1. Background
2. Objectives
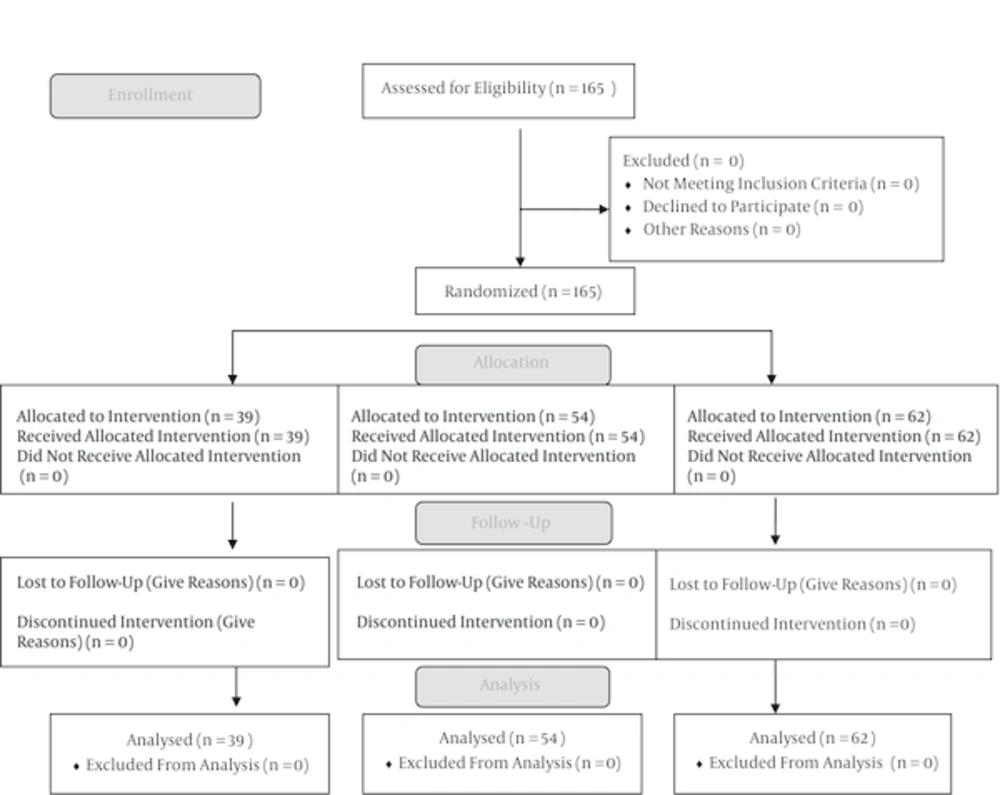
3. Methods
3.1. Study Populations
3.2. Study Design
3.3. Variable Definitions and Diagnostic Criteria
3.4. Statistical Analysis
4. Results
| Variable | Type of Surfactant | Total (%) | |||
|---|---|---|---|---|---|
| Curosurf No. (%) | Alveofact No. (%) | Survanta No. (%) | P Valuea | ||
| Sex | 0.07 | ||||
| Male | 37 (33.6) | 39 (35.5) | 34 (30.9) | 110 (67) | |
| Female | 25 (45.5) | 10 (18.2) | 20 (36.3) | 55 (33) | |
| Gestational age (weeks) | 0.8 | ||||
| ≤ 32 | 41 (39.4) | 30 (28.9) | 33 (31.7) | 104 (63) | |
| > 32 | 21 (34.4) | 19 (31.2) | 21 (34.4) | 61 (37) | |
| Birth weight (gr) | 0.3 | ||||
| < 1,500 | 28 (45.2) | 24 (38.7) | 10 (16.1) | 62 (42) | |
| 1,500 - 2,500 | 18 (36.7) | 22 (44.9) | 9 (18.4) | 49 (37) | |
| > 2,500 | 24 (44.4) | 15 (27.8) | 15 (27.8) | 54 (21) | |
| Sepsis | 0.5 | ||||
| Yes | 13 (30.2) | 15 (34.9) | 15 (34.9) | 43 (26) | |
| No | 49 (40.2) | 34 (27.9) | 39 (31.9) | 122 (74) | |
| Pneumothorax | 0.07 | ||||
| Yes | 2 (16.7) | 7 (58.3) | 3 (25) | 12 (7) | |
| No | 60 (39.2) | 42 (27.5) | 51 (33.3) | 153 (93) | |
| Mode of delivery | 0.5 | ||||
| Vaginal | 16 (31.4) | 16 (31.4) | 19 (37.2) | 51 (31) | |
| C/S | 46 (40.4) | 33 (28.9) | 35 (30.7) | 114 (69) | |
| Patent ductus arteriosus | 0.4 | ||||
| Yes | 8 (29.6) | 11 (40.7) | 8 (29.7) | 27 (16) | |
| No | 54 (39.1) | 38 (27.5) | 46 (33.4) | 138 (84) | |
| Ventilator therapy | 0.1 | ||||
| Yes | 19 (29.2) | 24 (36.9) | 22 (33.9) | 65 (39) | |
| No | 43 (43) | 25 (25) | 32 (32) | 100 (61) | |
| Retinopathy | 0.3 | ||||
| Yes | 1 (33.3) | 2 (66.7) | 0 | 3 (2) | |
| No | 61 (37.7) | 47 (29) | 54 (33.3) | 162 (98) | |
| Bronchopulmonary dysplasia | 0.9 | ||||
| Yes | 2 (50) | 1 (25) | 1 (25) | 4 (2) | |
| No | 60 (37.3) | 48 (29.8) | 53 (32.9) | 161 (98) | |
| Intracranial hemorrhage | 0.8 | ||||
| Yes | 7 (41.2) | 4 (23.5) | 6 (35.3) | 17 (10) | |
| No | 55 (37.2) | 45 (30.4) | 48 (32.4) | 148 (90) | |
| NCPAP support | 0.6 | ||||
| 0 | 42 (42) | 25 (25) | 33 (33) | 100 (80) | |
| Pulmonary hemorrhage | 0.06 | ||||
