1. Background
Considerable microorganisms exist in human body, especially in the intestinal tract, with maximum numbers and types as well as complex compositions. It is well-documented that intestinal microorganisms are significantly associated with human diseases such as inflammation, obesity, diabetes mellitus and tumors (1, 2). The first 1000 days of human life play a key role in the colonization of intestinal flora which is affected by genetic factors, environment, age, delivery mode and feeding mode (3, 4). For instance, the intestinal flora of 6-month-old infants is evidently affected by feeding mode (5) that also exerts remarkable effects on growth and development (6, 7). As the first choice of newborns and infants, breast milk can provide necessary nutrients together with abundant signaling and immune factors of microorganisms to rapidly colonize them in the intestinal tract, facilitating the formation of innate immune system (8). The newborns and infants fed with breast milk are significantly less prone to common diseases (9). However, due to social, physiological and health factors, formula milk instead of breast milk has been widely used. Recently, the bioavailability and functions of goat milk have been extensively studied. It has become popular as a matrix for formula milk products, especially for the cases with allergy or intestinal diseases (10). Nevertheless, the effects of goat milk formula, cow milk formula and breast milk on the intestinal flora of newborns and infants have seldom been reported hitherto. On the other hand, their intestinal flora is also affected by delivery mode (11, 12). The composition and diversity of intestinal flora in newborns are predominantly influenced through maternal-neonatal flora exchange. In contrast, cesarean section destroys the maternal- neonatal flora exchange, then increasing the risks of abdominal diseases and obesity (13). Currently, the influence of delivery mode on the intestinal flora of newborns and infants with the same age and feeding mode remains elusive.
2. Objectives
Thereby motivated, we herein used high-throughput sequencing to assess the effects of different modes of delivery and feeding on the intestinal flora of newborns and infants with different ages, aiming to provide a clinical basis for food development and scientific feeding.
3. Methods
3.1. Reagents and Apparatus
Fecal genomic DNA extraction kit was purchased from Shanghai Shuangying Biotechnology Co., Ltd. (China). High-throughput sequencing platform was bought from Illumina (USA).
3.2. Collection of Fecal Samples
The fecal samples of 108 full-term newborns and infants aged 0 - 36 months delivered in obstetrics and pediatrics departments of The First Hospital of Lanzhou University were collected. This study has been approved by the Ethics Committee of our hospital, and written consent has been obtained from the guardians of all subjects. Sample size (n) = Z2×P(1-P)/E2, where Z is the statistical magnitude (confidence interval: 95%), which is 1.96 in this study; E is the error, which is 0.3; and P is the probability, which is 0.5. The sample size was then calculated as ≥ 11, so 12 cases were selected for each group. The demographic and baseline characteristics of all participants were obtained from their birth certificates.
The subjects all had normal body weights, without history of diarrhea or gastrointestinal diseases within 1 week or using any antibiotics. There were twelve 0 - 6-month-old cases of natural labor and breastfeeding (MV1), twelve 0 - 6-month-old cases of cesarean section and breastfeeding (MF1), twelve 0 - 6-month-old cases of natural labor and goat milk formula (YV1), twelve 6 - 12-month-old cases of natural labor and goat milk formula (YV2), twelve 12 - 36-month-old cases of natural labor and goat milk formula (YV3), twelve 6 - 12-month-old cases of cesarean section and goat milk formula (YF2), twelve 12 - 36-month-old cases of cesarean section and goat milk formula (YF3), twelve 6 - 12-month-old cases of natural labor and cow milk formula (NV2), and twelve 12 - 36-month-old cases of natural labor and cow milk formula (NV3). Phase I: 0 - 6 months; phase II: 6 - 12 months; phase III: 12 - 36 months. Internal feces samples were collected to prevent contamination by urine or other impurities, placed in sterile tubes and stored in a -80°C refrigerator prior to use.
