1. Background
Attention is a core component and an integral player in the human sensation, perception, and cognition organization that hierarchically affects elemental processing and higher-level mental operations (1). Deficits in attentional capacity profoundly disturb the formation of advanced adaptive behaviors and have repeatedly been evidenced in various neuropsychiatric pathologies such as dementia (2), psychotic (3), mood (4), and personality disorders (5). Notably, our ability to focus on the ongoing task, selective attention, stems from the coherent suppression of the task-irrelevant attributes that necessitate the recruitment of complicated specialized mechanisms for producing this intricacy throughout the limited channel of attention (6, 7). A dual-mechanism control (DMC) framework implies that the variations in attentional control could be the immediate consequence of the rivalry between two categories of controlling routes: A proactive process that contributes to the features of the stimulus (e.g., paying attention to color and ignoring the word) and a reactive pathway that plays roles after the stimulus is processed. Both of them trigger some conflicts represented in different segments of neural correlates (8, 9). Determining these steps and their interaction could provide the field with a valuable tenet. Among the currently existing paradigms, the Stroop task explicitly evaluates the concept of interference and conflict processing (monitoring, detection, and resolution), such that reaction time in a color-naming task would be increased due to the incongruent semantic characteristics in the target stimuli (10). In the color word Stroop task (11), the responding process is formed according to the task-related information in the presence of distractors. Through the Stroop task two differential mind representations are competing, and the response should be for relevant stimuli. Specifically, the color is requested to be detected while mentally eliminating the words and vice versa. Naturally, word reading is more automatic, inhibitory control must override the automatic response to the word. Slower responses are recorded when the word competes with the color of the word (incongruent condition, the word blue written in green ink) than when it is not (congruent condition: the word blue written in blue color) (12).
Since the scalp EEG and event-related potentials (ERPs) allow the real-time demonstration of the neural underpinnings, simultaneous Stroop task and EEG recording might yield further insights (13). Evidently, the representation of the Stroop effect-related incongruity evokes distinct fluctuations in the EEG signal that could statistically be discriminated from the constituents generated within the congruent trials. The brain pulses of the incongruent stimuli might phenomenally originate from neuronal activities aimed at classifying and inhibiting the irrelevancy throughout the task performance. To date, each individual design has considered one or more sections of the associated signal to elucidate the neural correlates of conflict monitoring. Among them, the temporal interval between 300 - 500 ms has gained much significance as the most supported finding. This period comprises cognitive control domains and mainly negative deflections (called N450) (14-19), while other sections have also had roles in mismatch and interference processing (the earlier segments encompassing 150 - 300 ms). Subtraction of the congruent-related epoch signal from equivalent incongruent-associated epochs is assumed to produce a differentiated signal that encompasses the inhibition of task-irrelevant information and irrelevance detection effects. Additionally, within the frequency domain of the EEG signal, the Delta band has also shown some relevance to the task-related inhibition. It has been indicated that midfrontal delta could be a potentially selective marker of motor inhibition (20).
2. Objectives
Current computational modeling of the Stroop task has confirmed that color and word processing are performed via the non-dependent pathways that unite on a final constrained response (13). Machine learning-based classification methods have recently been applied to draw possible distinctions between task performance states and underlying neuronal constituents (21, 22). Hence, we used a machine learning-based classification to examine whether earlier or later epochs are more representative. So, through the present preliminary investigation, Gaussian SVM models were trained on the early (150 - 300 ms) and late (350 - 500 ms) intervals.
3. Methods
3.1. Subject Characteristics
Ninety-six trials were recorded from nine healthy persons (one left-handed and six right-handed based on the Edinburg Handedness Inventory Scores) in the age range of 20 - 25 years as paid volunteers. The educational level was within the range of 13 - 17 years. Regarding the state of the possible neuropsychiatric disorders, a trained clinical psychologist retrospectively performed a telephone-based unstructured interview (based on SCID-5 platform). One of them reported the existence of current anxiety-related traits (that do not fulfill the criteria of any items of the anxiety category in DSM-5). Accordingly, all subjects were free from neurological disorders (the lack of brain surgery, head traumas, epilepsy, seizure, and brain implant without any current medication, drug, or alcohol use), with normal EEG, and signed written informed consent for participating in the study. A validated Persian version of PANAS (positive and negative affect schedule) was also performed for participants. None of the subjects was familiar with the aims of the research work. The level of English fluency was approximately identical for all. The visual state was normal or corrected-to-normal. Before the experiment, the volunteers received detailed instructions on all the details of the task they would perform. Each participant was fitted with an EEG headset by a trained experimenter and seated comfortably in a lab room.
