1. Background
Respiratory distress syndrome (RDS) is the most common respiratory disease in premature neonates with low birth weight (1). Congenital surfactant deficiency is among the etiologies, as well. Surfactant insufficiency leads to alveolar collapse and multiple micro-atelectasis. To provide sufficient Functional Residual Capacity (FRC), surfactant administration and mechanical ventilation are utilized. However, tracheal intubation and long term mechanical ventilation increase the risk of pneumothorax, respiratory infections and trauma (e.g. barotrauma, volutrauma) (2). Despite brilliant improvements in treatment of newborns with RDS, controversies exist in defining the most effective method and investigations with the lowest complications are still necessary (3). The most recent guideline published in 2013 suggests using nasal ventilation as the first step, even before surfactant administration (3).
On the other hand, NCPAP is approved as a method of ventilation support in neonates with RDS (4, 5). Although it reduced the need for intubation in premature neonates (6, 7), it is failure rate is reported as high as 15-50% (8, 9). Thus, using a non-invasive method with lower failure rates seems reasonable. NIPPV increases minute ventilation and tidal volume in comparison to NCPAP (10). Moreover, extubation failure rate was lower, as well (11, 12). NIPPV is able to use PIP in addition to appropriate PEEP to utilize more alveoli, which is very valuable (13). Besides, PEEP can be set lower in NIPPV.
