1. Background
Pistachio has a high nutritional value and popularity among consumers. It is produced approximately 500 to 700 thousand tons per year in different world regions with warm weather, mainly in Eastern Mediterranean countries such as Iran, Syria, and Turkey. The largest global producers/exporters of pistachio are Iran and the United States (1). Kerman province in Iran, with more than 200,000 hectares of pistachio cultivated land, approximately half of which are located in Rafsanjan city, is one of the largest geographical regions of pistachio production. Most of Iran's pistachios have been exported to other countries, around 20 to 30 metric tons to European Union annually during 2012 - 2018 (2). The contamination of pistachio nuts with fungal toxins, especially aflatoxin, is a significant health challenge that consumers face, creating devastating economic consequences to producers (3). All varieties of this agro product are seriously contaminated by aflatoxin. Therefore, the prediction and control of aflatoxin contamination in pistachios are challenging issues hard to be managed. It should be mentioned that the mechanism of aflatoxin contamination is unknown since it is complex and depends on multifactors (4).
Furthermore, the aflatoxin contamination processes are considerably varied over some consecutive years. It can occur during the process of agro-products from storage, production, processing to trade, which seriously threatens consumption safety. The risk of aflatoxin contamination in agro-products needs to be assessed to control aflatoxin contamination, ensure consumer safety, develop prediction, prevent, and identify critical control points (CCPs) for precise control in practice (4, 5).
AFs are secondary metabolites and polyketides produced by some Aspergillus species, especially Aspergillus flavus and Parasitocus. AFs contaminate most foodstuffs, such as corn, rice, peas, wheat, and tree nuts (6-8). AFs are carcinogenic, teratogenic, and mutagenic to humans and animals (9). Human exposure to AFs contaminants has been a matter of increased concern over the last decades. It has been estimated that half of the people worldwide are exposed to dietary aflatoxins (10). There are four major naturally occurring aflatoxins (B1, B2, G1, and G2) in foodstuff, among which Aflatoxin B1 is considered the most toxic (11). The International Agency for Research on Cancer (IARC) classified AFB1 as a Group 1 human carcinogen (12). The highest rate of aflatoxin contamination in pistachio nuts is associated with aflatoxin B1 (80 - 100% contamination) (13). Epidemiological studies on AFs effects reveal some evidence that links human diseases to AFs contamination. In this respect, AFs are considered to be correlated with liver diseases. The possible role of AFs in human liver cancer has already been discussed. AFs contamination has been known to induce liver carcinogen [hepatocellular carcinoma (HCC)] in humans and animals. Previous studies suggest that HCC risk is multiplicatively (0.3 per 105 population) higher for individuals exposed to aflatoxin and chronic infection with hepatitis B virusthan individuals who are only exposed to aflatoxin (0.01 per 105 population). Exposure to AFs via dietary intake could be regarded as a potentially severe risk (14). Risk assessment is used to determine the health effects of mycotoxin exposure and guide food regulators to set thresholds for these chemicals in foodstuffs (15).
There is a method called quantitative exposure assessment by which scientific data are analyzed so that undesirable events can be assessed in terms of their severity and probability (16). This method is the case to measure the risks caused by consuming food containing xenobiotics. Risk assessments are advantageous, providing more effective risk management strategies. They are mainly associated with how much food an individual receives in a certain period and the number of xenobiotics absorbed into the body. The estimated human exposure to genotoxic carcinogens, including AFB1, can be evaluated by different criteria such as tolerated daily intake (TDI), dietary exposure, and the margin of exposure (MOE), which is regarded as valid references have been commonly used in this respect (17).
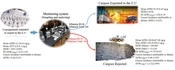
To control the aflatoxins level, monitoring programs for AFs in various commodities are strictly scrutinized by the authorities in many countries and international organizations. The monitoring system for sampling, analyzing, and issuing pistachio consignments exported to the E.U. has been established in Iran by the Ministry of Health to control hygienic conditions of pistachio cultivation, harvesting, and post-harvesting practices. All cultivated and processed pistachios are evaluated in food control laboratories. The exported shipments to the E.U. are rejected if their aflatoxin levels exceed the E.U. commission levels (AFB1 = 8 ppb, AFT = 10 ppb). Iran is the leading exporter of pistachios to Europe; therefore, regular monitoring of pistachio nuts is critical to control their safety and minimize mycotoxins contamination. This paper reports the results of an extensive survey on aflatoxins occurrence done by food control labs to monitor the contamination of raw pistachio intended to export to the E.U. from November 2012 to October 2018. This study aimed to investigate the risk assessment of AFB1 based on dietary exposure, the margin of exposure (MOE), Hazard index, the overall estimation of liver cancer risk (cases/100,000 population/year), and the percent of cancer incidence attributed to dietary AFB1 from consuming pistachio nuts for European people and investigate the impact of the monitoring system on these factors.
2. Methods
2.1. Sample Collection
Sampling was performed from all consignments (7298 pistachio nut samples) by food control inspectors in Kerman and Rafsanjan from Nov. 2012 to Oct 2018. A total of 928, 1016, 1343, 1444, 1227, and 1340 samples were analyzed in 2012-3 (Nov. 2012 - Oct. 2013), 2013 - 4, 2014 - 5, 2015 - 6, 2016 - 7, and 2017 - 8, respectively. The sampling was based on the E.U. sampling procedures described in Commission Regulation (E.U.) NO 178/2010 (18). According to the process, 100 incremental samples weighing 200 g were randomly taken from each consignment. They were mixed and then divided into two 10 kg sub-samples. Preparation of sub-samples for analysis and further analytical experiments were then carried out in Toxicology Labs of Food Control Laboratories located in Rafsanjan and Kerman.
