1. Introduction
Neonatal pneumoperitoneum denotes the occurrence of free gas within the abdominal cavity of newborns for various reasons, often manifesting as abdominal distension. Around 90% of neonatal pneumoperitoneum instances result from gastrointestinal perforation, necessitating immediate surgical intervention. However, a minority of these newborns exhibit no signs of perforation within the pneumoperitoneum, presenting no symptoms other than abdominal distension. Abdominal paracentesis in these cases reveals the presence of gas without fluid, indicating a rare condition known as neonatal benign pneumoperitoneum (NBPP), also referred to as idiopathic pneumoperitoneum, non-operative pneumoperitoneum, or unexplained pneumoperitoneum. Diagnosing NBPP requires caution; it is essential to carefully rule out pneumoperitoneum resulting from perforation or peritonitis. Unlike the common approach leading to unnecessary laparotomies, conservative treatment is advised for newborns diagnosed with NBPP. This paper reports three instances of NBPP successfully managed at our hospital over the past decade and integrates these experiences with existing literature to offer clinical guidance for the accurate diagnosis and effective treatment of this condition.
2. Case Presentation
2.1. Case 1
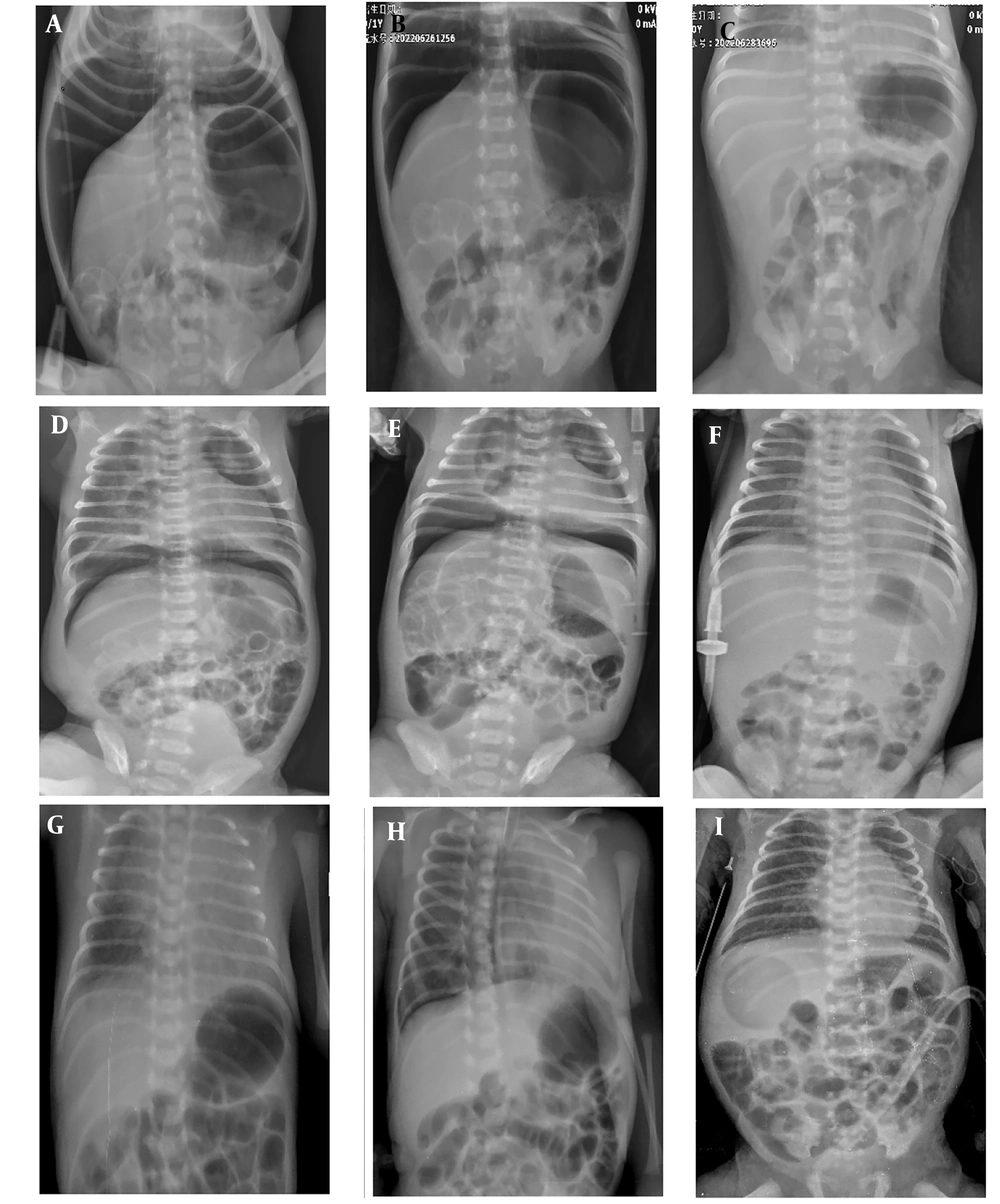
The patient was a male infant born at 33 + 6 weeks gestation, part of a twin pregnancy, transferred from an external hospital due to "abdominal distension" on his first day of life. The pregnancy was notable for a first-time mother with chronic hypertension and a twin gestation. The infant was delivered via cesarean section and