NCPAP and NIPPV are both continued distending pressure (CDP) methods. They improve diaphragm function, increase pulmonary compliance and reduce upper and lower airways resistance. What’s more, tidal volume and compliance increase, whereas alveolar edema declines.
2. Objectives
Herein, complications and benefits of these two non-invasive methods are compared.
3. Patients and Methods
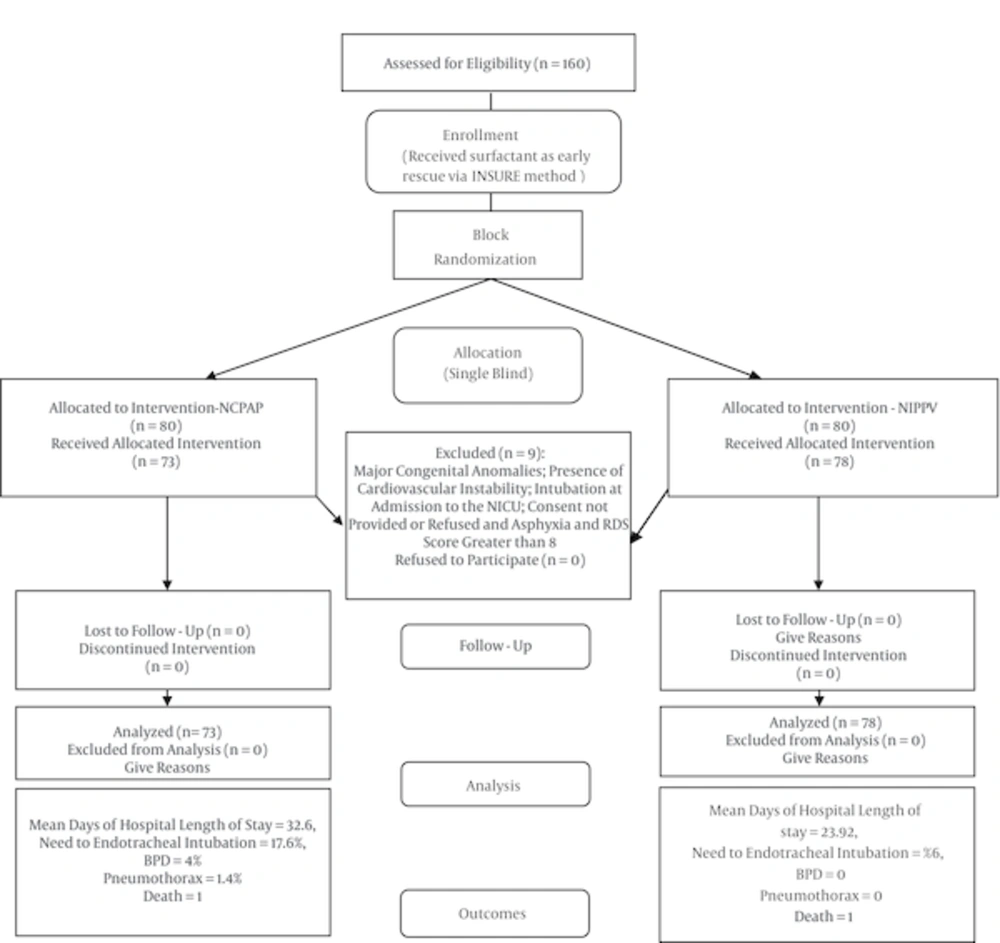
This study is a randomized clinical trial. Newborns with RDS and gestational age < 34 weeks admitted to NICU in Vali-Asr hospital, Tehran, Iran during 2012-2013 were enrolled in the study, consisting of 151 neonates with respiratory score (8) between 5-8. Informed consent was taken from the guardians. This study was approved by the institutional research ethics committee of Tehran University of Medical Sciences (Ethics code: 12046-91-04-89). Infants were excluded for any of the following reasons: major congenital anomalies, presence of cardiovascular instability and intubation at admission to the NICU, consent not provided or refused and asphyxia (Apgar ≤ 3, BE ≥ -12, multiple organ failure). Thirty-five of totally 151 newborns in this study received noninvasive ventilation as a primary mode (without surfactant therapy); 18 infants received NCPAP and 17 infants received NIPPV randomly. 13 (72%) infants of NIPPV group cured without surfactant therapy and only 4 (28%) infants needed surfactant therapy as INSURE, all of them again received NIPPV after INSURE and cured. In NCPAP group as primary mode 8 (44%) infants cured without surfactant therapy and 10 (56%) infants needed surfactant therapy.
