1. Background
Chronic kidney diseases (CKD) indicate the irreversible loss of renal function, which eventually requires renal replacement therapy. According to the National Kidney Foundation, CKD is functional or structural damage of the kidney or a decrease in glomerular filtration rate (GFR) by < 60 mL/min/1.73 m2 over more than three months (1). The exact prevalence of CKD in children is not well known since no national statistics is released in this regard. However, in Bangabandhu sheikh Mujib Medical University (BSMMU), Dhaka, Bangladesh, the prevalence rate of CKD in children was 7.9% in 2013 (2). CKD has several complications, including growth retardation, malnutrition, hypertension, acidosis, metabolic bone disease, and a tendency to progress to end-stage renal disease even when the primary disease process is no longer active. A recent accepted treatment for ESRD in developed countries is renal replacement therapy. Donor shortage for renal transplantation is also a significant challenge to providing optimal treatment. Accordingly, the clinicians need to halt the progression of the CKD stage by preventing the deterioration of the renal function. The most important independent risk factor for renal disease progression is proteinuria; hence, the existing therapeutic strategies to prevent progression are to decrease urinary protein excretion (3). The restriction of dietary protein intake is associated with a significant decrease in the deterioration rate of renal function in both experimental animals and men (4). Low protein can ameliorating several uremic symptoms and complications through retention of waste products and toxins resulting from protein catabolism and excreted with the urine. Low protein may help to slow the progressive GFR decline due to the effects on glomerular hemodynamics; hence, protein restriction can delay the onset of chronic dialysis. In other words, dietary protein restriction is the heart of the nutritional therapy for CKD (5).
2. Objectives
This study aimed to analyze the effect of decreasing the RDA of protein to a minimum recommended level on growth status and the renal function rate.
3. Methods
This observational analytical study was conducted in the Department of Pediatrics, Dhaka Shishu Hospital, from January 2016 to December 2017. In this study, thirty 2-18-year-old children with CKD (all five stages), without dialysis and having a normal diet, referring to the outpatient department of the hospital were selected purposively as the study sample. All the children have retarded renal function induced by CKD. For the last six months, children with CKD on a normal protein diet with no restriction were selected purposively, the duration of whose illness was 3 - 9 months. The protein RDA was considered for these children. After six months, their nutritional and renal status were observed. The CKD children with heart disease, congenital diseases, diabetic nephropathy, renal transplantation, and dialysis were excluded from the study. Five patients with CKD stage V refused to take dialysis due to financial problems. A predesigned Performa containing the participants’ clinical details and their history by age, gender, duration of illness, and factors inducing CKD were filled. A thorough clinical examination with particular attention to CKD stage was performed. Before the intervention considering the protein RDA, necessary baseline investigations were conducted to obtain preliminary data from previous records of the patients (namely Hb, RBS, serum albumin, and serum ferritin) and creatinine clearance rate (CCr). An anthropometric assessment addressing weight and height was conducted by using a bathroom scale and stadiometer.
Body mass index (BMI) was calculated, and Z-scores (WAZ and HAZ) obtained from CDC-2000 growth charts (6) were represented as standard deviation scores (SDS). After obtaining the parents’ informed consent, 4 mL of venous blood sample was collected from each patient under all aseptic precautions for laboratory assessment of Hb, RBS, serum albumin, and serum ferritin. The CCr was calculated by using the standard Schwartz equation (3). The protein RDA was provided to the children according to the WHO’s dietary guideline (7). In this guideline, the safe level of protein intake for infants and children is calculated in g/kg/day according to their weight and gender. The protein and other supplementary micronutrients (namely iron and calcium) were given to the children by a skilled pediatric dietitian. During the active intervention periods, the participants were individually advised and monitored by a dietician to achieve the planned protein intake by selecting normal foods with no restriction in carbohydrate and fat intakes. Dietary adherence was monitored via phone calls to achieve the planned protein intake. After three and six months of intervention, the data from three days of dietary recalls were used to assess the dietary intake. The parents, especially mothers, were interviewed regarding the assessment of dietary intake. Standard-sized cups, glasses, spoons, and small bowls were shown to the mothers to calculate the average portion in the diet. Each patient was observed for six months, and his/her growth and renal function progression was assessed using anthropometric and laboratory assessments over three- and six-month periods. The collected data were checked, edited, and then analyzed using SPSS software. After six months of intervention, the child’s growth and the renal function progression rate were assessed and compared using the previously documented data by running the paired sample t-test. In this study, the interval data are presented as mean ± SD, and p < 0.05 was set as the level of significance.
4. Results
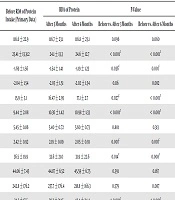
In this study, there was no patient in CKD stage I, and most of the patients were in stage III (36.7%) and stage IV (33.3%). Moreover, 13.3% of patients were in stage II, and 16.7% of the patients were in stage V (Table 1). Regarding the stages, the participants’ mean age was 12 (stage II), 9 (stage III), 6 (stage IV), and 6.4 (stage V) years. More male patients were in the stages II, III, and IV; however, females were dominant in stage V. Among the 30 CKD children, most of the patients were suffering from obstructive uropathy (36.66%), vesicourethral reflex (20%), reflux nephropathy (16.66%), glomerulonephritis (16.66%), and neurogenic bladder (10%). In Table 2, anthropometric measurements were compared with primary data after three and six months of RDA protein intake. In this regard, weight, WAZ, and BMI increased significantly after three and six months, compared to primary data (P < 0.05). Different laboratory data were compared with primary data after three and six months of RDA protein intake. Hb level increased significantly during three and six months of RDA protein intake, compared to the pre-intervention data (P < 0.05). Furthermore, other renal function tests such as S. creatinine, Blood urea, and CCr improved significantly after three and six months of RDA protein intake without dialysis (P < 0.05).
