-lupus FHN patients had no other features and were deemed idiopathic non-lupus FHN. 12 patients were thought to have secondary non-lupus FHN due to various reasons: positive Anti PLA2R membranous nephropathy, oncology-related membranous nephropathy, Ig A nephropathy, infection-related glomerulonephritis and anti-neutrophil cytoplasmic antibody-associated glomerulonephritis. | The histopathological lesions described as no lesions (10%), focal segmental glomerulosclerosis (5%), mesangioproliferative (5%), focal proliferative (25%), segmental chronic lesion (10%), diffuse proliferative (10%), active crescentic (10%), membranoproliferative (5%). | 50% of the non-lupus FHN received immunosuppressive therapy. | None developed lupus even after 20 years of follow-up. Overall, ESRD developed in about half of the cohort (12 out of 20 in the idiopathic group and 6 out of 12 in the secondary non-lupus FHN group). Renal and overall survival were not significantly different for the non-lupus FHN and lupus FHN groups. Their findings suggest the heterogeneity of the non-lupus FHN cohort with a relatively poorer outcome. |
Reported Cases of Adults with ANA Negative FHN
Data from literature suggests that FHN may not be exclusive to lupus. In general, close to 20% may not have serological evidence to support the underlying pathophysiology of SLE. Both of our patients showed FHN on renal biopsy. The ANA tests were carried out using the EIA method and these were repeated several times over a period of a few years and they remained negative till the time of writing. A deeper focus on the immunofluorescent pattern was not conforming to the clinical presentation. Such observations suggest that these cases with ANA negative FHN should be treated as a different entity and much is not readily known. Both of them received immunosuppressants at the presentation. They were not subjected to maintenance immunosuppression and their glucocorticoid therapy was weaned off gradually as well. Both demonstrated good long-term kidney outcomes.
In conclusion, ANA negative FHN could be immune-mediated glomerulonephritis, which may or may not be part of SLE. The clinical course is unpredictable despite having similar renal histology. More importantly, it seems that aggressive initial induction therapy is beneficial. Long-term maintenance with immunosuppressants may not be required. These children may benefit from surveillance, but much is needed to be studied regarding the optimal period of observation. The uncertainties in the long-term outcome can leave patients and their family members in perpetual mental stress.