1. Introduction
Homocysteine is a nonessential amino acid derived from methionine with demethylation. Homocysteine can be converted to methionine by re-methylation or to cysteine by transsulfuration. Methylenetetrahydrofolate Reductase (MTHFR) is an important enzyme of homocysteine metabolism. This enzyme catalyzes the synthesis of 5-MTHF from THF (1).
It is shown that the elevated serum homocysteine level is associated with an increased overall risk of ischemic diseases such as ischemic heart disease (2). Also, hyperhomocysteinemia is related to many neurological and psychiatric disorders, including dementia (3), stroke (4), epilepsy (5), Parkinson’s disease (6), schizophrenia (7), mood disorders (1), and some other neuropsychiatric diseases. The plasma concentration of tHcy can predict the cognitive decline in the elderly (3). Otherwise, a modifiable risk factor for vascular dementia could be an elevated plasma tHcy concentration (8).
The association between methylation and psychiatric disorder has been detected for several decades. Some studies showed elevated homocysteine in depressed patients. On the other hand, some population-based studies relieved that elderly persons with a history of B12 deficiency and secondary hyperhomocysteinemia are more likely to have depressive symptoms. Eventually, determining the level of homocysteine, B12, and folate in the broad list of neuropsychiatric disorders is critical, and therefore, the treatment of primary and secondary cases of hyperhomocysteinemia in these cases could result in relieving some symptoms of these diseases and ultimately prevent disease manifestation (9).
Impaired metabolism due to a genetic alteration in homocysteine metabolism may lead to hyperhomocysteinemia. This alteration includes enzymes such as mutations in MTHFR or deficiency in cofactors (vitamin B6, B12, and folate). In the MTHFR gene, more than 40 polymorphisms have been reported, of which two mutations C677T (rs1801133) and A1298C (rs1801131), have been extensively studied. It has been reported that the C677T mutation decreases MTHFR activity and increases the plasma homocysteine level, while the second polymorphism A1298C results in reduced enzymatic activity but to a lesser extent than does the C677T mutation. The reduced enzymatic activity of MTHFR could result in decreased 5- MTHF concentrations increased homocysteine concentrations, and reduced methylation capacity. Polymorphisms such as A1298C and C667T in the MTHFR encoding gene are associated with the impaired function of MTHFR and the decreased bioavailability of folate, resembling folate deficiency (10).
Previous case reports regarding hyperhomocysteinemia have presented patients with stroke or psychiatric symptoms and seizures. But, this time, we present a patient with mood symptoms, psychosis, stroke, epilepsy with hyperhomocysteinemia, and possible drug complications, which indicates the necessity of choosing the drug appropriately, especially those related to underlying neurometabolic disorders.
2. Case Presentation
A 20-year-old lady was referred to the emergency ward of Roozbeh hospital, Tehran, Iran. The reason for referral was talkativeness, aggressive and violent behavior, and paranoid ideas. First psychiatric manifestations including anxiety and obsessive-compulsive symptoms developed after a psychological stressor, which was treated with fluoxetine by referring to a neurologist. With this treatment, the patient begins to experience the exacerbation of manic symptoms. As a result, fluoxetine was discontinued, and risperidone was started. Risperidone was prescribed at a dose of 2 mg per night, and after two weeks, rest tremor and hypokinesia began and then evolved. After that, pramipexole at a dose of 0.7 mg 1/2 daily was administrated, which led to visual hallucination in our patient.
