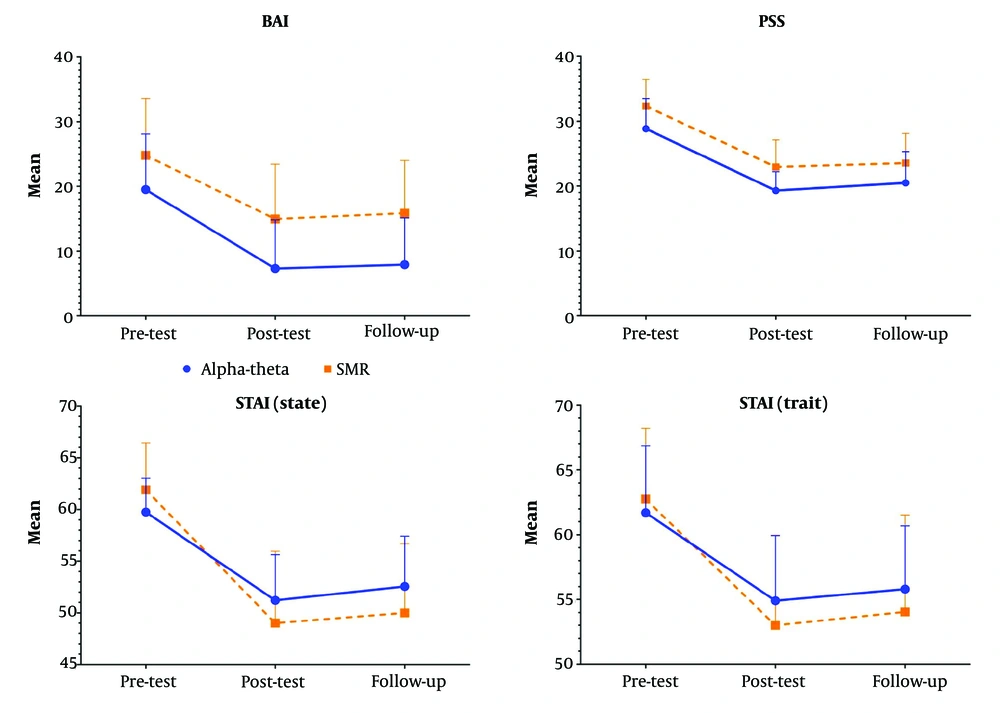
In this study, we evaluated the effectiveness of two EEG neurofeedback protocols (AT and SMR) in reducing anxiety symptoms and perceived stress among individuals with GAD. Participants underwent 15 sessions of either the AT protocol at Pz or the SMR protocol at Cz. The results demonstrated a significant reduction over time in BAI, state anxiety, trait anxiety, and PSS scores, confirming the overall efficacy of both neurofeedback protocols in alleviating anxiety symptoms. For state anxiety, there was a significant time × group interaction, implying that the reduction patterns differed between the two protocols. Although both groups experienced a decrease in state anxiety over time, the SMR group showed a slightly greater immediate reduction in the post-test compared to the AT group, suggesting that SMR training may have a more immediate impact on momentary anxiety states.
Regarding trait anxiety, both groups exhibited a significant reduction over time, but no significant group differences or interaction effects were observed, suggesting that both protocols were equally effective in reducing long-term anxiety traits. These findings suggest that while both neurofeedback protocols effectively reduced anxiety symptoms and perceived stress, the SMR protocol might provide more immediate relief for state anxiety. Future studies incorporating objective neurophysiological markers are needed to further validate these findings and explore the underlying mechanisms of each protocol.
Results indicated that the AT protocol significantly reduced anxiety levels. These findings align with Dadashi et al.'s research, which demonstrated that enhancing alpha waves can alleviate symptoms of GAD (
20). After EEG neurofeedback training to boost AT activity, individuals with GAD experienced expected decreases in PSS, STAI, and BAI scores. Notably, our study observed a similar pattern in the reduction of BAI scores across both protocols. However, more detailed investigations using objective tools are needed to assess brain activity following each protocol and to better understand the specific effects of each protocol on brain function.
Anxiety disorders are frequently triggered and maintained by biases in processing threat-related information. This suggests that the attention system of anxious individuals is more sensitive to threat-related than to neutral stimuli. These biases play a crucial role in the onset and continuation of anxiety disorders like GAD (
19,
38). Mansell's top-down model of processing biases in anxiety suggests that the anterior cingulate cortex, lateral prefrontal cortex, and parietal cortex play key roles in regulating attention (
39).
