1. Background
Perinatal asphyxia is the third-leading (23%) cause of neonatal death worldwide (1). Approximately 2 - 30 neonates out of every 1000 live full-term births experience perinatal asphyxia (2). Of these asphyxiated neonates, 15% - 20% die during the neonatal period, and up to 25% of survivors suffer from neurological disabilities (2, 3). Moreover, asphyxia can lead to hypoxic injury to the brain, heart, lungs, liver, gut and kidneys, and it is especially likely to cause neurodevelopmental deficits (4-7). Perinatal asphyxia is diagnosed via multiple clinical measures, including Apgar scores, cord pH, delayed respiration, fetal distress and intrapartum electronic fetal monitoring (8, 9).
MicroRNAs (miRNAs) are non-coding single-stranded RNA molecules that are approximately 22 nucleotides in length. miRNAs bind homologous seed sequences in the 3’-UTR of target genes and impair gene expression (10). Several studies have demonstrated that miRNAs participate in cell metabolism, proliferation, differentiation, autophagy and apoptosis, and they play a crucial role in development- and disease-related processes (11-14). Moreover, miRNAs have been extensively researched as biomarkers and as therapeutic targets for certain diseases (15-17). The expression of miR-210, which is located on chromosome 11, can be induced by hypoxia (18, 19) and it is elevated in individuals with cardiac diseases (20, 21) and tumors (22, 23).
2. Objectives
In the present study, we evaluated miR-210 expression in peripheral blood samples obtained from 42 asphyxiated neonates and 41 healthy neonates using quantitative real-time PCR (qPCR). Statistical analysis was performed for predicting the relationship between miR-210 expression and other indicators associated with the diagnosis of asphyxia. Lastly, bioinformatics analysis was performed for exploring the functional classifications of putative miR-210 target genes, as well as for determining the associated pathways and diseases.
3. Methods
3.1. Patients
The Ethics Committee of the Third People’s Hospital in Dongguan, China, approved the present study. Written informed consent was obtained from the parents of the subjects. A total of 42 asphyxiated neonates who were born at the Third People’s Hospital between September 2015 and August 2017 after at least 37 weeks of gestation period were included in this study. Forty-one full-term healthy neonates were included as healthy controls. On the basis of strict criteria used in previous studies (24, 25), the neonates were diagnosed with asphyxia if they had a cord pH of < 7.15 and 5 minutes Apgar score of ≤ 7. White blood cell (WBC), procalcitonin (PCT) and blood gas analysis were also performed using blood obtained from the umbilical artery after birth.
3.2. Sample Collection
At 72 hours after birth, peripheral blood samples were obtained from the radial artery in asphyxiated and healthy neonates. The collected samples were immediately stored at -80°C until further use for RNA extraction.
3.3. qPCR
Total RNA was isolated from the peripheral blood samples using a HiPure Blood/Liquid RNA Kit in accordance with the manufacturer’s instructions. RNA concentrations were quantified using a NanoDrop™ 2000 (Thermo, MA, USA). Subsequently, RNA was reverse-transcribed using a miR-210-specific oligonucleotide (5’-GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGA CCAGTGT-3’) and a U6-specific oligonucleotide (5’-AACGCTTCACGAATTTGCGT-3’). The qPCR reaction was performed on a MiniOpticon™ real-time PCR system (Bio-Rad, CA, USA) with GoTaq® qPCR Master Mix (Promega, WI, USA), following standard procedures. The primers used in this study were as follows: miR-210 F: 5’-AGCCCCTGCCCACCGC-3’, miR-210 R: 5’-GTGCAGGGTCCGAGGT-3’, U6-F: 5’-CTCGCTTCGGCAGCACA-3’ and U6-R: 5’-AACGCTTCACGAATTTGCGT-3’. The 20-µL qPCR system contained 10 µL of 2 × SsoAdvanced™ Universal SYBR® Green Supermix (Bio-Rad, USA), 1 µL of primers (5 pmol each), 1 µL of DNA template (approximately 20 ng) and 8 µL of ddH2O. The thermal steps were as follows: 94°C for 3 minutes, followed by 40 cycles of 95°C for 15 seconds and 60°C for 25 seconds. U6 was used as the reference gene. The 2-ΔΔCt method was applied for calculating the relative expression level of each miRNA.