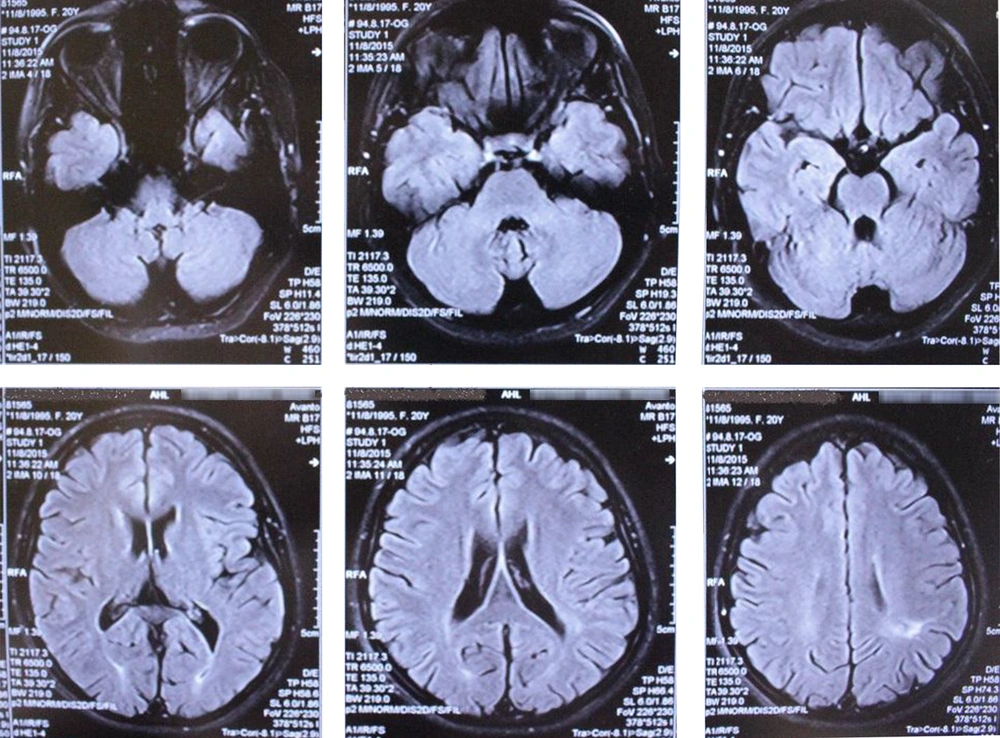
Later on, hemiparesis, face asymmetry, and ptosis occurred. In paraclinical assessment, brain magnetic resonance imaging (MRI) and electroencephalography (EEG) were applied. The brain MRI showed “gliosis and encephalomalacia adjacent to left occipital horn”, as well as “diffused brain atrophy” (Figure 1). The parietooccipital infarct was diagnosed in brain imaging, and AcetylSalicylic Acid (ASA) was prescribed.
In approach to the patient with young stroke, in the first stance, we evaluated all essential assessments, including heart echocardiography, TEE, and coagulative tests such as C3, C4, CH50, PrC, PrS, and anti-thrombin III. Also, we performed all vasculitis tests, including ANA, RF, anti-ds-DNA, and anti-phospholipid-Ab.
In our evaluation, all laboratory assessments were negative, except for homocysteine which was 74 µmol/L, at the time of the cerebrovascular event. At that time, the levels of B12 and folate were within the normal limit. The laboratory tests are summarized in Table 1.
| Test | Normal Range | Our Patient |
|---|---|---|
| C3 | 66 - 185 | 90 |
| C4 | 15 - 52 | 42 |
| CH50 | 42 - 95 | 63 |
| PrC | 65 - 135 | 80 |
| PrS | 15 - 35 | 23 |
| Anti-thrombin III | 17 - 30 | 25 |
| FANA | Up to 1/100 | 1/40 |
| RF | 0 - 20 | 5 |
| Anti-ds-DNA | > 75 | 35 |
| Anti-phospholipid-Ab-IgM | < 12 | Not detected |
| Vit B12 | 187 - 883 | 780 |
| Folic Acid | 3.1 - 20.5 | Over 20 |
There was no history of alcohol, substance abuse, and PPI prescription, which could potentially elevate the level of homocysteine or induce secondary hyperhomocysteinemia. Ultimately, clozapine was prescribed to control hallucination in patients with probable drug-induced parkinsonism. The psychiatrist supposed that parkinsonism in the patient could be due to risperidone administration and decided to select anti-psychotics with less extrapyramidal effect, above them, clozapine. Then, the patient experienced two attacks of loss of consciousness with tonic-clonic movement and upward gaze. Each attack lasted about five minutes, after which the patient had the post-ictal phase with about 10 minutes of drowsiness. Initially, the neurologist started levetiracetam with a dose of 500 mg to control seizures in the patient.
Due to the symptoms of psychosis in the patient, the neurologist discontinued levetiracetam and replaced carbamazepine. In outpatient follow-up, she experienced visual distortions (seeing the mother's face in another way) that were eliminated with increasing the carbamazepine dose. In cognitive assessment, memory impairment was prominent such that in delayed recall tests, the score was 0 out of 5.