In remaining infants surfactant was administered via INSURE method and then, they randomly (via block randomization) were divided into NCPAP (73 cases) and NIPPV (78 cases) group (Figure 1). Both groups received Survanta 100 mg/kg/dose with INSURE technique. Early (primary) complications including, re-intubation, pneumothorax, GI perforation, apnea, IVH and mortality rates were compared. Secondary outcomes compared were chronic lung disease (CLD) and length of hospital stay. To display the result graphically, Kaplan-Meier curves were plotted.
4. Results
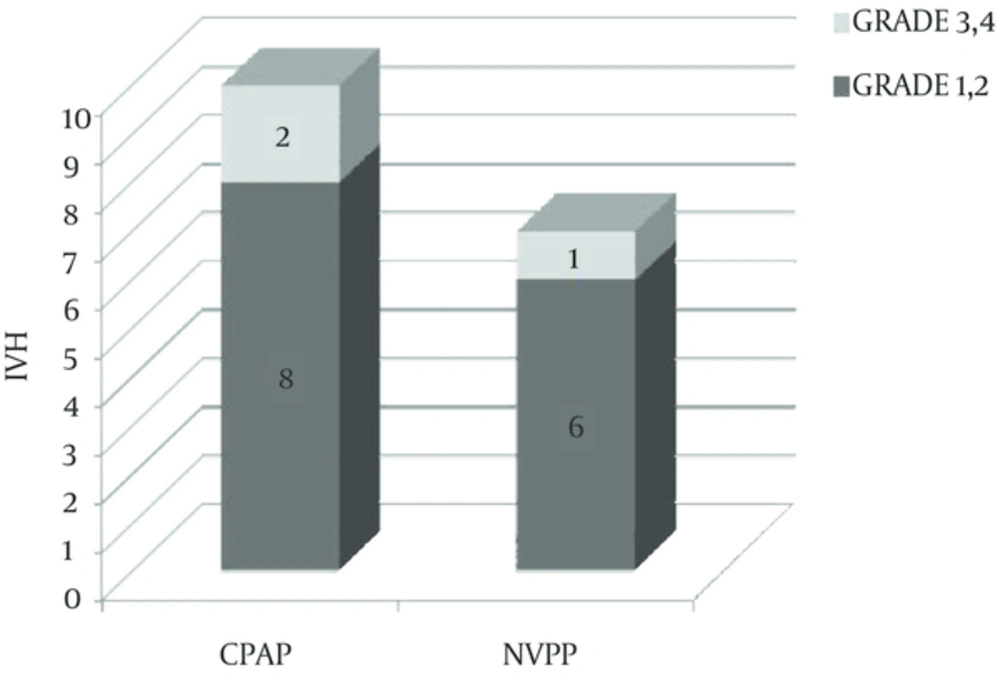
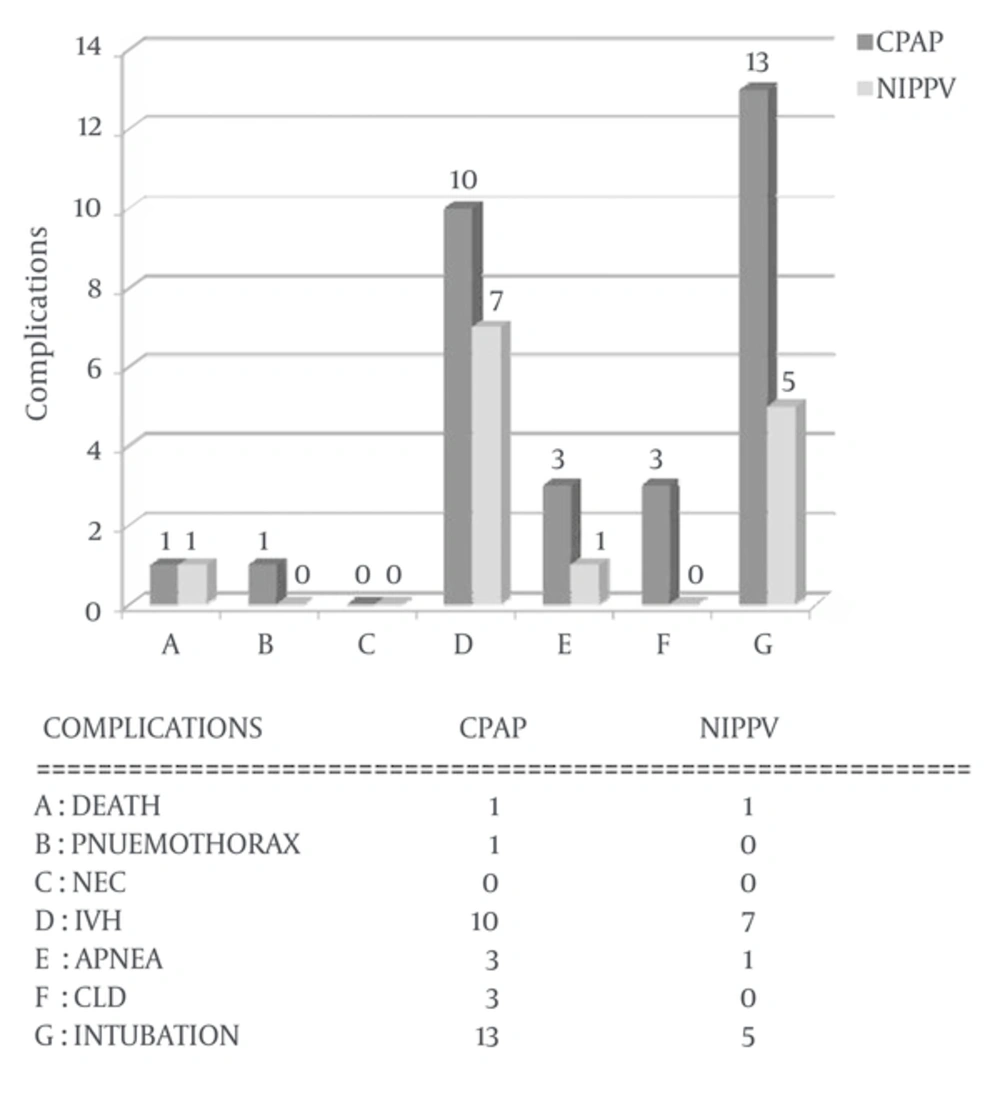
Demographic parameters did not differ significantly among NCPAP and NIPPV group (Table 1). The difference of pneumothorax, apnea, GI perforation and mean duration of ventilation support between the two groups was not statistically significant. Also the difference of intraventricular hemorrhage (IVH) was not statistically significant, IVH occurred less common and less severe in NIPPV group. IVH was detected in 10 (13.7%) neonates in NCPAP and 7 (9%) in NIPPV group, which was not statistically different. Eighty percent of IVH cases in NCPAP group were of grade 1-2 and twenty percent of grade 3-4. However, in NIPPV group 86% of IVH cases had grade 1-2 and 14% were in grade 3-4. The need for intratracheal intubation in first 72 hours was statistically significant between the two groups (17.6% in NCPAP vs. 6% in NIPPV, P = 0.031). The difference of CLD was statistically significant (4% in NCPAP group vs. no case in NIPPV group. P = 0.035).