| Yes | 3 (18.8) | 9 (56.3) | 4 (24.9) | 16 (10) | |
| No | 59 (39.6) | 40 (26.9) | 50 (33.5) | 149 (90) | |
| Preterm rupture of membrane | 0.1 | ||||
| Yes | 3 (25) | 2 (16.7) | 7 (58.3) | 12 (7) | |
| No | 59 (38.6) | 47 (30.7) | 47 (30.7) | 153 (93) | |
| Preeclampsia | 0.3 | ||||
| Yes | 4 (21.1) | 7 (36.8) | 8 (42.1) | 19 (11) | |
| No | 58 (39.7) | 42 (28.8) | 46 (31.5) | 146 (89) | |
| IDM | 0.2 | ||||
| Yes | 0 | 1 (25) | 3 (75) | 4 (2) | |
| No | 62 (38.5) | 48 (29.8) | 51 (31.7) | 161 (98) | |
| Death | 0.5 | ||||
| Yes | 11 (30.6) | 13 (36.1) | 12 (33.3) | 36 (22) | |
| No | 51 (39.5) | 36 (27.9) | 42 (32.6) | 129 (78) | |
aDescriptive test (crosstabs).
| Variable | Type of Surfactant | |||
|---|---|---|---|---|
| Survanta | Alveofact | Curosurf | P Valuea | |
| Duration of hospital stay (mean ± SD) (days) | 15.37 ± 14.2 | 15.47 ± 12.5 | 14.35 ± 12.6 | 0.892 |
| Birth weight (mean ± SD) (gr) | 1829 ± 782 | 1815 ± 729 | 1873 ± 859 | 0.926 |
| Gestational age (mean ± SD) (weeks) | 31.56 ± 3.8 | 31.46 ± 3.6 | 31.70 ± 3.8 | 0.949 |
| Ventilation time (mean ± SD) (days) | 1.08 ± 1.9 | 1.89 ± 2.9 | 1.57 ± 3.7 | 0.765 |
| Number of surfactant injections (mean ± SD) | 1.08 ± 0.33 | 1.55 ± 1.12 | 1.06 ± 0.3 | 0.043 |
| Pneumothorax | 0.070 | |||
| Yes | 2 (16.7) | 7 (58.3) | 3 (25) | |
| No | 60 (39.2) | 42 (27.5) | 51 (33.3) | |
| Patent ductus arteriosus | 0.375 | |||
| Yes | 8 (29.6) | 11 (40.8) | 8 (29.6) | |
| No | 54 (39.1) | 38 (27.5) | 46 (33.4) | |
| BPD | 0.782 | |||
| Yes | 2 (50) | 1 (25) | 1 (25) | |
| No | 60 (37.3) | 48 (29.8) | 53 (32.9) | |
| Intracranial hemorrhage | 0.841 | |||
| Yes | 7 (41.2) | 4 (23.5) | 6 (35.3) | |
| No | 55 (37.2) | 45 (30.4) | 48 (32.4) | |
| NCPAP support | 0.542 | |||
| Yes | 51 (35.9) | 43 (30.3) | 48 (33.8) | |
| No | 11 (47.8) | 6 (26.1) | 6 (26.1) | |
| Pulmonary hemorrhage | 0.055 | |||
| Yes | 3 (18.6) | 9 (56.4) | 4 (25) | |
| No | 59 (39.6) | 40 (26.8) | 50 (33.6) | |
| Retinopathy | 0.298 | |||
| Yes | 1 (33.3) | 2 (66.7) | 0 (0) | |
| No | 61 (37.7) | 47 (29) | 54 (33.3) | |
aANOVA and crosstabs tests.