3.3. DNA Extraction and Sequencing
Genomic DNA was extracted, and its purity and concentration were detected by SDS-PAGE. An appropriate amount of sample was added into a centrifuge tube and diluted to 1 ng/μL with sterile water. Using the diluted genomic DNA as template, the V3-V4 region of 16S rRNA gene was subjected to PCR. Primer sequences: Pr: 5’-ATTACCGCGGCTGCTGG-3’, Pf: 5’-CGCCCGCCGCGCGCGGCGGGCGGGGCGGGGGCACGGGGGG CCTACGGGAGGCAGCAG-3’. The expected length of amplification product was approximately 230 bp. The primers were synthesized by Life Technologies (Shanghai, China). After agarose gel electrophoresis, the purified PCR products were obtained by Gene JET gel extraction kit (USA). Library was constructed with Ion Plus Fragment Library Kit (48 rxns; Thermo Fisher, USA), quantified and detected by Qubit software, and sequenced on Ion S5TMXL system.
3.4. Bioinformatics Analysis
All clean reads were clustered into operational taxonomic units (OTUs) based on 97% similarity by using Uparse software (14). Afterwards, the representative sequences of OTUs were annotated, and cross-species annotation was performed with the SSUr RNA database (15) of SILVA (16) using the Mothur method, giving taxonomic information and community composition. Rapid multi-sequence alignment was conducted by MUSCLE software (17). Finally, the data were unified for subsequent alpha and beta diversity analyses. Alpha diversity analysis, which represents the abundance and diversity of flora, is described with Chao1, ACE, Observed species, Shannon and Simpson. Higher Chao1 and ACE indices suggest a higher flora abundance. Besides, higher Shannon index and Simpson index mean higher and lower flora diversities respectively.
3.5. Statistical Analysis
All data were analyzed by SPSS V. 16.0 software. The normally distributed categorical data were expressed as mean ± standard deviation, and intergroup comparisons were performed by the two independent-sample t-test. The data not conforming to normal distribution were represented as median and interquartile range, written as M (P25 ~ P75), and compared by using the nonparametric U test. Rates were compared by the χ2 test. P < 0.05 was considered statistically significant.
4. Results
4.1. Species Abundance and Diversity
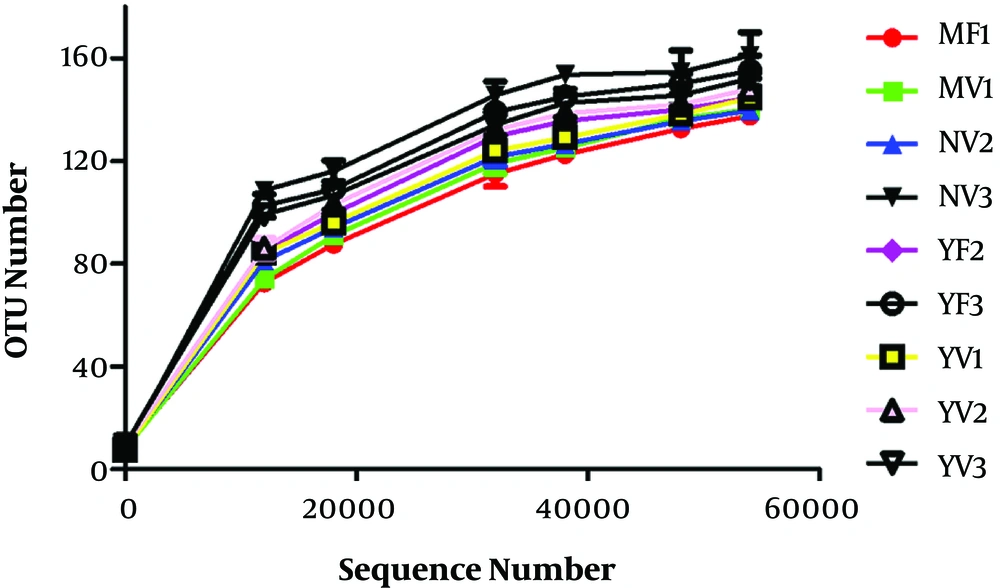
Figure 1 shows that as the number of sequencing increases, the curve tends to level off, indicating that the sequencing depth had substantially covered all species in each sample. When the amount of sequencing data was the same, the diversity of bacteria was higher in the infants of natural labor than those of cesarean section, except for Phase III infants, because cesarean section destroyed the mother-newborn flora exchange. The bacterial flora diversity of the goat milk formula group was higher than those of breastfeeding group and cow milk formula group. In the infants of natural labor fed with goat milk formula, the flora of Phase II infants had the highest diversity, followed by those of Phase III and I infants sequentially, probably being associated with the healthy development of infants. Phase II, as the key period for the mature development of infants, needs the most nutrients and heat energy required for growth, so the intestinal flora is most abundant and diverse in Phase II infants.