3.4. Determination of Neonatal Nervous Behaviors
A previous study demonstrated that neonatal behavior neurological assessment (NBNA) can be used for the comprehensive assessment of the nervous system, including general condition, action behavior, muscular tension and primitive reflexes (26). In this study, NBNA was performed 7 days after birth.
3.5. Statistical Analysis
All experiments were performed at least three times. Data are expressed as the mean ± standard error (SEM). Statistical analysis including the receiver operating characteristic (ROC) curve plot was performed using SPSS (version 19.0). Chi-square tests and Student’s t-tests (two-tailed) were used for analyzing the differences between the groups. Statistical significance was set at P < 0.05.
3.6. Bioinformatics Analysis
Putative miR-210 target genes were predicted using miRanda 3.3a. GO enrichment analysis, which was used for revealing genetic regulatory networks of miR-210 targets by forming hierarchical categories on the basis of molecular functions, biological processes and cellular components, was performed at the Gene Ontology Consortium website (http://www.geneontology.org). The Kyoto Encyclopaedia of Genes and Genomes (KEGG) pathway and disease analysis were performed at the KEGG website (http://www.genome.jp/kegg/) to explore the significant pathways and diseases of the differentially expressed mRNA targets, respectively. An autism spectrum disorder database (AutDB, http://autism.mindspec.org/autdb/Welcome.do) was used for creating a list of autism susceptibility genes. An epilepsy database (EpiGAD, http://www.epigad.org/) and the Epilepsy Genetics Initiative database (https://www.cureepilepsy.org/egi/genes.asp) were utilized for creating a list of epilepsy susceptibility genes. A miRNA-target interaction network was constructed and visualised with Cytoscape-V2-8-3 (https://www.innatedb.ca/cytoscape-v2.8.3/plugins/).
4. Results
4.1. Differing Characteristics of Asphyxiated Neonates and Control Groups in Clinical Diagnosis
A total of 83 neonates were recruited in this study, including 42 asphyxiated neonates (cord pH < 7.15; 5 or 10 minutes Apgar score < 7) and 41 healthy neonates. The clinical characteristics of the healthy controls and asphyxiated neonates are summarised in Tables 1 - 3. Large significant differences (P < 0.01) in delivery mode, Apgar (5 minutes and 10 minutes) scores, day 7 NBNA, WBC and blood gas analysis (PCO2, PO2, lactic acid and pH) scores were observed between the control and asphyxia groups. However, no significant difference in sex, gestational age or PCT was observed between the control and asphyxia groups.
| Number | Gender, Male/Female | Delivery Mode, Vaginal Birth/Cesarean** | Gestational Age, d | |
|---|---|---|---|---|
| Healthy controls | 41 | 21/20 | 35/6 | 272.00 ± 1.24 |
| Asphyxiated neonates | 42 | 22/20 | 21/21 | 275.50 ± 1.55 |
| χ2/t-value | - | 0.01 | 11.82 | 1.75 |
| P value | - | 0.9157 | 0.0006 | 0.0835 |
aχ2 value, gender, delivery mode; t-value, Gestational age.
b*, P < 0.05 vs. control; **, P < 0.01 vs. control.
| 5 Minutes Apgar Score** | 10 Minutes Apgar Score** | Day 7 NBNA Score** | |
|---|---|---|---|
| Healthy controls | 9.95 ± 0.03 | 9.95 ± 0.03 | 38.80 ± 0.22 |
| Asphyxiated neonates | 8.50 ± 0.21 | 9.24 ± 0.18 | 36.61 ± 0.34 |
| t-value | 6.81 | 3.92 | 5.46 |
| P value | < 0.0001 | 0.0002 | < 0.0001 |
a*, P < 0.05 vs. control; **, P < 0.01 vs. control.
