1. Background
Every year, 14.9 million (more than 1 in 10) children are born prematurely all over the world (1). Following the advances in perinatal care, an increasing number of preterm infants reach childhood (2). Negative effects of preterm birth on neurodevelopmental and psychiatric development of children and adolescents have been well documented (3-8). Severe neurological impairment has been reported in 10 to 12% of school-aged children, who were born very preterm (less than 32 weeks of gestation) (9-12). Mild neurocognitive disorders such as lower IQ and poor academic achievement have been reported more frequently (4, 13-18).
Attention deficit hyperactivity disorder (ADHD) is a neurobehavioral syndrome characterized by attention deficit, impulsivity and hyperactivity. It can be considered a generalized impulsivity disorder, with the traits of impulsivity manifesting at the motor, emotional, social, and attention levels (19). This disorder was described more than a century ago, as a behavioral disorder seen more often in boys. Attention deficit hyperactivity disorder is the most common neurodevelopmental disorder in Western countries (17) with a reported prevalence rate of 3 to 9% and a male to female ratio of 3:1 to 5:1 in previous studies (20-22). Investigations by the American Psychiatric Association, which were published in the Diagnostic and statistical manual of mental disorders 4th edition (DSM-IV), showed a prevalence of 3 to 5% for ADHD in adults with an equal male-female ratio (23-25).
The exact etiology of ADHD is still unknown, although many factors have been suggested as risk factors for the development of this disorder, including: preterm birth, low birth weight, genetic factors, environmental factors such as contact with toxic substances like lead, alcohol, and Poly-Chlorinated Biphenyl (PCP), inappropriate socioeconomic status, and perinatal complications such as intrauterine growth retardation and perinatal asphyxia (16, 26-33).
Since 1961, many improvements in devices and care have taken place. Although with the invention of neonatal intensive care unit, infant mortality rates, especially among preterm and low birth weight infants have been significantly reduced, these babies face many complications. These infants are under pressure and irritation, due to their specific condition. Moreover, they are influenced by potentially harmful environmental factors such as light and sound, magnetic fields, radiation, inactive ingredients of drugs and chemical substances. Premature infants are more vulnerable compared with older children and adults due to the evolution of organ systems, and are less able to deal with the problems of life outside the uterus (34).
Several studies have reported a relationship between prematurity, low birth weight (which are the leading causes of hospitalization in NICU) and the incidence of ADHD during childhood and adolescence (4, 16, 33, 35, 36). The question now is whether NICU hospitalization can increase the risk of ADHD. No study has ever investigated the relationship between NICU hospitalization and development of ADHD in the future, as an independent factor.
2. Objectives
The aim of the current study was to investigate the effects of hospitalization at the NICU, independent of other variables such as preterm birth and low birth weight, on the incidence of ADHD.
3. Materials and Methods
This study was a retrospective cohort study. Regarding the prevalence of ADHD in previous studies, the sample size needed for the study was calculated as 100 samples in the group with a history of NICU hospitalization and 100 samples in the group without a history of hospitalization.
Six to seven-year-old children with a history of neonatal intensive care unit hospitalization in Amirkola children’s hospital were included in the exposed group of our study. We asked parents about the child’s history of NICU hospitalization and then checked the hospital archives. Samples in the unexposed group were preschool children without a history of hospitalization, who were selected randomly among Babol’s preschool centers. Exposed and unexposed groups were matched for age. Children hospitalized in the NICU because of severe congenital defects, chromosomal anomalies, maternal drug abuse, parental separation, serious injuries during birth, and intractable seizures were excluded from the study. High and low birth weight infants (> 6 and < 3 standard deviation from the Marsal’s growth chart) (37), who could lead to a possible coding error (38), were also excluded.
After initial sample selection, the mothers of these children were contacted and sampling was continued by obtaining informed consent to reach the sample volume calculated. Finally, our study population consisted of 200 preschool children, who were born to Iranian mothers.
To diagnose ADHD, the standard checklist of DSM-IV was used. The DSM criteria were used for clinical-medical studies, and neurological evaluations were performed for diagnosis of ADHD by a psychiatrist (39). Other information such as gender, weight, birth weight, and gestational weeks at birth, delivery type, and days hospitalized in the NICU were collected and finally all the data were entered into the statistical analysis software.
The data from the study was analyzed using SPSS v.20. First, descriptive statistics was used to summarize the data. Then, appropriate statistical tests (chi-square, t-test and Mann-Whitney) were performed to estimate the association between studied variables and ADHD incidence. Finally, to estimate the impact of NICU hospitalization on the incidence of ADHD as the outcome variable, unconditional logistic regression was used to calculate odds ratio with 95% confidence interval. Since risk ratio should be reported in cohort studies, odd ratio was converted to risk ratio (40, 41).
4. Results
Amongst a total of 200 children, 103 were boys (51.5%) and 97 children were girls (48.5%) (P = 0.038). In terms of age, 49.2% of children were six years old, and 50.8% were seven years old (P = 0.15).