| Variable | Type of Surfactant | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Survanta No. (%) | Alveofact No. (%) | P Valuea | Curosurf No. (%) | Alveofact No. (%) | Curosurf No. (%) | P Valuea | Survanta No. (%) | P Valuea | |
| Pneumothorax | 0.03 | 0.1 | 0.5 | ||||||
| Yes | 2 (22.2) | 7 (77.8) | 3 (30) | 7 (70) | 3 (60) | 2 (40) | |||
| No | 60 (58.8) | 42 (41.2) | 51 (54.8) | 42 (45.2) | 51 (46) | 60 (54) | |||
| Patent ductus arteriosus | 0.2 | 0.3 | 0.8 | ||||||
| Yes | 8 (42.1) | 11 (57.9) | 8 (42.1) | 11 (57.9) | 8 (50) | 8 (50) | |||
| No | 54 (58.7) | 38 (41.3) | 46 (54.8) | 38 (45.2) | 46 (46) | 54 (54) | |||
| BPD | 0.6 | 0.9 | 0.6 | ||||||
| Yes | 2 (66.7) | 1 (33.3) | 1 (50) | 1 (50) | 1 (33.3) | 2 (66.7) | |||
| No | 60 (55.6) | 48 (44.4) | 53 (52.5) | 48 (47.5) | 53 (46.9) | 60 (53.1) | |||
| Intracranial hemorrhage | 0.8 | 0.6 | 0.9 | ||||||
| Yes | 7 (63.6) | 4 (36.4) | 6 (60) | 4 (40) | 6 (46.2) | 7 (53.8) | |||
| No | 55 (55) | 45 (45) | 48 (51.6) | 45 (48.4) | 48 (46.6) | 55 (53.4) | |||
| NCPAP support | 0.1 | 0.3 | 0.8 | ||||||
| Yes | 51 (54.3) | 43 (45.7) | 48 (52.8) | 43 (47.2) | 48 (48.5) | 51 (51.5) | |||
| No | 11 (64.7) | 6 (35.3) | 6 (50) | 6 (50) | 6 (35.3) | 11 (64.7) | |||
| Pulmonary hemorrhage | 0.03 | 0.09 | 0.5 | ||||||
| Yes | 3 (25) | 9 (75) | 4 (30.8) | 9 (69.2) | 4 (57.1) | 3 (42.9) | |||
| No | 59 (59.6) | 40 (40.4) | 50 (55.6) | 40 (44.4) | 50 (45.9) | 59 (54.1) | |||
| Retinopathy | 0.6 | 0.2 | 0.4 | ||||||
| Yes | 1 (33.3) | 2 (66.7) | 0 | 2 (100) | 0 (0) | 1 (100) | |||
| No | 61 (56.5) | 47 (43.5) | 54 (53.5) | 47 (46.5) | 54 (47) | 61 (53) | |||
aDescriptive test (crosstabs).
