1. Background
Impairments in visual processing have been reported in patients with schizophrenia, ranging from abnormalities in simple visual discrimination to high level visual perception tasks (1). It is suggested that certain visual impairments may contribute to cognitive deficits and poor functioning of the patients (2). A recent study found that disturbance in visual processing could be used as a predictive factor for development of the disorder among high risk individuals (3). Moreover, compared to other forms of sensory impairment, visual abnormalities in the general population are correlated with later development of schizophrenia (4). Given that the visual system has been extensively explored and is best understood through neuroscience, examination of visual processing in schizophrenia may clarify the impaired neural circuitry contributing to the development of this disorder.
It is well established that during early visual processing, each cerebral hemisphere only receives information from the contralateral hemifield (5), and as visual information moves up the hierarchy, more complex processing is achieved (6). Double simultaneous stimulation (DSS), in which 2 stimuli are presented at the same time in both visual fields, is a method of testing afferent visual pathway for signs of unilateral brain damage (7). In this regard, some studies have suggested that visual processing has a limited capacity to process multiple objects concurrently (8). In fact, when 2 or more stimuli are presented, competing stimuli require continuous prioritization of visual attention and locus-specific processing (8). Another theory assumes that the 2 hemisphere might cooperate instead of competition in order to process bilateral targets, which is known as ‘bilateral advantage’ (9). The present study aimed at examining visual processing during bilateral compared to unilateral visual stimulus presentation in patients with schizophrenia.
The right posterior parietal cortex (PPC) plays an essential role in spatial attention, preferentially processing the left hemispace in healthy individuals (10). Previous studies have examined visual processing of ipsilesional and contralesional, as well as bilateral simultaneous stimuli presentation in patients with unilateral parietal damage, and suggested distinct neurological syndromes, namely hemispatial neglect, extinction, and anti-extinction (11-16). While in patients with hemispatial neglect fail to detect contralesional stimuli, in extinction they are able to detect a single stimulus presented to either visual field, but fail to respond to the presentation of bilateral simultaneous stimuli. Opposite to extinction effect is anti-extinction, in which patients are able to detect two stimuli presented simultaneously, yet show impaired attention to contralesional stimulus when no competitive ipsilateral stimulus is present (11-19). While some studies have suggested that extinction is a mild form of neglect, others proposed that the two syndromes can occur independently (20).
It should be noted that neuropsychological and neuroimaging studies have demonstrated disruption of the parietal association cortex in patients with schizophrenia (10). Particularly, researchers have reported evidence of visual and representational hemispatial neglect in these patients, as evidence for parietal lobe dysfunction (21-24). However, to date no study has examined extinction and anti-extinction in patients with schizophrenia. The present study, aimed at comparing performances of patients on detection of single and double visual stimuli to obtain further insight to specific visual processing deficits in schizophrenia.
2. Objectives
In order to elucidate visual processing impairment in schizophrenia, 2 simple detection tasks were designed, since tasks, which require only target detection are less attention-demanding than tasks involving stimulus identification, localization or orientation (25). This experimental allowed: 1, distinguishing differences in visual processing of unilateral versus bilateral displays; and 2, examined differences in task-related performance patterns among patients with schizophrenia when compared to controls.
3. Materials and Methods
3.1. Participants
A group of 27 patients with schizophrenia (20 males) were recruited from outpatients of a psychiatric hospital. All patients met the diagnostic and statistical manual for mental disorders fourth edition (DSM-IV) criteria for a lifetime diagnosis of schizophrenia. They were assessed using the positive and negative syndrome scale (PANSS) for schizophrenia (26). Patients continued their antipsychotic medications throughout the assessment. The mean chlorpromazine (CPZ) equivalent dose was 369.2 mg (27). Patients did not have a history of electroconvulsive therapy during the 6 months prior to the testing time.
The control group comprised of 27 individuals (20 males), which were recruited from university staff and their relatives, screened for a personal and family history of psychotic illnesses. The one-to-one individual matching principle was used and the 2 groups were matched for age, gender, and educational level. Exclusion criteria for all participants included a history of head injury, neurological disorders, and current substances abuse. All participants were right-handed and had normal or corrected-to-normal vision. Written informed consent was obtained from all participants, and the study was approved by the ethics committees of Kerman University of Medical Sciences.