The results showed that 33.5% of children had one of three subtypes of ADHD (ADHD-I: inattention disorder, ADHD-II: hyperactivity and impulsivity disorder, ADHD-C: combined disorder). The incidence of ADHD-C (20.5%) was more than the two other subtypes (6.5% and 6% in ADHD-I and ADHD-II, respectively).
The incidence of ADHD was 27% in children with a history of NICU hospitalization and 14% in children with no history of NICU hospitalization. Among children with a history of NICU hospitalization, 23.9% had low birth weight (< 2500 gram) and 30.8% were preterm (< 37 gestational weeks).
Table 1, summarizes the association between study variables and ADHD in preschool children. Fischer’s exact test showed that the incidence of ADHD is more frequent in boys (22%) than girls (19%) (P value = 0.038). The test also showed that there was no significant difference between six- and seven-year-old children in terms of ADHD (P value = 0.23). Also, there was no statistically significant association between delivery type and ADHD (P value = 0.55).
| Variable | Frequency in Children With ADHD, % | P Value |
|---|---|---|
| Age, y | 0.150 | |
| 6 | 38.6 | |
| 7 | 30.6 | |
| Gender | 0.038 | |
| Boy | 22 | |
| Girl | 19 | |
| Delivery Type | 0.873 | |
| C-section | 21 | |
| Vaginal | 20 | |
| Low Birth Weight | 0.000 | |
| Yes | 34 | |
| No | 16 | |
| Preterm Birth | 0.005 | |
| Yes | 31 | |
| No | 16 | |
| NICU Hospitalization | 0.001 | |
| Yes | 27 | |
| No | 14 |
To assess the association between gestational age and ADHD, Fischer’s exact test was used and the results showed that the incidence of ADHD is significantly more frequent in premature infants (31%) than mature infants (16%) (P value = 0.002). T-test revealed a statistically significant association between gestational weeks and the incidence of ADHD (P value = 0.00). The average gestational age was 35.49 ± 3.84 weeks in children with ADHD and 36.78 ± 2.31 in children without ADHD (Table 2).
| Variable | Average and Standard Deviation Values in Children | P Value | |
|---|---|---|---|
| With ADHD | Without ADHD | ||
| Current Weight, kg | 21.8 ± 4.4 | 21.3 ± 4.8 | 0.461 |
| Birth Weight, kg | 2.658 ± 0.861 | 2.937 ± 0.684 | 0.852 |
| Gestational Age, w | 35.49 ± 3.84 | 36.78 ± 2.31 | 0.001 |
| Days Hospitalized in NICU, d | 6.31 ± 5.38 | 3.11 ± 4.59 | 0.000 |
Evaluation of the association between birth weight and incidence of ADHD using T-test showed that there was no statistically significant relationship between these variables (P value = 0.85). By categorizing the infants to low and normal birth weight and using Fischer’s exact test, there was a significant association between low birth weight and ADHD (P value = 0.003). The prevalence of ADHD was 34% and 16% in children with low birth weight and normal birth weight, respectively. Current weight of children had no significant association with ADHD (P value = 0.46) (Table 1).
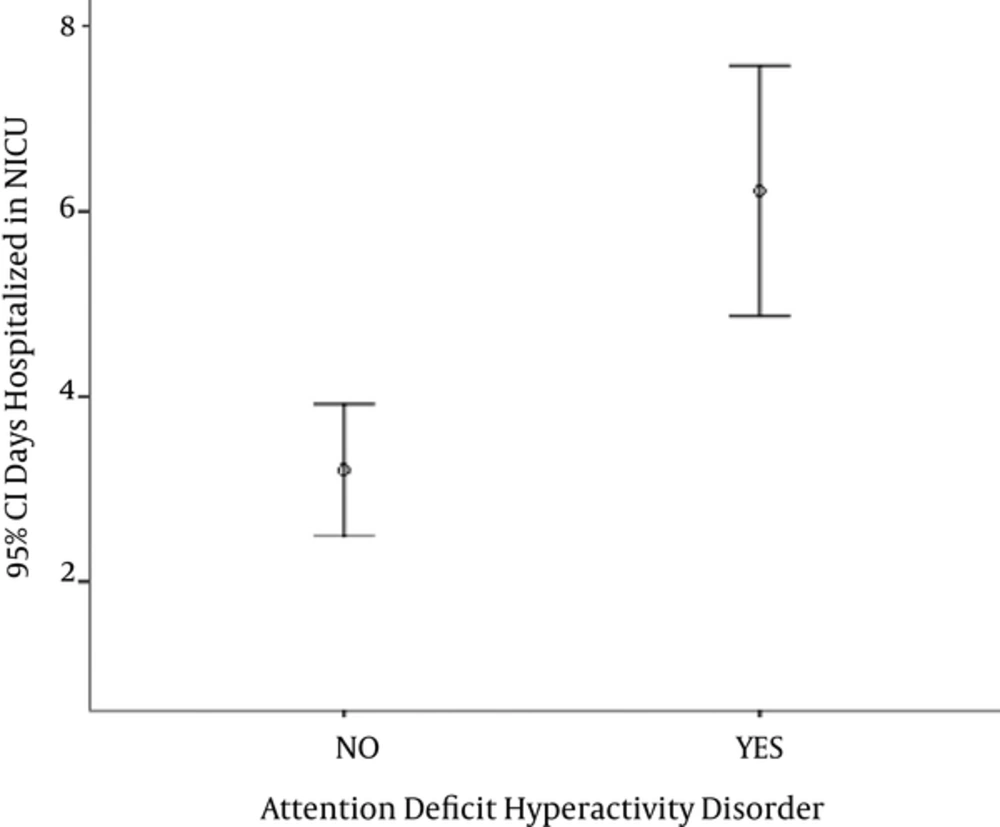
As mentioned earlier, the prevalence of ADHD in children with a history of NICU hospitalization (case group) was more than children without such a history (control group). To evaluate the association between history of NICU hospitalization and incidence of ADHD in preschool ages, Fischer’s exact test was initially used. The results revealed a significant difference between the two groups (P value = 0.00). The average NICU hospitalization days were 6.31 ± 5.38 and 3.11 ± 4.59 days in children with and without ADHD, respectively (Figure 1). The association between days hospitalized in NICU and the incidence of ADHD was evaluated using the T-test. The results showed a statistically significant association between days hospitalized and ADHD (P value = 0.01) (Table 2).