Recent functional magnetic resonance imaging (fMRI) studies have identified three distinct attentional networks. The first is the alerting network, which is activated in the frontoparietal cortex and thalamus. The second is the orienting network, characterized by high activity in the superior parietal region and temporal-parietal junction, with a bias towards the right hemisphere. Finally, the executive control network is activated in the anterior cingulate and both right and left frontal areas (
40).
Building on this model and its findings, Hou et al. demonstrated the effectiveness of EEG neurofeedback training targeting the parietal lobe in enhancing attention control (
19,
41), and our findings support the efficacy of this protocol in GAD patients. However, since we did not conduct a biological evaluation in our study, we cannot be certain whether the results obtained are due to changes in the activity of brain networks or other factors. This statement should help to clarify the limitations of our study for the academic journal.
Group two, undergoing the SMR protocol, demonstrated a significant decrease in BAI, PSS, and STAI scores. The SMR training regimen is known to improve attention (
42). This occurs when an individual is still yet alert, supported by brain activity in central scalp regions (
43,
44). Research has validated the efficacy of a 12 - 15 Hz frequency range in anxiety management (
23). Nonetheless, its extended duration, delayed impact, and variability among individuals limit its clinical application (
22,
45,
46).
The SMR EEG neurofeedback has proven effective in treating children with attention-deficit hyperactivity disorder (ADHD), and it has also shown positive effects on the attentional performance of healthy individuals (
25,
47,
48). One possible explanation for the effectiveness of SMR EEG neurofeedback training is that it functions as a bottom-up mechanism within the thalamic-cortical circuitry, enhancing its inhibitory processes. By increasing SMR, this training may boost the brain's ability to filter out irrelevant sensory information, leading to improved somatosensory processing (
25,
43).
Moreover, SMR training may stabilize vigilance by regulating the locus coeruleus noradrenergic system. Its activation has been demonstrated to affect the sleep spindle circuit (
49). Research has shown that engaging in attention-focused practices, such as yoga and meditation, can be highly effective in reducing symptoms of anxiety (
50,
51). In addition, in clinical settings, lack of focus is a common anxiety symptom. Therefore, it was hypothesized that improving attention-related SMR activity could alleviate anxiety (
52). Our study supports the effectiveness of this protocol for anxiety, but further research is necessary to assess its impact on brain function.
The SMR training appears to enhance cortical regulation and promote a state of focused calm. This mechanism likely leads to more immediate reductions in physiological arousal and, consequently, state anxiety. In contrast, the AT protocol — designed to facilitate a hypnagogic state and promote long-term mood and creativity changes — may not be as effective for rapid anxiety relief. The more direct modulation of arousal-related neural circuits via SMR training might therefore explain its superiority in alleviating state anxiety during the intervention (
25,
27,
53).
It should be noted that this study had some limitations. Only university students were recruited for this study, allowing for a more homogeneous sample and controlled variables. However, this may limit the generalizability of our findings to a broader population with more diverse demographic and clinical characteristics. The EEG neurofeedback training usually involves more sessions; however, our participants could not undergo additional sessions because neurofeedback is not considered a first-line treatment. Despite obtaining informed consent from the participants, hospital policy restricted us from enrolling participants in longer training. Increasing the number of sessions could potentially reduce the differences between the two protocols.
Additionally, a significant limitation of this study was the inability to include a placebo group, as we could not exclude patients referred to the treatment center, which made it challenging to establish a proper control group. The lack of a placebo group may have impacted the ability to fully isolate the effects of the treatment from other factors. Furthermore, this study relied solely on subjective data, which can be influenced by participant biases and perceptions.
Future research should incorporate objective data collection methods, such as EEG analysis, to provide a more comprehensive understanding of the effectiveness of different neurofeedback protocols. Using objective measures will not only enhance the reliability of the findings but also contribute to more robust conclusions. Prioritizing objective data will significantly advance the field, improve the validity of research outcomes, and better inform treatment strategies.
5.1. Conclusions
The results indicated that both the AT and SMR EEG neurofeedback protocols were effective in reducing GAD symptoms. Notably, the SMR protocol was superior in reducing state anxiety, while improvements in other variables were similar between the two protocols. Given the high treatment costs associated with neurofeedback, understanding the distinct effects of each protocol may help optimize cost-effectiveness and enhance treatment outcomes by guiding the selection of the most appropriate approach based on individual symptom profiles.
In clinical practice, these findings suggest that SMR neurofeedback may be particularly beneficial for individuals experiencing situational or performance-related anxiety, while both protocols remain viable options for broader anxiety symptom reduction. Future studies should explore the integration of neurofeedback with other therapeutic modalities and incorporate objective measures such as neuroimaging, heart rate variability, and other physiological markers to further elucidate the neural mechanisms underlying the efficacy of these protocols and validate these results.