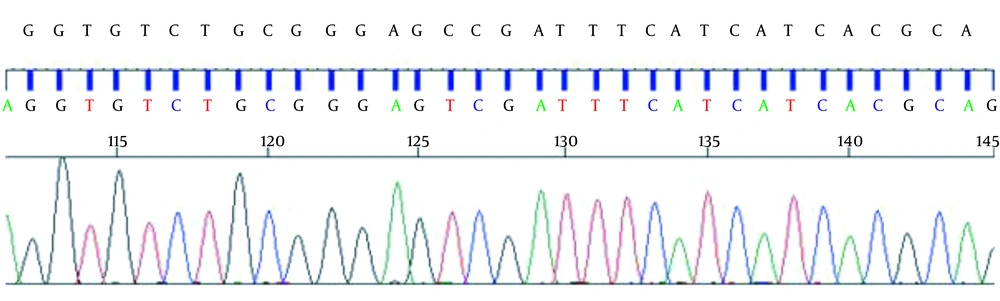
After discharge, genetic consultation was considered for the patient. Because of the elevated homocysteine level and also the low level of methionine, it was expected that MTHFR was reduced. It has been shown that the most prevalent mutation in this gene is in the C677T region (rs1801133). This part of the gene was sequenced, and as expected, it showed that the cytosine base, normally seen in this SNP, was replaced with the thymine base (Figure 2).
It should be noted that the patient's condition was the result of a consanguineous marriage. Regarding family history, epilepsy was reported in one sister of her father, and depression in the other sister of her father. In addition, two cousins experienced post-partum depression. Besides psychiatric and neurological symptoms, she had different manifestations endocrinopathy. As mentioned previously, she suffered from hypothyroidism with a high level of anti-TPO, and also was affected by polycystic ovary syndrome, elevated serum insulin level (> 30, normal value: 2 - 22), low level of 25OH vitamin D (10.1), and elevated parathyroid hormone (153, normal value: 9-94).
Three years prior to admission, the patient was evaluated for menstrual irregularity, through which treatment with metformin (1000 mg/day) was considered for polycystic ovary syndrome. In laboratory evaluation, serum homocysteine was 74 µmol/L (normal range: 5 - 15 µmol/L). In plasma amino acid HPLC, the methionine level was normal. During these three years, she was advised to use folate and vitamin B12 orally. Furthermore, hypothyroidism (with anti-TPO > 600 IU/mL) was detected and treated with levothyroxine.
3. Discussion
In general, it seems that by emerging mood symptoms and initiating the treatment of fluoxetine and risperidone, stroke occurred in the patient, and parietooccipital stroke or clozapine prescription caused a secondary seizure. By the use of levetiracetam to control the seizure, behavioral symptoms were exacerbated. Therefore, with initiating risperidone, drug-induced parkinsonism added to the patient's symptoms. Pramipexole was prescribed to treat drug-induced Parkinsonism, which led to an exacerbation of psychosis and ultimately patient hospitalization.
A review of case reports of patients with hyperhomocysteinemia indicated the evaluation and description of each patient with one major problem exclusively, but in our report, the patient was suffering from different medical conditions and neuropsychiatric disorders including stroke, seizures, mood problems, extrapyramidal complications, and many drugs side effects, along with hyperhomocysteinemia, which posed a lot of challenges to the patient's treatment. There were a few challenging points in this patient that should be reviewed and reassessed. The origin of the patient's psychotic and behavioral symptoms was the main challenge; whether these signs and symptoms were secondary to epilepsy or occurred due to primary psychiatric disorder.
Visual distortion could be a type of occipital lobe epilepsy due to intracerebral infarct. In a similar case, a 70-year-old man experienced visual hallucination after the occurrence of a hemorrhagic infarct. He saw a well-known face in a transient pattern but at recurrent times a day. In the DWI sequence of brain MRI, the territory of stroke was similar to that of our patient: The temporo-occipital lobe (11). In a parallel pathway, the stimulation of the visual association cortex could be responsible for this complex hallucination. On the other hand, it was probably related to an epileptic basis.