OTU number results of intestinal flora. OUT, operational taxonomic unit; MV1, 0 - 6-month-old, natural labor and breastfeeding; MF1, 0 - 6-month-old, cesarean section and breastfeeding; YV1, 0 - 6-month-old, natural labor and goat milk formula; YV2, 6 - 12-month-old, natural labor and goat milk formula; YV3, 12 - 36-month-old, natural labor and goat milk formula; YF2, 6 - 12-month-old, cesarean section and goat milk formula; YF3, 12 - 36-month-old, cesarean section and goat milk formula; NV2, 6 - 12-month-old, natural labor and cow milk formula; NV3, 12 - 36-month-old, natural labor and cow milk formula.
4.2. Effects of Different Modes of Delivery on Intestinal Flora of Cases with the Same Feeding Method and Age
The clean reads of all samples were clustered using Uparse software. By default, the sequences were clustered into OTUs with 97% consistency, and species annotations were performed for the representative sequences. In all samples (Table 1), there was no significant difference in the microbial abundance between the infants of Phase I breastfeeding group and Phase II goat milk formula group (P > 0.05). In the Phase III goat milk formula group, the microbial abundance varied greatly in different modes of delivery due to diverse food supplements for Phase III infants (18, 19). Meanwhile, Alpha diversity analysis also showed that there was no significant difference between natural labor and cesarean section in Phase I and II breastfeeding groups (P > 0.05), but the Chao1 values were significantly different in the Phase I breastfeeding group (P < 0.05). In the Phase III breastfeeding group, the degrees of bacterial flora in the infants with different delivery modes were significantly different (P < 0.05), being consistent with the results mentioned above.
| Group | OTU Number | ACE | Chao1 | Shannon | Simpson |
|---|---|---|---|---|---|
| Phase I natural labor + breastfeeding | 147 ± 7 | 147.67 ± 8.93 | 142.80 ± 6.30 | 2.73 ± 0.90 | 0.68 ± 0.17 |
| Phase I cesarean section + breastfeeding | 149 ± 8 | 152.18 ± 7.93 | 152.01 ± 6.77 | 2.85 ± 0.69 | 0.75 ± 0.18 |
| t | 0.652 | 1.308 | 3.450 | 0.367 | 0.979 |
| P valuea | 0.510 | 0.204 | 0.002 | 0.717 | 0.338 |
| Phase II natural labor + goat milk formula | 168 ± 26 | 165.08 ± 24.52 | 163.25 ± 22.14 | 3.48 ± 0.55 | 0.82 ± 0.06 |
| Phase II cesarean section + goat milk formula | 160 ± 21 | 162.47 ± 20.44 | 163.30 ± 23.29 | 3.35 ± 0.55 | 0.80 ± 0.07 |
| t | 0.829 | 0.283 | 0.005 | 0.579 | 0.751 |
| P valuea | 0.416 | 0.780 | 0.996 | 0.568 | 0.460 |
| Phase III natural labor + goat milk formula | 161 ± 17 | 159.06 ± 18 | 156.26 ± 16.63 | 3.66 ± 0.61 | 0.84 ± 0.11 |
| Phase III cesarean section + goat milk formula | 189 ± 16 | 191.25 ± 17.75 | 188.05 ± 18.56 | 4.18 ± 0.5 | 0.89 ± 0.04 |
| t | 4.155 | 4.411 | 4.419 | 2.284 | 1.480 |
| P valuea | 0.000 | 0.000 | 0.000 | 0.032 | 0.152 |
Abbreviations: OUT, operational taxonomic unit; phase I, 0 - 6 months; phase II, 6 - 12 months; phase III, 12 - 36 months.
aIndependent t-test.