required no resuscitation at birth beyond routine warming, drying, and tactile stimulation. His Apgar scores were 8 and 10 at one and five minutes, respectively, and his birth weight was 1750 g. Upon admission to the NICU, he was placed on nasal continuous positive airway pressure (NCPAP) for respiratory support. Shortly afterward, abdominal distension was observed. The infant had passed meconium post-delivery. An abdominal X-ray revealed free air under the diaphragm, prompting his transfer to our level 4 NICU for potential surgical intervention for "neonatal pneumoperitoneum." On physical examination upon admission, his abdomen was notably distended but soft, with no redness or tenderness of the abdominal wall. Laboratory tests showed a white blood cell count (WBC) of 6.47 × 109 / L and a C-reactive protein (CRP) level of 1.65mg/L (Table 1). The abdominal X-ray indicated a significant accumulation of free air (Figure 1A), and no signs of calcification. Abdominal paracentesis detected only air, without any fluid. Given the patient's stable clinical condition on minimal respiratory support, neonatal benign pneumoperitoneum (NBPP) was diagnosed. Management included nothing by mouth (NPO), gastrointestinal decompression, oxygen therapy via facemask (FiO2, 30%, 5L/min), and antibiotics. Subsequent abdominal X-rays showed a significant reduction in air below the diaphragm. Enteral feeding began on the third day of life and increased as tolerated. By the fifth day post-birth, the air under the diaphragm had completely resolved (Figure 1C). The patient achieved full enteral feeds by the tenth day and was subsequently discharged home without complications.