Our patient saw the mother's distorted face repeatedly, and short-lasting probably related to epileptiform discharge in EEG. Fortunately, this symptom resolved after increasing carbamazepine. According to this evidence, we supposed that this symptom could be a form of occipital epilepsy. Interestingly, a type of misidentification syndrome happened based on background psychosis in our patient. Capgras syndrome is the best-known form of misidentification syndrome defined with this idea "A close relative is replaced by another person." Although in the review of case reports, schizophrenia is the principal disorder associated with capgras, multifarious medical diseases accounted for etiology in 19% of the cases (12).
Another major challenge was choosing the appropriate medications for treating seizures. An Antiepileptic Drug (AED) with the least effect on homocysteine metabolism, as well as minimal cognitive, mood, and behavioral complications, is preferred. Some studies showed classic AEDs such as phenytoin (13), phenobarbital (14), valproic acid (15), and carbamazepine (15) most probably could be associated with increased homocysteine levels. However, increased homocysteine levels may also be observed in patients treated with such new-generation AEDs as oxcarbazepine and topiramate (16). Also, the use of metformin can exacerbate hyperhomocysteinemia, while metformin was prescribed for this patient for weight management (17).
Phenytoin is an enzyme inducer; so, it can modulate the activity of liver enzymes. Consequently, it causes cofactor depletion, particularly folic acid and vitamin B12, leading to hyperhomocysteinemia. It is well-known that patients on phenytoin often have low serum folic acid levels. Studies showed that valproic acid is associated with lower folic acid levels. Sodium valproate is not the first choice due to the possibility of cognitive impairment, parkinsonism, and its effect on the homocysteine metabolism cycle (18, 19).
Besides, levetiracetam can induce nervousness and psychotic symptoms in the patient and is not recommended due to the history of psychotic symptoms exacerbation. Lamotrigine can also affect homocysteine metabolism and is not preferred to control seizure attacks. Finally, considering the limitations and challenges in choosing an anti-epileptic treatment, carbamazepine was initiated in the patient. Carbamazepine does not affect cognitive impairment and has minimal effects on homocysteine metabolism; due to its mood-stabilizing properties, it has positive effects on behavioral and psychiatric symptoms. Ultimately, the seizure and behavioral symptoms of the patient were partially controlled. Visual distortion in the patient was effectively reduced by increasing the dose of carbamazepine.
Ischemic stroke that happens at young ages can be attributed to hypercoagulability state due to hyperhomocysteinemia. In patients with hyperhomocysteinemia, to prevent ischemic stroke, the use of pyridoxine (B6), cobalamin (B12), and folate might be considered, but its effectiveness is not well established. Regarding the treatment and prevention of recurrent stroke, ASA should be administered and continued in patients. On the other hand, homocysteine should be decreased with the administration of vitamin B-group supplements, including B12 and folic acid, and should be maintained at a level below 14 µmol/L (9).
Several articles investigated homocysteine levels during antipsychotic therapy, specifically second-generation antipsychotics. There are conflicting data regarding homocysteine levels and antipsychotic treatment. Results showed that all second-generation antipsychotics could increase the homocysteine level, and there is no recommendation on the best choice of antipsychotics in patients with hyperhomocysteinemia (20).
As mentioned above, the rational use of drugs for psychosis treatment in this complicated case had many challenges and complexities. Appropriate treatment for these patients should have the least minimal extrapyramidal and cognitive side effects. This method also must not affect the metabolism of homocysteine or seizure threshold.
In general, the multidisciplinary approach is critical in such patients, and interdisciplinary collaboration and consultation with various specialties of psychiatry, neurology, endocrinology, and clinical pharmacy can lead to fewer complications and better treatment. The holistic vision and comprehensive approach to the patient with considering the underlying disorder and using the appropriate treatment of choice with minimal complications due to the possibility of drug interactions can lead to safe treatment and symptom control in our patient.
3.1. Conclusions
In patients with elevated serum homocysteine and psychiatric disorder, it is imperative to consider MTHFR gene mutation. A challenge in these patients is to select the medication that does not deteriorate hyperhomocysteinemia. Furthermore, clinicians must note various complications of this condition including vascular events, psychosis, and seizure. In this complex case, we used carbamazepine that was accompanied by clinical improvement.