| WBC, 109/L** | PCT, ng/mg | PCO2, mmHg** | PO2, mmHg** | Lactic Acid, mmol/L** | pH** | |
|---|---|---|---|---|---|---|
| Healthy controls | 14.29 ± 1.08 | 0.50 ± 0.12 | 48.05 ± 1.38 | 53.12 ± 4.11 | 2.03 ± 0.19 | 7.32 ± 0.01 |
| Asphyxiated neonates | 49.80 ± 11.85 | 4.11 ± 2.21 | 63.61 ± 1.41 | 34.39 ± 1.10 | 7.50 ± 0.44 | 7.08 ± 0.01 |
| t-value | 2.95 | 1.61 | 7.87 | 4.41 | 11.43 | 14.58 |
| P value | 0.0042 | 0.1119 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 |
a*, P < 0.05 vs. control; **, P < 0.01 vs. control.
4.2. qPCR and Statistical Analysis
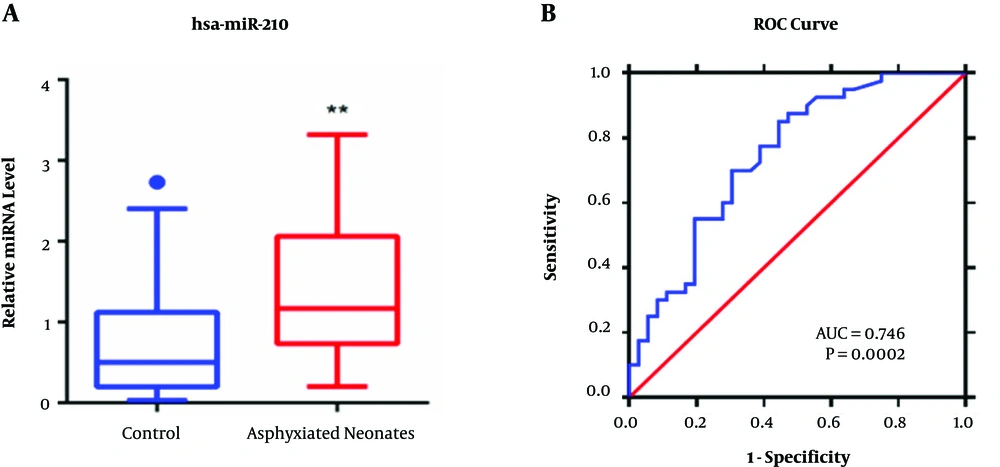
We analyzed miR-210 expression in peripheral blood samples obtained from 42 asphyxiated neonates and 41 healthy neonates using qPCR. As shown in Figure 1A, miR-210 expression was noted to be 1.8-fold higher in the peripheral blood of asphyxiated neonates (P < 0.01) compared with that of healthy controls. Moreover, a large significant negative correlation (P < 0.01) was noted between miR-210 expression with the day 7 NBNA score and lactic acid (Table 4). Figure 1B also showed that the area under curve (AUC) of miR-210 expression in the ROC curve was > 0.7 (AUC = 0.746, P = 0.0002). The 95% CI of miR-210 expression was 0.6352 to 0.8572, suggesting that miR-210 expression could be a useful indicator in diagnosing neonates with asphyxia.
| Parameter | miR-210 Expression | 95% CI | |
|---|---|---|---|
| r | P Value | ||
| 5 min Apgar score * | -0.2825 | 0.0178 | -0.4907 ~ -0.04373 |
| 10 min Apgar score | -0.1615 | 0.1818 | -0.3880 ~ 0.08351 |
| Day 7 NBNA score ** | -0.3303 | 0.0052 | -0.5297 ~ -0.0962 |
| WBC, 109/L | 0.1711 | 0.1567 | -0.07365 ~ 0.3964 |
| PCO2, mmHg * | 0.3032 | 0.0107 | 0.06639 ~ 0.5077 |
| PO2, mmHg | -0.1836 | 0.1282 | -0.4072 ~ 0.0608 |
| Lactic acid, mmol/L ** | 0.4597 | < 0.0001 | 0.2452 ~ 0.6312 |
| pH ** | -0.4111 | 0.0004 | -0.5938 ~ -0.1881 |
a*, P < 0.05 vs. control; **, P < 0.01 vs. control.