| No. | Age at onset(days) | Gender | Gestation(weeks) | Birth Weight (g) | Delivery | Apgar Score | Symptom | WBC (109/L) | CRP (mg/L) | Treatment | Survival |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | Male | 33+6 | 1750 | Cesarean | 8-10 | AD, T, vomiting | 6.47 | 1.65 | Conservative | Y |
| 2 | 2 | Male | 39+4 | 3250 | Vagina | 4-7-9 | AD, T, Cyanosis | 12.1 | 94 | Conservative | Y |
| 3 | 1 | Male | 30+5 | 1710 | Cesarean | 9-10 | AD, T | 24.7 | 18 | Laparotomy | Y |
The Detailed Demographic and Clinical Information of Three Patients
The radiographs of patients (case 1, ABC; case 2, DEF; and case 3, GHI). A, D, G: Radiographs revealed free gas under the diaphragm at hospital admission. B, E, H: Radiographs showed free gas under the diaphragm after 24 hours. C, F, I: Radiographs showed resolution of free gas in the abdomen after treatment
2.2. Case 2
The patient was a male infant born at 39 + 4 weeks gestation, transferred from an external hospital at 2 days old for the treatment of respiratory failure resulting from meconium aspiration syndrome and the presence of free air under the diaphragm. The infant was born vaginally to a mother with four pregnancies and two previous births, experiencing prolonged rupture of membranes, fetal distress, and thick meconium-stained amniotic fluid, with the umbilical cord wrapped around the neck. His birth weight was 3250g (Table 1). At birth, the infant was cyanotic, flaccid, and showed no respiratory effort. He was resuscitated in the delivery room with endotracheal intubation and positive pressure ventilation. The Apgar scores were 4 at 1 minute, 7 at 5 minutes, and 9 at 10 minutes. The admission chest X-ray indicated meconium aspiration syndrome. He was initially placed on a high-frequency oscillator at the local hospital, with settings of a mean airway pressure (MAP) of 25 cmH2O, a frequency of 10 Hz, FiO2 at 99%, and an amplitude of 55. Two days later, despite an improvement in his respiratory status, the patient developed sudden abdominal distension. An abdominal X-ray revealed a significant amount of free air under the diaphragm, leading to concerns about gastrointestinal perforation and his subsequent transfer to our hospital. Upon admission, his vital signs were stable on the ventilator, and the abdomen was significantly distended yet soft, without redness or tenderness. Echocardiography identified a patent ductus arteriosus and moderate pulmonary hypertension. Laboratory tests showed a white blood cell count (WBC) of 7.0 × 109/L and a CRP level of 92 mg/L. After performing abdominal paracentesis and observing a reduction in gas on the X-ray, no further surgical interventions were made. The patient received treatment including nothing by mouth (NPO), gastrointestinal decompression, continued endotracheal intubation, high-frequency oscillatory ventilation, and other supportive care such as antibiotics. By the 6th day of life, his respiratory support was significantly reduced, and a follow-up abdominal X-ray indicated the free air had substantially decreased. By the 7th day post-birth, the abdominal X-ray showed the free air had essentially resolved. The patient was discharged home without complications after a 15-day hospital stay.
2.3. Case 3
This case involves a male premature infant born at 30 + 5 weeks gestation via cesarean section due to placenta previa. His Apgar scores were 9 at 1 minute and 10 at 5 minutes, with a birth weight of 1710 g. Post-birth, the infant was placed on NCPAP to manage respiratory distress and exhibited abdominal distension on the first day of life. The abdomen was soft and non-tender, without any redness on the abdominal wall. Laboratory tests showed a white blood cell count (WBC) of 24.7 × 109/L and a C-reactive protein (CRP) level of 18 mg/L (Table 1). A chest-abdominal X-ray revealed free air under the right diaphragm. Given the clinical presentation, spontaneous intestinal perforation could not be ruled out, leading to an exploratory laparotomy. During the surgery, a small amount of clear fluid was found in the abdominal cavity, but no perforation was detected in the stomach or intestines. The infant was ultimately diagnosed with NBPP (1). By the 5th day post-birth, the free air under the diaphragm had been completely absorbed. The patient was eventually discharged home with a discharge weight of 2500 g.
3. Discussion
Neonatal benign pneumoperitoneum (NBPP) is an infrequent variant of neonatal pneumoperitoneum characterized by a soft abdominal wall and an absence of systemic symptoms (2). Radiologically, NBPP is identified by abdominal distension, with patients often displaying free air under the diaphragm or within the abdomen; abdominal paracentesis typically reveals only gas or a minimal amount of clear fluid, and exploratory laparotomy does not uncover any gastrointestinal perforation. The condition is mostly reported in premature infants and shows no preference for gender. In this report, we present 3 cases of NBPP, all of which exhibited abdominal distension, and most also presented with respiratory distress. All patients recovered and were successfully discharged. Further details are available in Table 2.