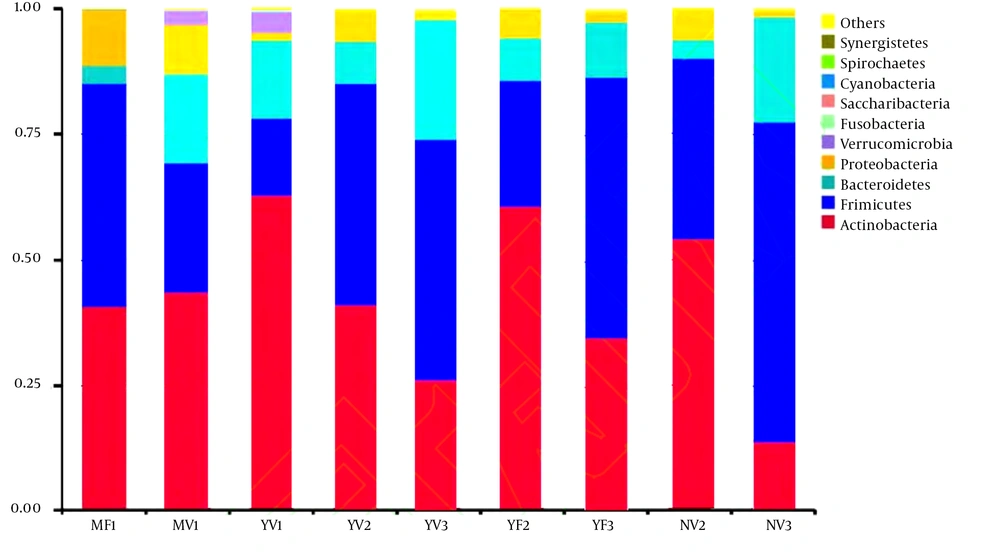
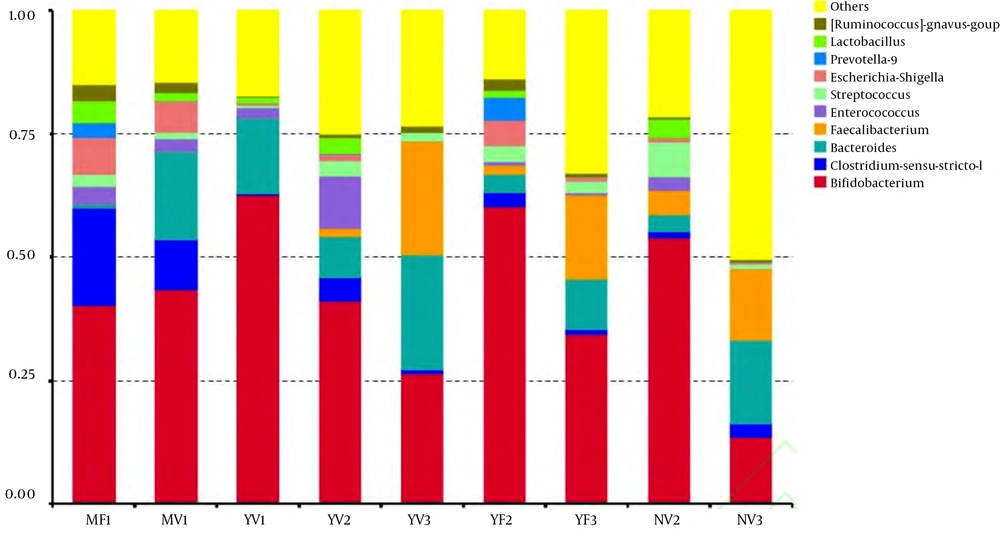
By sequencing the 16Sr RNA gene V3 - V4 region of all microorganisms in each sample, 10 major bacterial phyla were detected, but 99.09% of the sequences in all samples were mainly composed of 4 bacterial phyla, i.e. Actinobacteria, Firmicutes, Bacteroidetes and Proteobacteria. As exhibited in Figure 2, Actinobacteria and Firmicutes are dominant bacterial phyla of all groups. The relative abundances of Actinobacteria, Firmicutes, Bacteroidetes and Proteobacteria in the MV1 group were 43.74%, 25.48%, 17.93%, and 9.71%, respectively, and those of the MF1 group were 40.83%, 44.31%, 3.81%, and 11.01%, respectively. In the 0 - 6-month-old breastfeeding group, the dominant bacteria in the infants of natural labor and caesarean section had the same composition, but the abundance of Actinomycetes of the former was higher than that of the latter, and the abundance of Firmicutes of the latter was significantly higher than that of the former (P < 0.05). In the YV2 group, the relative abundances of Actinobacteria, Firmicutes, Bacteroidetes and Proteobacteria were 41.44%, 43.84%, 8.38%, and 6.27%, respectively. In the YF2 group, the relative abundances were 60.76%, 24.85%, 8.66%, and 5.70%, respectively. In the 6 - 12-month goat milk formula group, the abundance of Firmicutes in the infants of natural labor was higher than that in the infants of caesarean section (P < 0.05), but the abundance of Actinomycetes of the latter was higher than that of the former (P < 0.05). The relative abundances of Actinobacteria, Firmicutes, Bacteroidetes and Proteobacteria in the YV3 group were 26.58%, 47.73%, 24.07, and 1.58%, respectively. In the YF3 group, the relative abundances were 34.77%, 51.94%, 11.10%, and 2.16%, respectively. In the 12 - 36-month goat milk formula group, the abundances of Actinomycetes and Firmicutes in the infants of natural labor were lower than those in the infants of cesarean section (P < 0.05). In the 0 - 6-month-old breastfeeding group, Verrucomicrobia