| Variables | NCPAP (n = 73) | NIPPV (n = 77) | P Value |
|---|---|---|---|
| 0.371 | |||
| Male | 39 (53.5) | 36 (46) | |
| Female | 34 (46.5) | 42 (54) | |
| 1627 ± 539 | 1637 ± 631 | 0.916 | |
| 32.15 ± 2.03 | 32.04 ± 2.91 | 0.80 | |
| 36.98 ± 14 | 41.18 ± 6.5 | 0.055 | |
| 28.65 ± 5.91 | 29.01 ± 3.17 | 0.650 | |
| 6.86 ± 2.14 | 6.97 ± 1.84 | 0.733 | |
| 8.28 ± 1.12 | 8.07 ± 1.64 | 0.358 | |
| 7.98 ± 2.26 | 7.87 ± 2.50 | 0.020 | |
| 53 (73.3) | 64 (83.3) | 0.180 | |
| 51 (70) | 62 (81.7) | 0.130 |
a Values are expressed as No. (%) or mean ± SD.
Duration of hospitalization was 23.92 ± 13.5 and 32.61 ± 21.07 days in NIPPV and NCPAP group, respectively. The difference was statistically significant (P = 0.003) (Figure 2 and Table 1). Re-intubation rate in the first 72 hours in NCPAP group was 13 (17.6%) occasions and in NIPPV group it was 5 (6%) occasions which was significantly higher in NCPAP group (OR: 1.67, CI:1.13, P = 0.003) (Table 2 and Figure 3).
| Complication | NCPAP | NIPPV | P Value |
|---|---|---|---|
| 1 (1.3) | 1 (1.4) | 0.947 | |
| 1 (1.4) | 0 | 0.316 | |
| 0 | 0 | NA | |
| 3 (4) | 1 (1.3) | 0.280 | |
| 10 (14) | 7 (9) | 0.389 | |
| 3 (4) | 0 | 0.035 | |
| 13 (18) | 5 (6.5) | 0.031 | |
| 32.61 ± 21.07 | 23.92 ± 13.5 | 0.003 |
a Values are expressed as No. (%) or mean ± SD.
5. Discussion
There are several common points in this study with previous studies; however, there are some differences which are discussed. Re-intubated cases in first 72 hours were 5 (6%) in NIPPV vs. 13 (17.6%) in NCPAP group, which is statistically significant. Regarding re-intubation complications, this study found NIPPV as a feasible method. Extubation failure was defined as hypercapnia (pCO2 > 60), need to a PEEP > 6-7, need to PIP > 20, pH < 7.2 and recurrent apnea. Tang et al. reported a lower incidence of endotracheal intubation in NIPPV group (14). Davis et al. (13) and Bahman-Bijari et al. (12) found similar results. NIPPV reduces the need for high PEEP and provides higher functional residual capacity (FRC). More alveoli are utilized in this method because of using higher peak inspiratory pressure (PIP). Besides, respiratory muscles’ work decreases.
This study did not detect significant difference in pneumothorax incidence between the two groups. The same is reported by Meneses et al. (15). However, this parameter was not investigated in some other studies (12, 13). Using the least possible PEEP in NCPAP group may explain it. Sometimes in NCPAP mode, to reach O2 saturation > 90%, PEEP parameters higher than 6 cm H2O for newborns have to be used. These high PEEP parameters may cause pneumothorax. If we were allowed to use higher PEEP parameters in our study, we might have detected significant differences in pneumothorax incidence in both groups as evidenced in neonatal literature. Besides, using PIP in NIPPV group mandates lower PEEP to achieve sufficient ventilation. Here apnea rate did not differ significantly between the two groups (4% in NCPAP vs. 1.3% in NIPPV). Barrington et al. reported a lower rate of apnea in NIPPV group which was not statistically significant (10). After all, apnea has not been reported to be higher in newborns cared with NIPPV. CLD was 2.1 folds higher in NCPAP vs. NIPPV group in our study which was a significant (P = 0.035) difference. The diagnosis was made due to clinical, laboratory and radiologic findings (16). Similar results were reported by other studies (10-12). Tang et al. reported a lower rate of CLD in NIPPV vs. NCPAP group, as well (14).