| Ref. | Age (days) | Gender | Gestation (w) | Birthweight (g) | Delivery | Symptom | Treatment | Survival |
|---|---|---|---|---|---|---|---|---|
| Gupta et al. (3), 2014 | 2 | F | 36 | 2100 | V | AD, RD | Conservative | Y |
| He et al. (4), 2015 | 8 | F | 37 | 2950 | V | AD, TP | Conservative | Y |
| Al-Lawama et al. (5), 2016 | 2 | M | 34 | 2280 | C | AD, TP | Laparotomy | Y |
| Abdelmohsen and Osman (6), 2017 | 5 | F | 34 | 1750 | V | AD, RD, Fever | Laparotomy | N (died of respiratory failure) |
| Duan et al. (1), 2017 | 6 | M | U | U | U | AD | Conservative | Y |
| 4 | M | U | U | U | AD, TP | Abdominal paracentesis | Y | |
| 25 | M | U | U | U | AD | Conservative | Y | |
| 19 | F | U | U | U | AD, TP | Abdominal paracentesis | Y | |
| 23 | M | U | U | U | AD, Fever, Diarrhoea | Conservative | Y | |
| 23 | M | U | U | U | AD | Conservative | Y | |
| Sammut et al. (7), 2018 | 13 | F | 27+4 | 420 | C | AD, anaemia, hypotension | Laparotomy | Y |
| Nakajima et al. (8), 2020 | 9 | F | 33+6 | 1972 | C | AD, pneumomediastinum, RD | Laparotomy | N (died of serious sepsis) |
| Huang and Zhang (9), 2020 | 1 | M | 35+6 | 2600 | C | AD, TP | Conservative | Y |
| Wang (10), 2021 | 3 | M | 29+1 | 1440 | C | AD, TP | Laparotomy | Y |
| Wang et al. (11), 2021 | 1 | U | 33+5 | 2190 | C | AD | Laparotomy | Y |
Some Cases of Neonatal Benign Pneumoperitoneum Ordered by Years
The exact causes and mechanisms of NBPP remain largely undefined, though adult cases of benign pneumoperitoneum have been associated with factors related to chest, abdominal, gynecological, and iatrogenic conditions (12-14). Neonatal pneumoperitoneum has been linked to various medical issues, such as necrotizing enterocolitis (NEC), isolated gastrointestinal perforation, anorectal malformations, meconium intestinal obstruction, and congenital band obstruction, among others (2). Many clinicians and researchers propose that mechanical ventilation and pulmonary air leaks are prerequisites for NBPP. Nakajima (8) described a case of NBPP accompanying air leak syndrome in a low-birth-weight infant, where abdominal X-rays revealed gas under the diaphragm 11 days post-birth, and laparotomy confirmed NBPP. Similar instances involving pneumothorax and mechanical ventilation have also been documented (3, 9, 10). It is speculated that following pulmonary lesions or mechanical ventilation that causes alveolar rupture, gas might migrate into the pulmonary interstitium and mediastinum, then along the vascular or lymphatic sheaths, ultimately entering the abdominal cavity through the diaphragmatic hiatus or retroperitoneal space, thereby causing pneumoperitoneum. The use of surfactants in infants with respiratory distress syndrome has been reported to potentially induce pneumoperitoneum (8).
Wang et al. described a case of NBPP in a premature infant who had not experienced pneumothorax, respiratory distress syndrome, or mechanical ventilation prior to exploratory laparotomy (11). This suggests an alternative mechanism to pulmonary air leakage. It is proposed that newborns can ingest air when crying after birth, with the gas then entering the abdominal cavity through the fragile gastric mucosa. This process is facilitated by high pressure from thick meconium, leading to gas accumulation beneath the diaphragm. He et al. (4) reported a similar case in 2015, hypothesizing that pneumoperitoneum resulted from a perforation (ranging from 2 to 4 mm in diameter) on the anterior wall of the stomach due to anoxia and ischemia. As the infant cried, ingesting a significant amount of air, gastric peristalsis pushed the air through the perforation into the abdominal cavity. Given that the infant was lying supine for an extended period, the risk of gastric juice causing peritonitis through the perforation was relatively low. Additionally, the self-sealing nature of the gastric leak meant that the intraperitoneal free gas could be gradually absorbed and eliminated. Furthermore, prolonged corticosteroid use may lead to degeneration of mucosa-associated lymphoid tissue and thinning of the intestinal wall, thus increasing the risk of perforation and contributing to the development of pneumoperitoneum (15).