3.2. Experimental Procedure
The stimuli in both tasks were white shapes (circle, star, and triangle) presented on a black computer screen. Each trial was started with the presentation of a fixation point for 500 ms at the center of a computer screen. Then, shapes briefly appeared (70 to 140 ms) in one or two locations at the corners of an imaginary square centered around the fixation point. There were 3 types of display: unilateral-left, unilateral-right, and bilateral trials with identical display probability. The time between the onset of the 2 trials was 2.5 or 3 seconds. Participants sat 60 cm from the computer. They were asked to fixate at the center of the screen and to press the left key for unilateral and the right key for bilateral trials as quickly and accurately as possible, with a counterbalanced order. Both tasks were detection task and response time and error rate were registered via key press.
In the circle task, the target stimulus (circle) could be presented either unilaterally as 1 circle in left/right hemifield, or bilaterally as 2 circles presented, simultaneously. Each display was repeated 40 times resulting in 120 trials.
In the star and triangle task, similar to the previous task, there were 2 types of displays (unilateral and bilateral). Unilateral trials consisted of one shape (star or triangle) presented on the right or left hemifield. On bilateral trials, 2 identical or non-identical shapes were presented simultaneously (i.e. 2 stars, 2 triangles or 1 star and 1 triangle). Each display was repeated 40 times, which resulted in 320 computer randomized trials.
3.3. Statistical Analyses
A generalized linear model (GLM) for repeated measures was used to test for group (schizophrenia vs. controls) differences of the dependent variables (error rate and response time) across the 3 trials (unilateral-right, unilateral-left, and bilateral) (within-subject factor). Significant main effects, interaction, and follow-up pairwise comparisons were examined after adjustment for multiple testing (Bonferroni). A combination of chi-square and t-test was used on demographic and clinical data. The alpha level was set at 0.05 for all the statistical tests performed. All analyses were conducted using the SPSS 18.0 software.
4. Results
Table 1 presents the clinical and demographic characteristics of patients and controls. The 2 groups were well matched for age, gender, education, and hand preference.
| Variables | Patients | Controls | P Value |
|---|---|---|---|
| Age, y | 33.8 (8.2) | 34.4 (8.8) | NS |
| Educationa, y | 11.2 (2) | 11.7 (2.2) | NS |
| Sex-N, % males | 20 (65) | 20 (65) | NS |
| Edinburgh | 98.6 (7.2) | 0098.3 (5.4) | NS |
| Length of illness, y | 12.3 (6.3) | ||
| Age onset of illness, y | 21.4 (5.9) | ||
| Mean chlorpromazine | |||
| equivalent, mg | 369.2 (151.5) | ||
| PANS (+) | 8.1 (4) | ||
| PANS (-) | 0011.6 (6.9) |
Abbreviations NS, Non Significant; PANS, Positive and Negative Symptom Scale.
aEdinburgh handedness inventory.
4.1. Circle Task
The mean error rates and response times across the 3 trials (unilateral-right, unilateral-left, and bilateral) were calculated. Tables 2 and 3 show mean and SD of the error rates and response times of the participants.