Unconditional logistic regression (backward stepwise) was used to determine the relationship between the incidence of ADHD and NICU hospitalization (Table 3). This regression showed that the days hospitalized at the NICU and gender was associated with ADHD in preschool children.
| Variable | Risk Ratio | CI 95% | P Value | |
|---|---|---|---|---|
| Lower | Upper | |||
| Age (6 or 7) | 1.26 | 0.614 | 2.428 | 0.570 |
| Gender (boy or girl) | 1.16 | 0.252 | 0.919 | 0.027 |
| Current Weight | 1.00 | 1.000 | 1.000 | 0.427 |
| Delivery Type | 1.05 | 0.427 | 1.682 | 0.636 |
| Preterm Labor | 1.94 | 0.674 | 2.904 | 0.368 |
| Low Birth Weight | 1.00 | 1.000 | 1.000 | 0.719 |
| History of NICU Hospitalization | 1.93 | 0.397 | 3.197 | 0.824 |
| Days Hospitalized in NICU | 1.18 | 1.044 | 1.292 | 0.006 |
5. Discussion
In this retrospective cohort study, which was performed on 200 preschool aged children, the independent relationship between NICU hospitalization and the incidence of ADHD was tested. The prevalence of ADHD among children with a history of hospitalization in NICU and children without such a history was 27% and 14%, respectively. The difference between the two groups was statistically significant. Neonatal intensive care unit hospitalization makes the risk of developing ADHD in preschool ages 1.16 times greater. Therefore, after a week’s stay at NICU, the risk of ADHD at preschool ages becomes 1.93 times greater.
Several studies suggested that attention problems are more frequent among children born preterm. Farooqi et al. (33) found that eleven-year-old children born after 23 to 25 weeks of gestation develop ADHD, three to four times more than term infants (after 37 weeks of pregnancy). However, Stjernqvist and Svenningsen (16) stated a two-fold increase in a study carried out on ten-year-old children, who were born before 29 weeks of gestation. A recent French study on 1102 five-year-old children born between 22 and 32 weeks of gestation showed a two-fold risk of ADHD compared to the control term group (42). Follow-up studies of ADHD in preterm infants are concentrated with some exceptions on children, who were born very preterm and have received care in NICUs (4), and no study has investigated the difference in the incidence of ADHD between children with a history of NICU hospitalization and those without such a history.
There are evidences of developmental delay in children with ADHD (43). The delay has also been recognized using imaging techniques. Children with ADHD reach the maximum cortical narrowness three years later than healthy controls (44). It has been hypothesized that the effects of perinatal factors become evident during the development (45). It seems reasonable to assume that such factors are effective on development of ADHD symptoms in children, who are born preterm.
The current study is the first to report the impact of NICU hospitalization independent of factors such as preterm labor and low birth weight. A large number of infants hospitalized at the NICU are born preterm, the brain connections of whom are still not fully formed. The maximal amount of cortical connections refinement took place in weeks 24 to 28 of pregnancy. The corrected mean number of neurons reaches a maximum at 28 weeks of gestation, and estimated number of neurons is significantly higher at 28 to 32 weeks of gestation than at 17 to 20 gestational weeks and at 0 to 13 postnatal weeks (45, 46). As a result, infants who are admitted to the NICU are more vulnerable to environmental harmful factors affecting the neurobehavioral systems.
It can be concluded from the results of this study that NICU hospitalization increases the possibility of ADHD in the future, independently. This finding, which needs to be confirmed by further studies with greater sample sizes in a multi-central fashion and by including more mediator/confounding variables such as behavioral and socioeconomic factors related to parents, indicates the necessity of reducing possible damaging elements in the NICUs. The answer to the question of how neonatal intensive care unit can cause developmental damages requires comprehensive researches.