qPCR and ROC curve of miR-210 expression. A, miR-210 expression in the peripheral blood of asphyxiated neonates. qPCR analysis of miR-210 levels in the peripheral blood of asphyxiated neonates (n = 42) and healthy controls (n = 41). *P < 0.05, **P < 0.01. B, The ROC curve plot of miR-210 expression.
4.3. Bioinformatics Analysis of miR-210 Target Genes
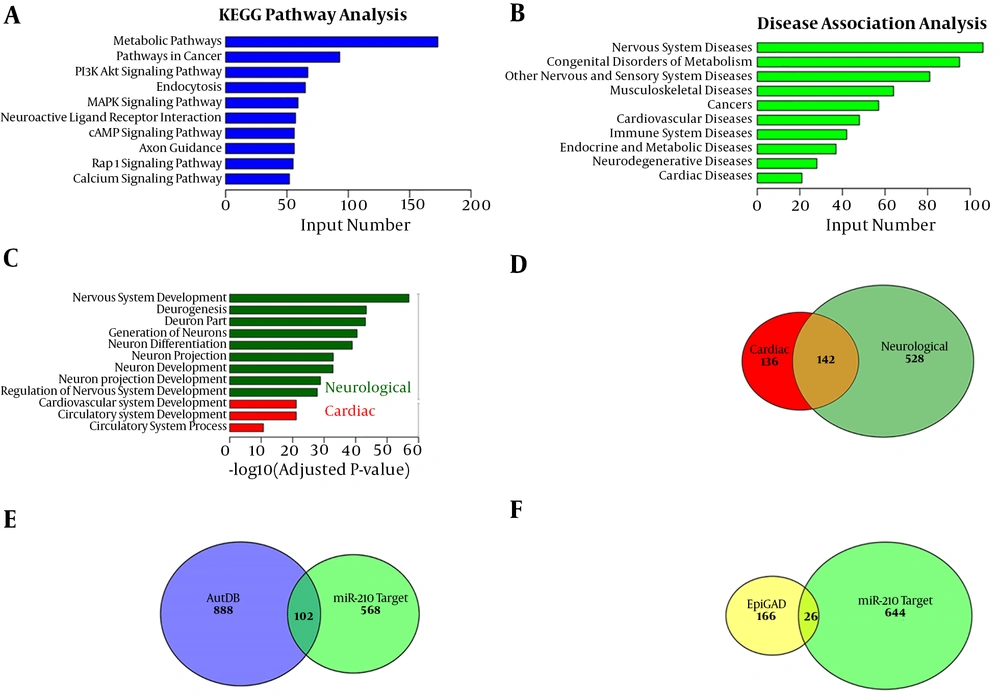
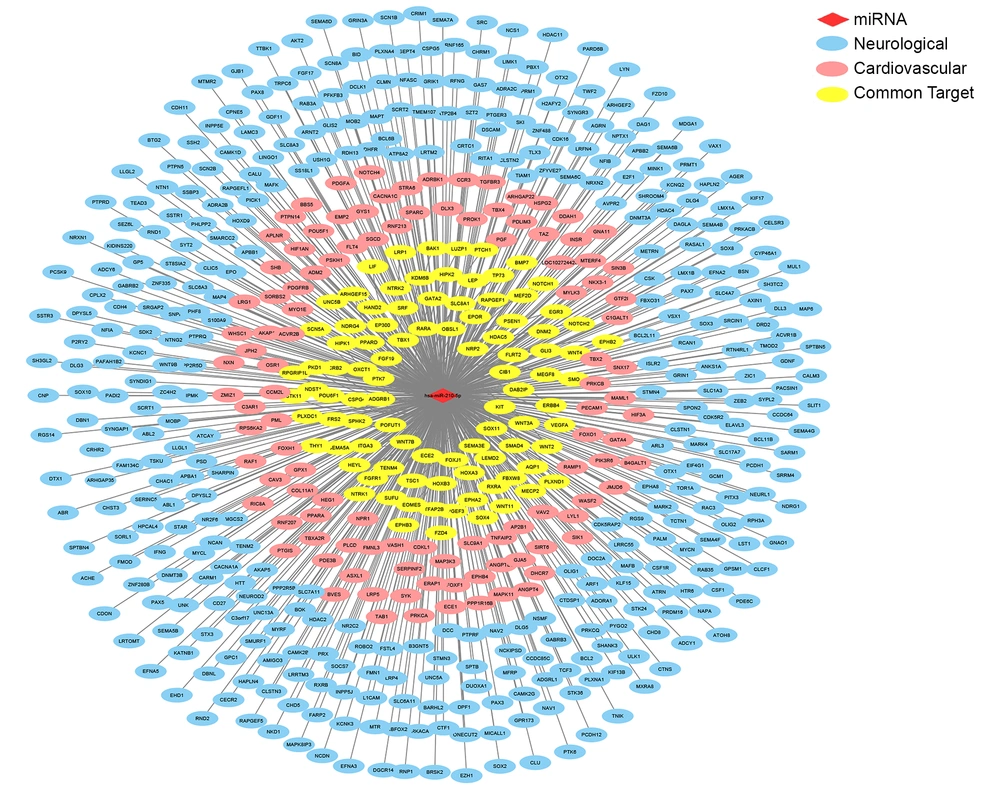
Mature miRNAs bind to the 3’-UTR of target genes and regulate gene expression. The miR-210 targets were predicted using miRanda 3.3a. KEGG and GO enrichment analysis were performed for exploring the functional classification of these 3848 putative miR-210 target genes and for determining their associated pathways and diseases. Findings of the KEGG pathway analysis indicated that miR-210 was primarily involved in the metabolic, cancer-related, PI3K/AKT, endocytosis and MAPK pathways (Figure 2A). Findings of the KEGG disease association analysis showed that miR-210 was primarily associated with diseases of the nervous system, other sensory systems and the cardiovascular system; congenital metabolic disorders; and cancer (Figure 2B). Furthermore, findings of the GO analysis showed that miR-210 targets were primarily enriched in nine neurological development pathways and three cardiac development pathways, respectively (Figure 2C). Findings of the GO analysis also demonstrated that 670 putative miR-210 target genes were enriched in neurological development processes, 278 target genes were enriched in cardiac development processes and 142 genes were implicated in both processes (Figure 2D). We compared the 670 putative miR-210 targets involved in neurological processes with a list of 990 autism susceptibility genes and 192 epilepsy susceptibility genes for examining the association between miR-210 and autism or epilepsy. A total of 102 putative miR-210 target genes were significantly associated with autism, and 26 were associated with epilepsy (Figure 2E and F). The interaction network of miR-210 and its targets in the neurologic and cardiovascular pathways revealed that most miR-210 targets were associated with neurological processes (Figure 3).
KEGG and GO enrichment analysis of putative miR-210 target genes. A, The 10 main KEGG pathway categories of 3848 putative miR-210 target genes. B, The 10 main KEGG disease categories of 3848 putative miR-210 target genes. C, Nine and three significantly enriched miR-210 targets of GO terms associated with neurological and cardiac development pathways, respectively. D, Venn diagram showing that 670 and 278 putative targets were enriched in the GO terms ‘neurological development’ and ‘cardiac development’, respectively, and 142 putative targets were enriched in both GO categories. E and F, Overlap between the 670 putative targets enriched in the GO term ‘neurological development’ and genes in the autism spectrum disorder (AutDB) and epilepsy (EpiGAD) databases.