| Variables | Unilateral-Left | Unilateral-Right | Bilateral |
|---|---|---|---|
| Error rate | |||
| patients | 11.5 ± (17.9) | 18.9 ± (28.9) | 7.1 ± (6.8) |
| controls | 0.8 ± (1.5) | 1.3 ± (1.9) | 3 ± (2.8) |
| Response time | |||
| patients | 512.1 ± 169.9 | 582.4 ± 186.3 | 604.6 ± 169.9 |
| controls | 401.2 ± 73.5 | 461.6 ± 77.8 | 481.8 ± 72.6 |
| Variables | Unilateral-Left | Unilateral-Right | Bilateral |
|---|---|---|---|
| Error rate | |||
| patients | 12.4 ± 21.1 | 17.4 ± 25.2 | 7.7 ± 6.3 |
| controls | 0.4 ± 0.9 | 2.3 ± 2.0 | 0.9 ± 1.0 |
| Response time | |||
| patients | 594 ± 172 | 604 ± 169 | 690 ± 219 |
| controls | 474 ± 78 | 481 ± 72 | 536 ± 79 |
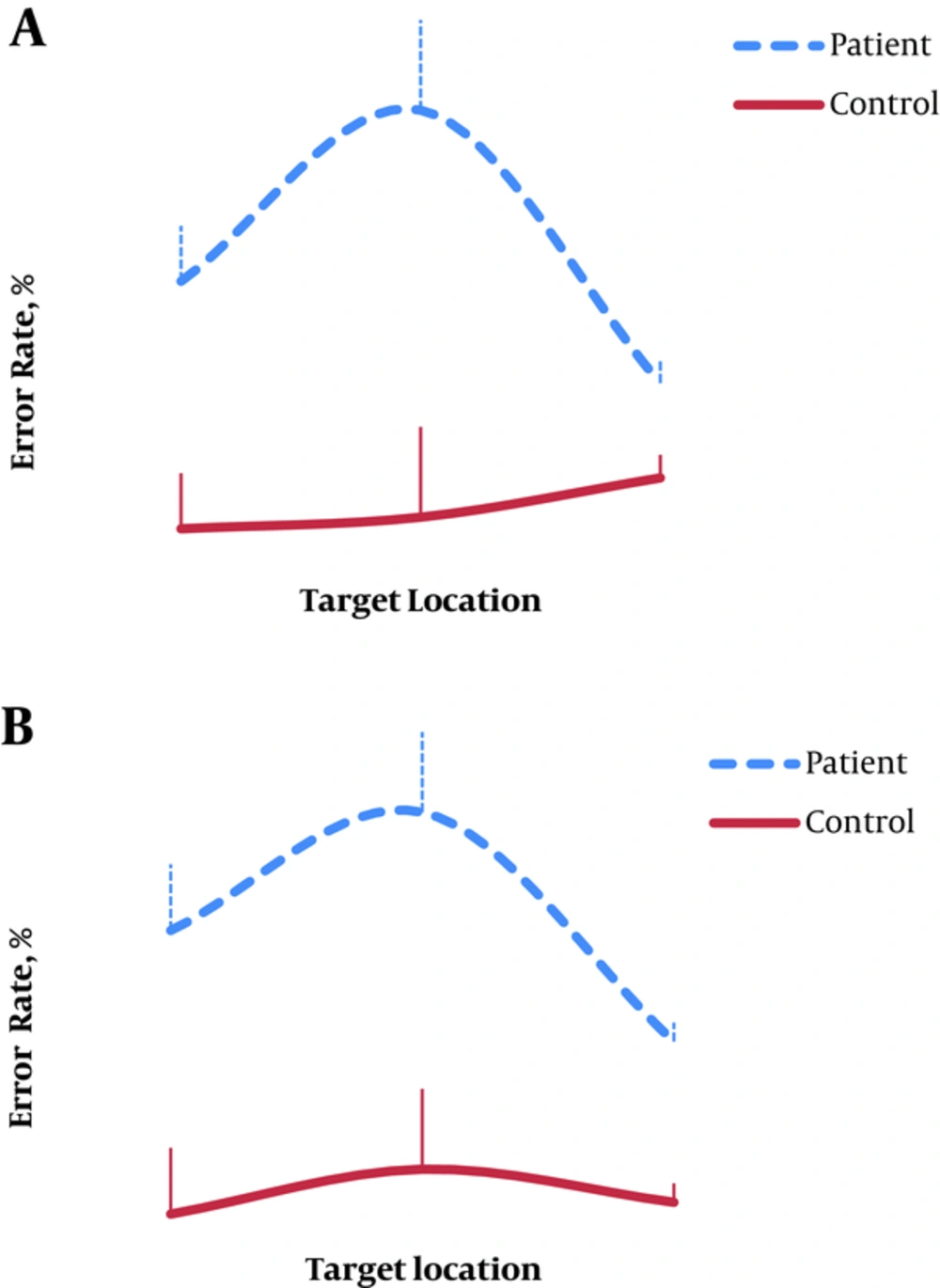
Regarding error rate: the results showed a significant main effect of group, F(1,52) = 11.08, P = 0.002, and η2 = 0.9, showing that the performance of patients was less accurate than that of controls. Pairwise comparisons of the error rates between the 2 groups showed significant group differences for all trials, all P < 0.006, (Table 2). The effect of trial was significant, F(2,104) = 4.1, P = 0.03, and η2 = 0.59, indicating that error rate differed across the 3 trials. Moreover, there was a significant interaction effect between trials and participant group, (F(2,104) = 6.7, P = 0.006, and η2 = 0.8) indicating differential changes in error rates between patients and control groups among the 3 trials. This interaction effect was examined by pairwise comparisons of the mean scores between the 3 trials in each group. The comparisons showed that for controls, error rates were similar across different trials. In contrast, patients had significantly higher error rate on unilateral-right compared to unilateral-left, P = 0.01, and compared to bilateral trials, P = 0.005. Also, they had significantly higher error rate on unilateral-left relative to bilateral trials, P = 0.01, (Figure 1A).