3.2. Task Design
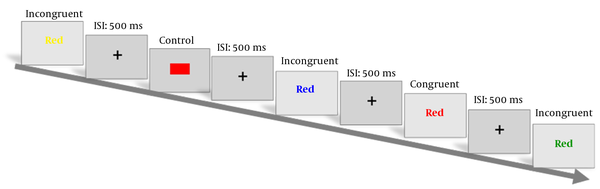
The Stroop task was implemented using Tasc v 4.3 software (http://sarmadtec.com, standard code: IEC62304). Three conditions comprised color-word and neutral stimuli (colored squares and colored rectangular shapes) that presented in a ratio as congruent (80 stimuli), neutral (80 stimuli), and incongruent (120 stimuli). The inter-stimulus interval was constant and set to 0.5 seconds, and the time duration for stimulus presentation depended on the person's response. Ultimately, the task was projected on a 21 inches LCD monitor situated approximately 70 - 75 centimeter in front of the participant and the stimuli were accurately displayed in the center of the screen (Zero degree of visual angle). The overall duration of task performance (based on the pre-task preparations and individual differences in reaction times) was around 40 minutes. The participant chose answers according to the labeled keyboard in front of him/her. The room was shielded without windows and the ambient light was always constant. Also, the environmental temperature kept in a range of 20 - 25 centigrade. Response times and accuracies were collected from behavioral tasks. Figure 1 is displaying the schematic structure of the task.
3.3. EEG Signal Acquisition
EEG data were recorded using a 21-electrode cap (Ag-Cl electrodes) with an Accur8TM amplifier and GET v4.15 acquisition software (http://sarmadtec.com, standard codes: IEC60601-2-26 and IEC60601-1). The impedance of each electrode was kept below 10 kiloohms (kΩ), and the sampling rate was 2 kHz. The environment of electroencephalography acquisition was a sound, EMI-proof, shielded room.
3.4. Analysis
All the steps were performed through Razin V2.0.9 software (http://sarmadtec.com).
3.5. EEG Preprocessing
For each Stroop trial, the first part of the task associated with the training phase was removed from the total signal. Blink removal was applied to all the sessions. A Butterworth filter was used for each channel's EEG data with a lowpass filter at 30 Hz (23, 24). Residual artifacts were denoised manually after the completion of these steps. All three conditions of congruent, incongruent, and neutral were redefined as two conditions: (1) Differentiated incongruent epochs (DIe), which are incongruent and their equivalent congruent epochs were subtracted from and (2) Neutral epochs, in which all epochs were selected from correctly answered trials. Within these epochs, two intervals of 150 - 300 ms and 350 - 500 ms post-stimulus were extracted and for each interval, the aforementioned conditions (differentiated incongruent and neutral) were categorized in two classes. Whole duration (150 ms) of each epoch was used as a variable to be compared between conditions. Additionally, EEG delta band power was calculated for classes in each interval using the Welch method.
The SVM algorithm was applied. SVM could provide the supervised classification system with a distinction based on the maximum margin hyperplane. Currently used SVM is a non-linear version with a Gaussian kernel. Cross-validation and learning curves were also applied to assess the fitting state (25, 26). The SVM classifier was chosen because of the acceptable performance, considerable robustness concerning the curse of dimensionality, and appropriate dealing with outlier values.
4. Results
An independent-samples t-test was conducted to compare negative affect for male and female. There was not a significant difference in the scores for male (M = 15.55, SD = 3.995) and female (M = 16.59, SD = 5.156); t (89) = -1.066, P = 0.289. Comparing Positive Affect for male and female yielded an insignificant difference in the scores for male (M = 34.10, SD = 8.384) and Female (M = 32.80, SD = 8.271); t (89) = 0.742, P = 0.460.