was detected in the natural labor group, but not in the cesarean section group. Although the four bacterial phyla were not significantly different in the infants with the same age and feeding mode (P > 0.05), there were some differences in the component proportions of dominant bacterial phyla due to different modes of delivery. In the 0 - 6-month-old breastfeeding group, the number of Actinobacteria in the natural labor group exceeded that in the cesarean section group (P < 0.05), but the number of Firmicutes was lower (P < 0.05), because cesarean section affected the balance of infantile intestinal flora, which destroyed the maternal-newborn flora exchange and caused the imbalance of intestinal flora of newborns when they were suddenly moved from the uterine environment to the external environment.
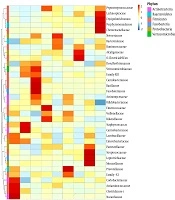
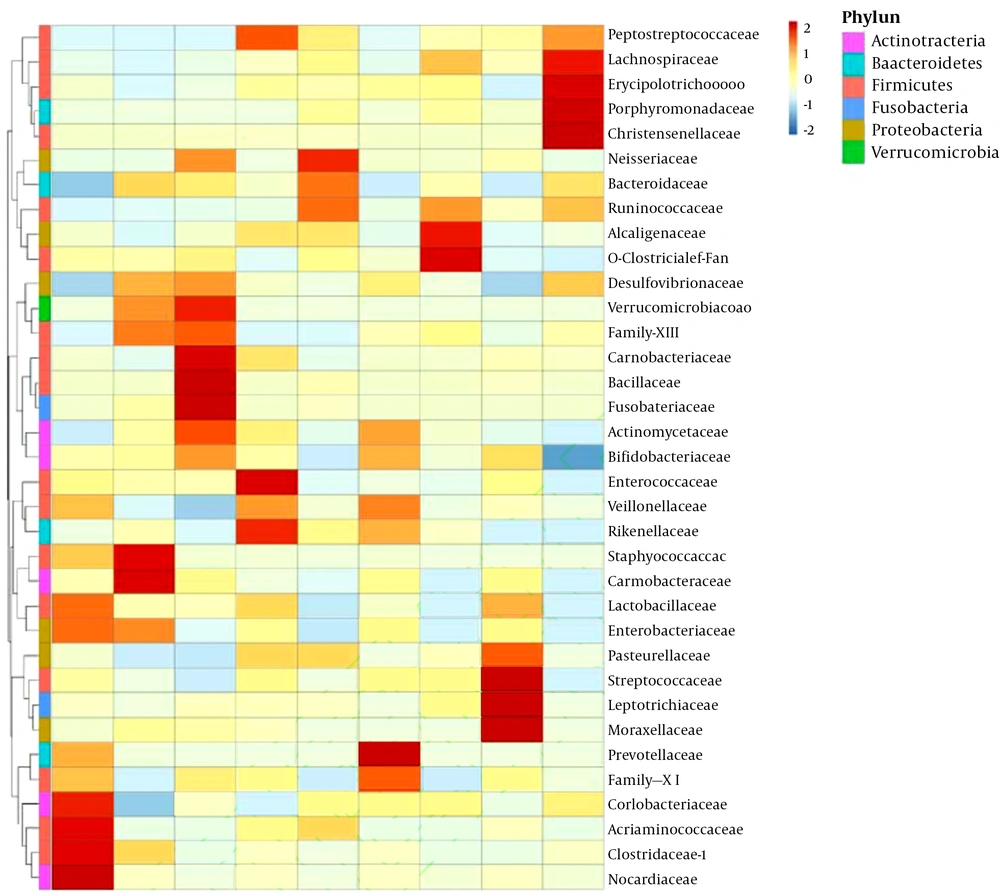
At the family level, in the 0 - 6-month-old breastfeeding group, Staphylococcaceae and Clostridiaceae were the dominant bacteria of natural labor group and cesarean section group, respectively (Figure 3). In the 6 - 8-month-old goat milk formula group, Enterobacteriaceae and Prevotellaceae were the dominant bacteria of natural labor group and cesarean section group, respectively. In the 12 - 36-month-old goat milk formula group, Bacteroidaceae and Ruminococcaceae were the dominant bacteria of the natural labor group, and Alcaligenaceae were dominant in the cesarean section group. In short, the mode of delivery barely affected the intestinal flora composition of the infants with the same age and feeding mode, but its effects on the abundance of dominant bacteria were significantly different.
Effect of different modes of delivery on intestinal flora of cases with the same feeding method and age. MV1, 0 - 6-month-old, natural labor and breastfeeding; MF1, 0 - 6-month-old, cesarean section and breastfeeding; YV1, 0 - 6-month-old, natural labor and goat milk formula; YV2, 6 - 12-month-old, natural labor and goat milk formula; YV3, 12 - 36-month-old, natural labor and goat milk formula; YF2, 6 - 12-month-old, cesarean section and goat milk formula; YF3, 12 - 36-month-old, cesarean section and goat milk formula; NV2, 6 - 12-month-old, natural labor and cow milk formula; NV3, 12 - 36-month-old, natural labor and cow milk formula.