Regarding response time (RT), the effect of group was significant (F(1,52) = 10.9, P = 0.002, η2 = 0.9) showing that patients were significantly slower than controls (Table 2). Pairwise comparisons of the mean scores between the 2 groups for each trial showed significant group differences for all trials, all P < 0.001. Moreover, there was a main effect of trial (F(2,104) = 80, P < 0.001, and η2 = 1) indicating differential changes in RT across trials. Post hoc analysis showed that RT in unilateral-left trials were significantly faster than both unilateral-right and bilateral trials, in both groups. The interaction between RT and group was not significant, (F(2,104) = 0.39, P = 0.6, and η2 = 0.1) showing similar pattern of changes in RT across the 3 trials for patients and controls.
4.2. Star and Triangle Task
Finding on error rate during this task, similar to the previous section, showed significant group effect (F(1,52) = 11.9 , P = 0.001, and η2 = 0.9) with higher patient error rate compared to controls, all P < 0.005 (Table 3). Also, there was a significant effect of trial (F(2,104) = 6.2, P = 0.003, and η2 = 0.8) and error rates significantly differed among various trials. Finally, the interaction effect between error rate and group was significant, (F(2,104) = 3.6, P = 0.05, and η2 = 0.6) showing that the performances of the 2 groups differed across the 3 trials. For controls, post-hoc comparisons showed that error rate did not differ between the different trials, all P>0.05. However, patients had significantly lower error rates on bilateral trials compared with both unilateral-right, P = 0.001, and unilateral-left trials, P = 0.04. They also had greater error rates on unilateral-right relative to unilateral-left trials, P < 0.001 (Figure 1B). Finally, in bilateral trials, the effect of similarity was not significant (F(1,52) = 2.3, P = 0.1, and η2 = 0.3) and error rate was similar in identical and non-identical bilateral trials in both groups.
Results of response time showed the significant main effect of group (F(1,52) = 12.2, P = 0.001, and η2 = 0.9) and patients responded more slowly than controls on all trials (Table 3). There was a significant effect of trial (F(2,104) = 47.1, P < 0.001, and η2 = 1) yet its interaction effect with group was not significant (F(2,104) = 2.2, P = 0.1, and η2 = 0.4). Follow-up comparisons showed that both groups had longer response times on bilateral compared to unilateral trials.
4.3. Correlation Analyses
In the group of patients, bivariate correlation analyses were performed between the error rates with the CPZ equivalent dose and clinical characteristics of the patients including PANS, duration of disease, and age of onset. No significant correlation was found (all P > 0.2).
5. Discussion
This study aimed at evaluating visual processing during unilateral displays compared to bilateral in patients with schizophrenia and healthy controls. To reach this goal, the study compared accuracies in unilateral-left, unilateral-right, and bilateral displays on 2 different tasks, namely the circle task, and the star and triangle task.