| CKD Groups | |||||
|---|---|---|---|---|---|
| Stage I | Stage II | Stage III | Stage IV | Stage V | |
| No. (%) | 0 | 4 (13.3) | 11 (36.7) | 10 (33.3) | 5 (16.7) |
| Age, mean ± SD | 0 | 12 ± 2.2 | 9 ± 3.8 | 6 ± 2.4 | 6.4 ± 1.8 |
| Sex, frequency | |||||
| Male/female | 0 | 4/0 | 8/3 | 8/2 | 1/4 |
Distribution of Study Subject by Age and Gender in Different CKD Stages (N = 30)
| Variables | Before RDA of Protein Intake (Primary Data) | RDA of Protein | P-Value | ||
|---|---|---|---|---|---|
| After 3 Months | After 6 Months | Before vs. After 3 Months | Before vs. After 6 Months | ||
| Height | 116.6 ± 22.9 | 116.7 ± 23.1 | 116.8 ± 23.1 | 0.096 | 0.060 |
| Weight | 23.41 ± 13.3c2 | 24.1 ± 13.3 | 24.6 ± 12.7 | < 0.001c | < 0.001c |
| WAZ | -1.69 ± 1.56 | -1.54 ± 1.41 | -1.19 ± 1.23 | 0.036c | 0.001c |
| HAZ | -2.04 ± 1.54 | -2.03 ± 1.51 | -2.02 ± 1.54 | 0.116 | 0.082 |
| BMI | 15.9 ± 3.1 | 16.47 ± 2.95 | 17.1 ± 2.7 | 0.027c | < 0.001c |
| Hb, mg/dL | 9.44 ± 2.08 | 10.36 ± 1.42 | 10.98 ± 1.33 | < 0.001c | < 0.001c |
| RBS, mmol/L | 5.45 ± 0.86 | 5.40 ± 0.72 | 5.60 ± 0.73 | 0.801 | 0.313 |
| Creatinine, mg/dL | 2.42 ± 0.92 | 2.19 ± 0.89 | 2.05 ± 0.91 | 0.001c | 0.001c |
| Urea, mmol/L | 36.5 ± 19.9 | 33.6 ± 21.0 | 31.9 ± 22.6 | 0.014c | 0.008c |
| Total albumin, mg/L | 44.06 ± 7.45 | 44.87 ± 6.52 | 45.59 ± 6.75 | 0.391 | 0.167 |
| Ferritin, ng/L | 242.8 ± 176.2 | 237.7 ± 176.4 | 218.8 ± 166.3 | 0.579 | 0.087 |
| GFR | 34.3 ± 17.5 | 38.2 ± 21.15 | 42.4 ± 24.4 | < 0.001c | < 0.001c |
5. Discussion
In this study, there were 11 patients (36.7%) in stage III, 10 patients (33.3%) in stage IV, 5 patients (16.7%) in stage V (without dialysis), and 4 patients (13.3%) in stage II. In a cross-sectional study by Gupta et al. (8), 27 (60%) children out of 45 persons had stage I, II, or III CKD while the rest (40%) had stage IV or V CKD (Kist-van Holthe tot Echten et al. (9) assessed 56 CKD children (aged 2 - 18 years) regarding protein restriction (safe protein levels by the WHO). After three years of follow-up, they observed growth in children with CKD, and their weight, WAZ, and BMI improved significantly. These findings were compatible with those of the present study (Table 2); hence, it is not recommended to reduce protein intake below safe levels (protein RDA of) by the WHO. In this study, after six months of intervention, the patients’ weight, WAZ, and BMI improved significantly. According to Butani et al. (10), dietary protein consumption increases serum creatinine level by protein catabolism; however, it does not decrease clearance. Hellerstein et al. (11) performed a study on 15 normal children and 33 pediatric renal children to evaluate creatinine excretion rate after protein meal (baked chicken) in normal children. In their study, renal child serum creatinine concentrations and creatinine excretion rates increased significantly. King and Levey (12) and Lew and Bosch (13) also showed that protein intake directly and quantitatively affected endogenous creatinine clearance in young, healthy subjects. In contrast, Rizzetto et al. (14) showed significant improvement in eGFR after a low protein diet (LPD) and its impact on the renal function of the nondialysis CKD patients. In this study, although the total albumin level increased after the intervention with RDA protein, the increase was not statistically significant. In the present study, the mean creatinine clearance rate (CCr) improved significantly after three and six months of RDA protein intake (Table 2). Similarly, Piccoli et al. (15) and Schwingshackl and Hoffmann (16) reported that low-protein diets in uremia patients reduced the symptoms of “uremic toxicity”. This is consistent with this study, indicating that S. urea level decreased significantly after the intervention. Koshy and Geary (17) demonstrated anemia and poor outcomes among children with CKD. Kaspar et al. (18) also reported that forty-five percent of children with CKD were anemic in the CKD cohort and that the lower glomerular filtration rate (GFR) was associated with lower levels of hemoglobin. After birth, a group of peritubular interstitial cells in the kidney was the source of erythropoietin production. According to Warady et al. (19), CKD children require additional parenteral and enteral iron to store. However, in this study, the Hb level significantly increased, and anemia improved significantly after intervention with RDA protein.
5.1. Conclusions
After intervention using the recommended dietary allowance protein for the CKD children at different stages with no dialysis, renal function (S. creatinine, s. urea, and CCr) and growth (Weight, WAZ, and BMI) significantly improved over three- and six-month periods (P < 0.05).