4.3. Effects of Feeding Modes on Intestinal Flora of Cases with the Same Age and Delivery Mode
According to Table 1, the OTU number of the goat milk formula group exceeds that of the breastfeeding group in the Phase I natural labor group (P < 0.05), indicating a higher flora abundance. The ACE and Chao1 indices of the goat milk formula group were higher than those of the breastfeeding group (P < 0.05), also suggesting that the abundance in the goat milk formula group was significantly higher. Nevertheless, there was no difference in the Shannon index or Simpson index between the two groups (P > 0.05). In the Phase II natural labor group, the flora abundance of the goat milk formula group was slightly higher than that of the cow milk formula group (P > 0.05). In the Phase III natural labor group, the two feeding modes exerted significantly different effects (P < 0.05), because the 12 - 36-month-old infants were supplemented with a large amount of complementary foods besides milk formula.
With the same age and delivery mode as well as different feeding modes, over 99.09% of the sequences were composed of four phyla, i.e. Actinobacteria, Firmicutes, Bacteroidetes and Proteobacteria (Figure 2). However, their abundances were different. The relative abundances of Actinobacteria, Firmicutes, Bacteroidetes and Proteobacteria in the MV1 group were 43.74%, 25.48%, 17.93%, and 9.71%, respectively, those in the YV1 group were 62.98%, 15.23%, 15.45%, and 1.51%, respectively, those in the YV2 group were 41.44%, 43.84%, 8.38%, and 6.27%, respectively, those in the NV2 group were 54.20%, 35.71%, 3.91%, and 6.11%, respectively, those in the YV3 group were 26.58%, 47.73%, 24.07%, and 1.58%, respectively, and those in the NV3 group were 13.87%, 63.53%, 21.29%, and 1.29%, respectively.