4.1. Individual and Pooled Event-related Epochs and Topographic Maps
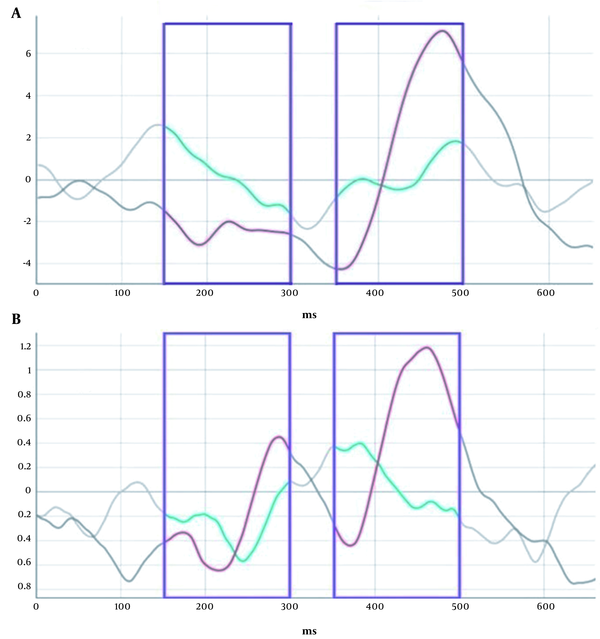
The signal epochs associated with the two intervals, early 150 - 300 ms and late 350 - 500 ms after stimulus presentation, were considered and classified for all seven participants as two classes of DIe and neutral, as depicted in Figure 2. Within each interval, two colored lines are related to the DIe and neutral conditions. There is a significant separation between these two states, within either early or later parts, regarding both configurational characteristics and the overall amplitude. Then, all the individual signals were pooled together for a more holistic representation (Figure 2B). As illustrated, two differentiable sections of the signal can be seen for this task, and the previously mentioned difference between the two conditions is also observed when pooling the data of all participants.
The averaged event-related signals of participants. (A) Temporal intervals for the early mismatch detection and late irrelevance processing are highlighted in purple. Within each selected epoch, two signals are observed; red demonstrates the neutral trials, and green is associated with the differentiated signal. (B) All individual pooled temporal intervals for the early mismatch detection and late irrelevance processing.
4.2. Pooled Event-related Scalp Distribution of the Delta Band Power
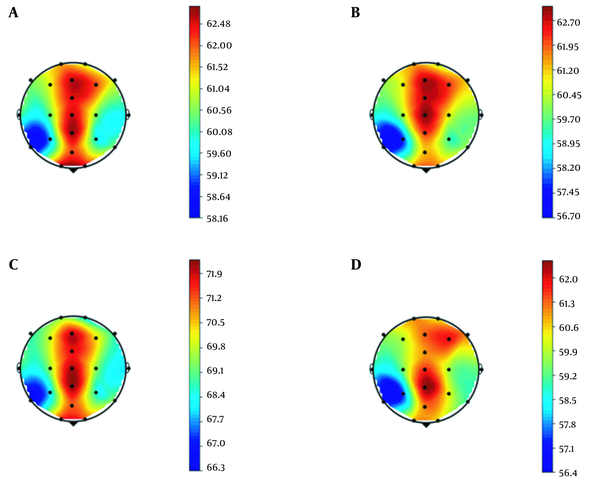
The band powers associated with the delta frequency were calculated for the two signal intervals. Figure 3 illustrates the pooled scalp distribution of the power values of the delta frequency band for two conditions, one for the neutral condition and one for DIe. As seen in the topographic map for Delta Power, there is no significant difference between the two conditions at different time intervals. Also, most of this activity is in the central areas, which might indicate the individual's efforts for motor response.
There was no statistical difference between the two states considering two temporal intervals.