Interaction network of miR-210 and its targets in neurologic and cardiovascular pathways. The red rhombus indicates miR-210; the blue ellipse refers to neurological genes that are regulated by miRNAs; the light red ellipse indicates cardiovascular genes that are regulated by miRNAs; and the yellow ellipse represents common genes in the neurologic and cardiovascular systems that are regulated by miRNAs.
5. Discussion
Asphyxia induces the up-regulation of miR-210 expression in vitro (18, 27) and in vivo (22, 28). Under hypoxic conditions, cells express hypoxia-inducible factor (HIF-1) in response to hypoxic stress. HIF-1 stimulates the transcription of the miR-210 primary transcript by binding to the hypoxia response element in the miR-210 promoter (29). Increased miR-210 expression suppresses the expression of prolyl hydroxylases and succinate dehydrogenase complex subunit D, which stabilises HIF-1 and promotes miR-210 production (30). Elevated miR-210 expression is used as a predictive marker for tumor hypoxia because of the association between miR-210 and HIF-1 (28). However, whether miR-210 is associated with asphyxia in neonates remains unknown. In the present study, we evaluated miR-210 expression in the peripheral blood of asphyxiated neonates (n = 42) and healthy neonates (n = 41). Our results showed that miR-210 expression was 1.8-fold higher in the asphyxiated neonates than in the healthy neonates (P < 0.01).
Perinatal asphyxia is defined as a lack of blood flow or impaired respiratory gas exchange accompanied with acidosis in a fetus (3). Asphyxia can occur immediately before birth, during birth or after birth, and it results in profound systemic and neurologic sequelae owing to hypoxia or anoxia (3, 31). Hypoxia and ischemia induce cortical sparing brain injury, deep grey matter injury and neuronal death in neonates, and they ultimately lead to neonatal hypoxic–ischemic encephalopathy (HIE) (32-34). HIE specifically refers to the neurologic sequelae of perinatal asphyxia, including mental retardation, visual motor or visual perceptive dysfunction, increased hyperactivity, cerebral palsy and epilepsy (33, 35, 36). In addition to neurologic injury, myocardial injury or cardiovascular dysfunction can occur in 50% - 80% of asphyxiated neonates (37, 38). Decreased contractility, decreased cardiac output, decreased stroke volume and increased pulmonary artery pressure are frequently observed after asphyxia, and these symptoms can lead to hypotension, metabolic acidosis and pulmonary hypertension (38). Several studies have recently demonstrated that the inhibition of miR-210 provides neuroprotection against hypoxic–ischemic brain injury in neonatal rats (39, 40). Therefore, we hypothesized that miR-210 plays a vital role in promoting the development of neurological sequelae of perinatal asphyxia by targeting genes associated with nervous system development. KEGG and GO enrichment analysis of putative miR-210 target genes indicated that miR-210 participates in neurologic and cardiovascular processes, and that these genes may be involved in the neurologic and cardiovascular injuries associated with asphyxia. In addition, miR-210 target genes were found to be significantly associated with autism and epilepsy, which cannot be assessed during neonatal health screening. Combined with the finding that miR-210 expression was closely associated with other clinical diagnostic indicators and the obtained ROC curve, the findings of this study suggest that the verification of miR-210 expression may be a useful indicator for diagnosing neonates with asphyxia. This indicator may help in identifying certain neurological and cardiac diseases that cannot be diagnosed during neonatal health screening and, therefore, provide early prevention and treatment for asphyxiated neonates.
5.1. Conclusions
In the present study, miR-210 expression was up-regulated in the peripheral blood of asphyxiated neonates, and its expression was also closely linked to other clinical diagnostic indicators. Thus, miR-210 expression could potentially be used as an indicator in diagnosing neonates with asphyxia. It could help identify some neurological and cardiac diseases that are not diagnosable during neonatal health screening and can provide early prevention and treatment for asphyxiated neonates.