Figure 4 shows that at the genus level, the intestinal bacteria of Phase I naturally born infants receiving breastfeeding and goat milk formula are mainly composed of Bifidobacterium, Clostridium-sensu-stricto-1 and Bacteroides. The abundance of Bifidobacterium in the infants fed with goat milk formula surpassed that of the breastfed infants (P < 0.05), while the abundances of Clostridium-sensu-stricto-1 and Bacteroides in the breastfed infants were slightly higher than those of the infants fed with goat milk formula (P < 0.05). In the Phase II naturally born infants, there was no significant difference in the composition of the genus-level flora between the infants fed with cow milk formula and goat milk formula (P > 0.05). Nevertheless, the abundances of Bacteroides and Enterococcus in the goat milk formula group were slightly higher than those of the cow milk formula group (P < 0.05). The abundances of Bifidobacterium and Streptococcus were slightly lower than those of the cow milk feeding group (P < 0.05). In the Phase III naturally born infants, the abundances of Bifidobacterium in the two groups of different feeding modes were significantly lower than those of Phase I and II infants (P < 0.05). The abundances of Bifidobacterium, Bacteroides and Prevotella-9 in the goat milk formula group were higher than those of the cow milk formula group (P < 0.05). For the infants with the same age and mode of delivery, there was no difference in the composition of intestinal flora among breastfeeding, goat milk formula and cow milk formula groups (P > 0.05), but their abundances were different.
Effect of feeding modes on intestinal flora of cases with the same age and delivery mode. MV1, 0 - 6-month-old, natural labor and breastfeeding; MF1, 0 - 6-month-old, cesarean section and breastfeeding; YV1, 0 - 6-month-old, natural labor and goat milk formula; YV2, 6 - 12-month-old, natural labor and goat milk formula; YV3, 12 - 36-month-old, natural labor and goat milk formula; YF2, 6 - 12-month-old, cesarean section and goat milk formula; YF3, 12 - 36-month-old, cesarean section and goat milk formula; NV2, 6 - 12-month-old, natural labor and cow milk formula; NV3, 12 - 36-month-old, natural labor and cow milk formula.
4.4. Effect of Age on Intestinal Flora of Cases Fed with Goat Milk Formula
At the phylum level, there was no difference in the composition of dominant bacteria (Actinobacteria, Firmicutes, Bacteroidetes and Proteobacteria) among the infants of natural labor aged 0 - 6 months, 6 - 12 months and 12 - 36 months in the goat milk formula group (P > 0.05) (Figure 2). However, the abundances were different, which were 95.17%, 99.94%, and 99.96%, respectively in Phase I, II, and III infants. Actinobacteria had the highest abundance in Phase I infants, followed by Phase II and III ones sequentially. The abundances of Firmicutes, Bacteroidetes and Proteobacteria were highest in Phase II infants, followed by Phase I and III ones sequentially. Hence, the intestinal flora changed obviously along with the growth and development of infants.
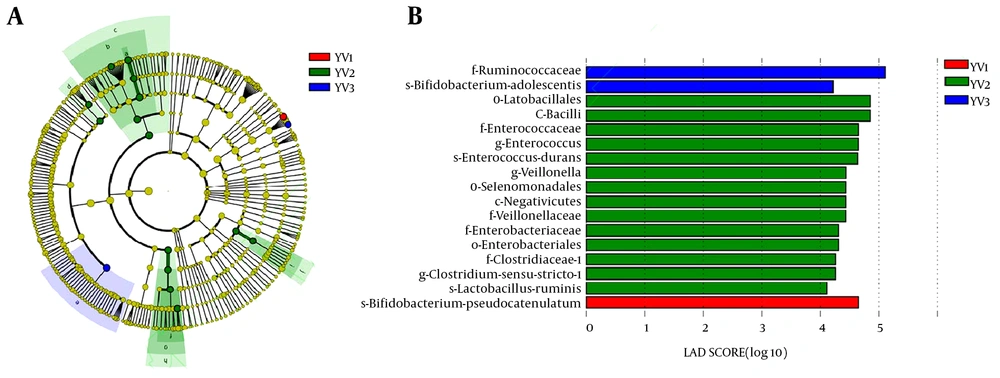
Lef Se analysis was performed for the infants fed with goat milk formula in the three age groups. Figure 5a shows bacteria that play crucial roles in the three groups. The intestinal flora of the 6 - 12-month-old infants of natural labor in the goat milk formula group different from those of the other two groups included Enterococcaceae, Lactobacillales, Bacilli, Clostridiaceae, Veillonellaceae, Selenomonadales, Negativicutes, Enterobacteriaceae and Enterobacteriales. As presented in Figure 5b, Bifidobacterium pseudocatenulatum is dominant in the infants aged between 0 and 6 months; Ruminococcaceae and Bifidobacterium adolescentis are dominant in the infants aged 12 - 36 months; Lactobacillus, Enterococcus and Clostridium are dominant in 6 - 12-month-old infants. Therefore, intestinal flora is most abundant in the infants aged between 6 and12 months.