| Variable | Type of Surfactant ≤ 32 Weeks | Type of Surfactant > 32 Weeks | ||||||
|---|---|---|---|---|---|---|---|---|
| Survanta | Alveofact | Curosurf | P Value* | Survanta | Alveofact | Curosurf | P Valuea | |
| Duration of hospital stay (mean ± SD) (days) | 19.14 ± 15.9 | 17.43 ± 14.7 | 18.12 ± 14.8 | 0.892 | 8.0 ± 4.84 | 12.37 ± 6.97 | 8.43 ± 3.12 | 0.018 |
| Birth weight (mean ± SD) (gr) | 1411 ± 42 | 1385 ± 33 | 1319±37 | 0.571 | 2645±68 | 2496±67 | 2745±66 | 0.504 |
| Gestational age (mean ± SD) (weeks) | 29.4 ± 2.2 | 29.1 ± 1.9 | 29.2 ± 2.1 | 0.845 | 35.9 ± 2.3 | 34.2±1.0 | 35.7±1.7 | 0.567 |
| Ventilation time (mean ± SD) (days) | 1.37 ± 2.1 | 1.87 ± 3.3 | 1.82 ± 4.2 | 0.765 | 0.52 ± 1.3 | 1.95 ± 2.4 | 1.19 ± 2.7 | 0.019 |
| Pneumothorax | 0.218 | |||||||
| Yes | 2 (28.6) | 5 (71.4) | 0 | 0.026 | 0 | 2 (40) | 3 (60) | |
| No | 39 (40.2) | 25 (25.8) | 33 (34) | 21 (37.5) | 17 (30.4) | 18 (32.1) | ||
| Patent ductus arteriosus | 0.011 | |||||||
| Yes | 8 (42.1) | 5 (26.3) | 6 (31.6) | 0.954 | 0 | 6 (75) | 2 (25) | |
| No | 33 (38.8) | 25 (29.4) | 27 (31.8) | 21 (39.6) | 13 (24.5) | 19 (35.9) | ||
| BPD | - | |||||||
| Yes | 2 (50) | 1 (25) | 1 (25) | 0.905 | 0 | 0 | 0 | - |
| No | 39 (39) | 29 (29) | 32 (32) | 21 (34.4) | 19 (31.2) | 21 (34.4) | ||
| Intracranial hemorrhage | - | |||||||
| Yes | 7 (41.2) | 4 (23.5) | 6 (35.3) | 0.862 | 0 | 0 | 0 | - |
| No | 34 (39.1) | 26 (29.9) | 27 (31) | 21 (34.4) | 19 (31.2) | 21 (34.4) | ||
| NCPAP support | 0.817 | 0.607 | ||||||
| Yes | 34 (38.2) | 26 (29.2) | 29 (32.6) | 17 (40.5) | 9 (21.4) | 16 (38.1) | ||
| No | 7 (46.6) | 4 (26.7) | 4 (26.7) | 4 (50) | 2 (25) | 2 (25) | ||
| Pulmonary hemorrhage | 0.077 | 0.077 | 0 | 0.325 | ||||
| Yes | 3 (20) | 8 (53.3) | 4 (26.7) | 0 | 001 (100) | 0 | ||
| No | 38 (42.7) | 22 (24.7) | 29 (32.6) | 21 (34.4) | 19 (31.2) | 21 (34.4) | ||
| Retinopathy | 0.600 | 0.325 | ||||||
| Yes | 1 (50) | 1 (50) | 0 | 1 (100) | 0 | |||
| No | 40 (39.2) | 29 (28.4) | 33 (32.4) | 21 (34.4) | 19 (31.2) | 21 (34.4) | ||
aANOVA and crosstabs tests.
| Variable | Type of Surfactant (Pre-Surfactant) | Type of Surfactant (Post-Surfactant) | ||||||
|---|---|---|---|---|---|---|---|---|
| Survanta | Alveofact | Curosurf | P Valuea | Survanta | Alveofact | Curosurf | P Valuea | |
| pH (mean ± SD) | 7.38 ± 0.1 | 7.27 ± 0.25 | 7.26 ± 0.28 | 0.014 | 7.37 ± 0.39 | 7.34 ± 0.22 | 7.28 ± 0.23 | 0.219 |
| PCO2b (mean ± SD) | 37.7 ± 11.9 | 42.6 ± 12.4 | 44.2 ± 14.7 | 0.014 | 33.7 ± 11 | 39.2 ± 12.9 | 43.7 ± 13.8 | 0.003 |
| PO2c (mean ± SD) | 65.6 ± 50.9 | 63.3 ± 25.8 | 62.5 ± 33.4 | 0.197 | 65.5 ± 35.6 | 51.4 ± 20.1 | 63.7 ± 49.3 | 0.428 |
| BE (mean ± SD) | -3.4 ± 5.2 | -3.5 ± 5.3 | -1.5 ± 8.5 | 0.355 | -1.6 ± 5.3 | -1.99 ± 5.8 | 0.30 ± 8.0 | 0.330 |
aANOVA.
bPCO2, partial pressure of carbon dioxide in the blood.
cPO2, partial pressure of oxygen in the blood.