4.3. Classification Method Values
The SVM model was trained with epochs from intervals of interest for two classes in DIe and neutral conditions. The whole epoch was considered as the input for the SVM model. Table 1 depicts the final results of the classification step. The table shows the sensitivity, specificity, and accuracy based on the obtained confusion matrix for three out of 96 example trial. Based on the classification results, the late interval performed better (P-value = 0.0731, F = 3.855) in classifying DIe and neutral conditions, suggesting strong correlation between irrelevant stimulus suppression and selective attention and the late 350 - 500 ms interval.
| Example Trials | 150 - 300 ms | 350 - 500 ms | ||||
|---|---|---|---|---|---|---|
| Specificity | Sensitivity | Accuracy (%) | Specificity | Sensitivity | Accuracy (%) | |
| T17 for S 04 | 0.68 | 0.66 | 67.19 | 0.68 | 0.7 | 69.34 |
| T35 for S 09 | 0.73 | 0.72 | 72.68 | 0.72 | 0.74 | 72.68 |
| T62 for S 05 | 0.70 | 0.74 | 72.26 | 0.84 | 0.8 | 82.51 |
| Average (for all trials) | 66.54 | 71.76 | ||||
Sensitivity, Specificity, and Accuracy Obtained Through Classification Model for Three Example Trials Out of Total 96 Trials a
5. Discussion
Selective attention is the ability to restrict cognitive processing and responses to a specific, task-relevant subset of present stimuli while excluding other attributes. EEG recording during the Stroop task, as one of the reliable and valid methods for representing goal-directed suppression, could reveal the neural correlates of ongoing cognitive processing.
The SVM model classified two distinct intervals within the event-related signal with acceptable accuracies. Different event-related potential studies and resting-state analyses (time domain, frequency domain, correlational, and associational approaches) have been done to provide more elucidations. These investigations have yielded inconsistent findings. In a very early study, Duncan-Johnson and Kopell showed that despite a significant distinction between reaction times of the congruent and incongruent paradigms, there is no remarkable difference in various dimensions, latency, or amplitude measures of the P3 component evoked by the congruent, neutral, or incongruent conditions in the Stroop test (27). The present findings support this result. These observations led to the conclusion that the interference effect seen in the reaction time is not the consequence of early neural processes. Other researchers reported that negativity in the signal around 400 ms after stimulus presentation could be considered a reliable and replicable finding that efficiently dissociated different conditions of the Strop task. This event-related segment (350 - 500 ms) has been widely reported to reflect neural correlates for semantic mismatch, violation detection, and processing (28).
Additionally, a negative deflection called the N270 component was considered the semantic and non-semantic discordance detector in response to non-anticipated stimulus location, arithmetic aspects, digit color or magnitude, shape, or even gender mismatch on different modality platforms (28, 29). A few studies showed some uncertainty about clearly dissociating the N400 and N2b ERP waves. There have been suggestions that N400 is a delayed member of the N2 waves (30, 31). Present confined findings partially support the existence of different patterns and profiles.
Current frequency assessments did not reveal significant results regarding power values or scalp distribution patterns. The frequency bands and values of the spectral powers have long been considered appropriate and relevant tools for EEG assessments and analyses within the rest or task conditions. Various lines of evidence imply the specific contributions of delta and theta oscillations in targeted inhibition for more efficient processing of task-relevant features and precise responses. Knyazev et al. confirmed that the functional delta frequencies seem important in synchronizing brain activity with autonomic functions, motivational functions, higher emotional engagement, cognitive operations of attention, and the identification of motivationally salient stimuli in the related context (32-34).
Regarding the resting-state signals, the frequency band of 13 - 20 Hz was sensitive to differentiate between the congruent and incongruent conditions according to the coherence measurement (35). One emotional Stroop task demonstrated that interference with threatening words could be associated with self-reported attentional inhibition capacity and frontal delta–beta coupling. Other EEG-related studies also reported a clear distinction between different states of the Stroop task and resulting semantic interference regarding EEG data (36-39). Moreover, lines of evidence confirm the discriminatory role of electroencephalographic items during rest for differentiation between conditions and states of mental health (40). Additionally, robust correlations in a study indicated that participants with stronger resting-state-related left-lateralized activity in different prefrontal regions were more likely to suppress intrusive attributes and confirmed that neurophysiological dissociations might provide explanations for interindividual variations (41).