Meneses et al. did not report a significant difference in CLD rate between the two groups (15). This may be due to different inclusion criteria. They had included in their study also neonates with low Apgar score at birth and those who received CPR and PPV. Consequently, these patients had developed pulmonary insults before receiving ventilation support. Therefore, CLD incidence was not different significantly between the two groups. Mean birth weight of their patients was 1100 gr in comparison to 1600 gr in this study. Therefore, some cases with low Apgar score and probable asphyxia had entered Meneses’ study, which might have affected the outcomes. For instance, BPD incidence was reported to be as high as 25% which is not reported elsewhere (15).
This study found a significant shorter hospitalization duration in NIPPV group (P = 0.003). Kaplan Mayer survival curve and regression test have confirmed this finding. This reveals NIPPV as a potential method to decrease expenses. Bahman-Bijari et al. reported a similar result (12). Meneses et al. reported no difference (15). Davis et al. did not find a significant difference. However, NIPPV was used in cases of NCPAP failure in this study, which was ignored in statistical analysis (13).
We found a higher incidence of IVH in NCPAP group, though not statistically significant. A randomized clinical trial performed by Bhandari et al. reported no difference between the two groups (17). We did not find a higher incidence of IVH in infants assisted with NIPPV in our cases.
This study detects no case of NEC or GI perforation in the two groups. Utilizing a F8 OG tube prevents abdominal distention. Besides, appropriate nursing care is crucial in careful measurement of the tube length and its fixation. Similar results were reported by Davis and coworkers (13), and Meneses et al. (15). Older studies have reported some cases of NEC and GI perforation, using NCPAP (18). However, Davis et al. and Lemyre et al. did not confirm this association (13, 19). Regarding all that, appropriate OG size is recommended in NCPAP use to prevent CPAP gastrointestinal complications.
Average time of ventilation support did not differ significantly between the two groups in our study. Meneses et al. published similar results (15). Nonetheless, Bahman-Bijari et al. (12) and Ramanathan et al. (20) reported significantly shorter duration of ventilation in NIPPV group. A probable explanation may be the difference in weaning criteria. Our criteria for weaning included: normal ABG, absence of respiratory distress, stable vital signs, no reduction in oxygen saturation, absence of apnea and spontaneous effective respiration by the neonate. Then, the infant was weaned and cared under oxyhood with 5 L/min flow rate. In the case of normal oxygen saturation, chest X-ray, vital signs and general condition, the oxyhood was removed. Weaning criteria in Bahman-Bijari et al. study were respiratory rate between 30-60, FiO2 < 30%, O2 sat > 90% and PEEP < 5 (12).
Traumatization of the skin around nose and mouth and irritation of nasal mucosa occurred in both groups. Yet, all of them recovered after weaning without any complication. Appropriate nursing care is fundamental in this field. Lower rates of barotraumas and air leak were found in this study.
Synchronization of NIPPV is focused in recent studies. This needs special skin sensors that are not available in Iran. Utilizing synchronized NIPPV (SNIPPV) revealed more successful results. A signal detector sensitive to abdominal motion is necessary for this method. It synchronizes ventilation support with infant’s thoraco-abdominal motions. What can be done instead is setting a ventilator setup similar to patient’s respiration efforts. However, Dumpa et al. reported no benefit for SNIPPV compared with NIPPV. No difference with PDA, IVH, BPD, ROP, PVL and mortality rates was significant. This confirms NIPPV as an effective method with least complications (21).
We found NIPPV to be more efficient compared with NCPAP in the management of RDS. Reduced length of hospital stay, lesser need to endotracheal intubation, and decreased BPD incidence are among the advantages which can totally reduce health care expenses (22, 23).