The diagnosis of NBPP relies on medical history, physical examination, radiological evaluation, and abdominal paracentesis. The chosen site for abdominal paracentesis is typically at McBurney's point or the contralateral McBurney’s point and occasionally beneath the xiphoid process (16). The needle is inserted diagonally into the abdominal cavity. Indications for paracentesis are: (1) Respiratory or fluid restriction due to pneumoperitoneum necessitating paracentesis for symptom relief; (2) suspected gastrointestinal perforation, peritonitis, NEC, etc., requiring paracentesis for diagnostic purposes; (3) the need for intraperitoneal administration in the treatment of infections, tuberculosis, tumors, and other conditions within the peritoneal cavity (17). The risk associated with abdominal paracentesis is minimal, given the lack of vital organs at the insertion site, the needle's diagonal trajectory, and the intestines' mobility, which reduces the risk of injury upon exposure to external forces. Typically, gastrointestinal radiography aids in diagnosis (8, 18). All three cases in our report presented with abdominal distension, with X-rays revealing varying degrees of free air under the diaphragm. Specifically, Case 3 was initially suspected of neonatal gastrointestinal perforation due to elevated CRP and WBC levels, leading to a laparotomy that revealed no perforation and only a small amount of clear fluid. A subsequent diagnosis of NBPP was made. The pneumoperitoneum in the other two cases appeared to resolve on its own with close monitoring and supportive care. Diagnostic criteria for NBPP include: (1) The infant's generally good condition, (2) abdominal X-rays indicating pneumoperitoneum, (3) the absence of peritoneal irritation signs, (4) the presence of only gas or a minimal amount of liquid during abdominal paracentesis, and (5) reduction or disappearance of intraperitoneal free air following supportive treatment (1). Differential diagnoses for NBPP should consider hollow organ perforation, subphrenic abscess, extraperitoneal gas, mediastinal emphysema, and intestinal cystic emphysema, among others (1, 5).
Treatment of NBPP is generally straightforward. Conservative and supportive measures, such as withholding enteral feeding, gastrointestinal decompression, administering antibiotics, and replacing fluids intravenously, have proven to be sufficiently effective. Alongside these conservative treatments, it is crucial to closely monitor the newborns' abdominal and vital signs and to conduct follow-up abdominal X-rays. Should the abdominal distension worsen, as indicated by the appearance of abdominal wall redness or compromised intestinal hemodynamics, abdominal paracentesis may be considered to alleviate the condition (1). Some experts also suggest that immediate fasting and abdominal paracentesis should be undertaken upon the appearance of pneumoperitoneum signs in neonates (19). Based on our experience, we recommend initiating fasting early in the disease's course until other conditions can be ruled out through differential diagnosis. Given the self-limiting nature of this condition, abdominal paracentesis should be reserved for cases presenting with severe abdominal distension. Although severe postoperative complications are rare in newborns with NBPP, Al-Lawama et al. (5) documented a case experiencing metabolic acidosis, hypotension, and a decrease in hemoglobin following exploratory laparotomy. Ultrasound findings included grade 3 intraventricular hemorrhage, and the neonate developed a significant ductus arteriosus. All patients treated conservatively, as summarized in Table 2, survived. Caution is advised when contemplating surgery for neonates with pneumoperitoneum that has not been definitively diagnosed.
3.1. Conclusions
In summary, neonatal patients presenting with unclassified pneumoperitoneum should be approached with caution, with NBPP considered a possible diagnosis. Initial management should prioritize close monitoring and supportive care for cases of non-severe NBPP. Further investigation into the sources of gas, etiology, and pathogenesis of NBPP is warranted.