The results showed that in both tasks, patients with schizophrenia: i, were significantly less accurate at detecting unilateral-right compared with unilateral-left stimulus; and ii, were significantly better at detecting 2 simultaneously presented visual stimuli versus a stimulus presented in isolation (anti-extinction effect). Altogether, these findings indicate an attentional deficit towards the right hemifield in patients, and some putative compensatory mechanisms, such as ‘priming effect’, ‘attentional waiting’, and ‘transient binding’, that helped them show a better performance in bilateral displays (12-16). In fact, possible explanations may be extrapolated from different theories that researchers have suggested to explain observed anti-extinction effect in patients with unilateral parietal damage. For example, Goodrich and Ward suggested that the competition for visual attention could be modulated by a priming effect, which might strengthen response mechanisms in bilateral trials (15). According to this theory, the researchers suggest that the presence of a right stimulus was not sufficient to activate response mechanisms in patients, yet the left visual stimulus activated the response. Indeed, the primed response mechanisms pulled out a stimulus that could be ignored, and the severity of attention deficits was reduced in bilateral trials (15). According to “attentional waiting” theory, it could be suggested that unilateral targets in the right visual field were missed because attention was captured on the left side, yet right side targets were detected under bilateral presentation because patients detected the targets in the left visual field, then shifted attention to the right side (12). The third explanation is the design of the study tasks. Based on “transient binding” theory, as the left and right stimuli in the current study had common onset and offset, common color, and belonged to the same semantic category, they could compensate left attentional deficit by linking them to each other and enhancing performances in bilateral presentations (14, 28). Moreover, in the task design, the distance between the 2 targets was small (less than 15°), thus grouping effect in this distance might have better helped patients detect bilateral stimuli (29).
It should be mentioned that both findings of this study, right hemi-neglect and anti-extinction effect, support other studies that have suggested parietal lobe dysfunction in patients with schizophrenia (21, 30, 31). Indeed, right hemi-spatial neglect might indicate either impairment of the left parietal lobe in allocation of attention to the right visual space, or hyperactivity of right parietal lobe leading to more saliency of the left visual field (21). In support of this, Posner et al. (1988) found that patients demonstrated attentional deficits to the targets in the right visual field similar to patients, who had a left hemisphere lesion (32). Moreover, in target cancellation tasks, patients with schizophrenia showed greater omissions in the right side compared to the left side (33, 34), and in line bisection tasks they demonstrated a significant leftward bias in their estimation of the center of a line, suggesting right hemispatial neglect (23, 35). Finally, all previous studies showed anti-extinction effect in patients with parietal lobe damage (12-16). Therefore, the current findings of anti-extinction effect in patients with schizophrenia could also suggest dysfunction of parietal lobe in the patients.
Interestingly, the current study demonstrated evidence of hemineglect, yet not extinction effect in patients with schizophrenia. A point of debate in the experimental literature is the extent to which the visual extinction and neglect are related and share a common underlying mechanism. Although some studies have reported both neglect and extinction in the same patients (36), others have shown that neglect and extinction can occur independently. The present results showed at least dissociation between neglect and extinction in patients with schizophrenia.
The 2 groups showed no differences in bilaterally distinguishing between identical and non-identical stimuli presented in the star and triangle task. The role of stimulus similarity in extinction has been examined (17, 28, 37) with inconsistent results. For example, Baylis et al. (1993) found that extinction was more pronounced for identical stimuli in color or form (37), in contrast, Ptak et al. (2002) showed that extinction was reduced for identical stimuli (17). The current findings extend previous studies in showing that similarity has no effect on anti-extinction phenomenon in schizophrenia.
Regarding limitations of the study, it could be mentioned that all patients were taking their medication, which might have affected the results. However, chlorpromazine equivalent doses were not significantly correlated with task measures, making any potential effect of medication on the finding unlikely. While there were greater percentages of male participants in the 2 groups, it could not affect the finding of anti-extinction in patients, since several studies have found that gender is not a determining factor in impaired visuospatial attention in patients with schizophrenia (38, 39). Also, the patient group was recruited mainly from an outpatient clinic and showed mild symptoms, thus they may not exactly represent individuals typically encountered in clinical practice or examined in previous schizophrenia studies, which may limit the ability to generalize the findings.
5.1. Conclusion
The results of this study show evidence of bilateral advantages in visual detection processing in patients with schizophrenia.