Effect of age on intestinal flora of cases fed with goat milk formula. A, Lef Se analysis results; B, relative abundances. YV1, 0 - 6-month-old, natural labor and goat milk formula; YV2, 6 - 12-month-old, natural labor and goat milk formula; YV3, 12 - 36-month-old, natural labor and goat milk formula.
5. Discussion
The colonization of intestinal flora in infants and young children is a complex process affected by many factors, such as age, feeding mode and delivery mode. Early flora colonization in infants is affected by the mode of delivery, i.e. natural labor or cesarean section (18-20). In this study, high-throughput sequencing was conducted to evaluate the effects of delivery modes on the intestinal flora of infants at different ages who were fed by the same method. In the Phase I and II groups with different modes of delivery, the flora abundances were similar, but the abundance of the Phase III group was significantly different, which can be ascribed to complementary food supplementation.
In the neonatal period, the intestinal flora of breastfed and artificially fed infants is dominated by bifidobacteria, the number of which, however, is higher in the intestines of breastfed infants (21). The numbers of Bacteroides, Enterobacter, Clostridium, Enterococcus and Streptococcus are lower in breastfed infants (10). Moreover, the feces of both breastfed and artificially fed infants contain considerable Enterobacter, and the number of Bifidobacteria is not significantly different between them (22). In the past decade, Haarman et al. used fluorescence in situ hybridization and nucleic acid probe to compare the intestinal flora of breastfed and artificially fed infants within 20 days after birth, and found that bifidobacteria were dominant in the intestines of breastfed infants, followed by Lactobacillus and Streptococcus. Moreover, the numbers of Bacteroides and Bifidobacteria in the intestines of artificially fed infants were similar (23). In contrast, we herein used high-throughput sequencing to study the effects of feeding modes on the intestinal flora of infants with the same age and delivery mode. Actinobacteria accounted for 62.98% of bacteria in 0 - 6-month-old infants of the goat milk formula group, which was much higher than that of the breastfeeding group (43.74%), and the abundance of bifidobacteria was also higher. The influence of different feeding modes on infantile intestinal flora is valuable for the development of milk formula products.
Although breast milk is the most ideal nutrient for infant growth and development, the feeding rate is still low, and goat milk is a suitable raw material for infant formula because of low allergenicity and high nutritional value (24). In this study, high-throughput sequencing revealed that there was a slight difference in the intestinal flora of Phase I infants of natural labor between breastfeeding and goat milk formula groups. The intestinal flora of breastfeeding and goat milk formula groups mainly comprised Bifidobacterium. Clostridium-sensu-stricto-1 and Bacteroides, and the abundance of Bifidobacterium in the infants fed with goat milk formula was higher than that of breastfed infants. There was no difference in the Phase II infants of natural labor between goat milk formula and cow milk formula groups. In addition, the abundances of Bifidobacterium, Bacteroides and Prevotella-9 in the goat milk formula group were higher than those of the cow milk formula group.
The colonization of intestinal flora in infants is a dynamic process, being closely related to pathological changes. Regardless, the changes of intestinal flora in 0 - 36-month-old infants have seldom been studied until now. In this study, the dominant bacteria in three phases (0 - 36 months) were composed of Actinobacteria, Firmicutes, Bacteroidetes and Proteobacteria, with significantly different abundances though. As the age increased, the abundances of Actinobacteria and Bifidobacterium gradually decreased. Additionally, the abundance of Lactobacillus was highest in 6 - 12-month-old infants and lowest in 12 - 36-month-old infants.
In conclusion, this study assessed the effects of delivery mode, feeding mode and age on the intestinal flora of newborns and infants through high-throughput sequencing. For the infants with the same feeding method and age, the mode of delivery affected their intestinal flora. Furthermore, the infants fed with goat milk formula with the same way of delivery and age had the highest bacterial abundance. The findings provide valuable evidence for improving the health, metabolism and disease treatment of infants, and also pave the way for developing milk formula products.