2.2. Sample Preparation
To minimize the sub-sampling errors in aflatoxin analysis, a water slurry of pistachio nut samples was prepared. Thus, 10 L water was added to each 10 kg sub-sample of pistachio nut; then, the mixture was mixed and ground using a slurry machine for 15 min. once done, 125 g of the slurry was used as the test portion for analysis (7).
2.3. Chemicals and Reagents
All aflatoxin standards were prepared from Sigma (MO, USA). High-performance liquid chromatography (HPLC) grade of methanol and acetonitrile supplied from Caledon Co. (Caledon laboratories Ltd., Canada). All materials used in the preparation of phosphate-buffered saline [pH 7.4; 0.20 g KCl, 0.20 g KH2PO4, 1.16 g anhydrous Na2HPO4 (or 2.92 g Na2HPO4,12H2O) and 8.0 g NaCl dissolved in 900 ml water and pH adjusted to 7.4 with 0.1 M HCl or 0.1 M NaOH and diluted to 1 L with water] were procured from Merck Co. (Merck, Darmstadt, Germany).
2.4. Apparatus
Liquid chromatography (LC) analysis was performed using a reverse-phase HPLC system (Dionex, Sunnyvale, California L.P., USA) equipped with a Gilson-Workstation (GX-271 Aspec Gilson, USA), vacuum degasser (Ultimate-3000, Dionex, Sunnyvale, California L.P., USA), temperature-controlled oven (Ultimate-3000, Dionex, Sunnyvale, California L.P., USA), and fluorescence detector (R.F. 2000, Dionex, Sunnyvale, California L.P., USA). The Dionex LC column was C18, 250 × 4.6 mm, 5 µm. The Aflatest immunoaffinity columns were purchased from Vicam (M.A., USA).
2.5. Standard Solutions
When standard solutions of each aflatoxin were prepared, their concentration was determined using a UV-Visible Spectrophotometer (Varian, CARY 100, USA) through AOAC method No. 971.22 (AOAC 2000) applying the following formula:
Where A is the absorbance measured at 365 nm, MW is the molecular weight of aflatoxin; CF is the correction factor for spectrophotometer and C molar absorptivity of aflatoxin.
These standards were used to prepare mixed standards containing 40 ng/mL AFs (B1, G1 = 16 ng/g; B2, G2 = 4 ng/g). The working standard solution was prepared by diluting mixed standards with methanol and water by pipetting appropriate volumes into a set of 20 mL calibrated volumetric flasks and diluting to volume. Concentrations of aflatoxins B1 and G1 were 0.025, 0.05, 0.1, 0.2, 0.4, 0.8, 1.6, 2.4 ng/mL and B2 and G2 were 0.00625, 0.0125, 0.025, 0.05, 0.1, 0.2, 0.4, 0.6 ng/mL, respectively. An eight-point calibration curve was built for each type of aflatoxin.
2.6. Extraction and Clean Up
Aflatoxin content of samples was analyzed using the official method AOAC No. 999.07 with minor modifications (19). Pistachio nut slurries were extracted with methanol/water/hexane (300 mL/75 mL/100 mL), after which the extract was filtered through MN 619 de filter paper. After filtration, 3.1 mL of extract was diluted with 9.9 mL deionized water then mixed with 8 mL air in the workstation. To clean up the diluted extract, aflatest immunoaffinity columns were used. First, 5 mL phosphate buffer saline was passed through the column by a workstation-cleaned needle. Then, 12.6 mL of the diluted filtrate was passed through the aflatest column at a flow rate of ca. one drop/s. The column was washed with 15 ml water and dried, applying a slight vacuum. Finally, aflatoxin was eluted with methanol through the following procedure. At first, 0.5 mL methanol was added to the column, which passed by gravity. After 1 min, the second portion of 0.75 mL methanol was applied and collected. The elute was diluted with 1.75 mL of deionized water, after which 100µL was injected into HPLC (13).
2.7. Chromatographic Conditions
The post-column bromination with KobraCell (Coring System, Germany) is used to perform Reversed-phase LC determination of aflatoxins. A flow rate, fluorescence detection at wavelength, and emission wavelength were 0.8 mL/min, 365 nm, and 435 nm, respectively. The column temperature was 36°C. Retention times for aflatoxins G2, G1, B2, and B1 were 10, 11.63, 13.6, and 15.06 min, respectively. The isocratic mobile phase was the water-acetonitrile-methanol solution and a ratio of 60: 20: 30 (v/v/v), containing 120 mg/L KBr and 350 μL HNO3 4M.
2.8. Calibration Graph
The calibration graph, prepared using the working solutions, covered the ranges stated in section 2.5. It was constructed before the analysis to check the pilot for linearity (r2 = 0.999), and it later was used to quantify aflatoxin. If the sample contained no toxin within the calibration range, a more appropriate calibration graph was prepared or diluted so that an aflatoxin concentration could be suitable for the calibration graph. Limit of detection (LOD) and quantification (LOQ) of the HPLC were estimated by making a calibration curve according to the following equations:
Where σ is the standard deviation of the response or standard deviation of y-intercepts, s is the slope of the calibration curve.
2.9. Quality Assurance
Validated methods were used to evaluate the reliability of results, and internal and external quality control experiments were performed. Regarding internal quality control, it was verified that the methods were accurate and precise. Therefore, recoveries of aflatoxins B1, B2, G1, and G2 were recorded by analyzing a blank pistachio nut sample spiked at four ng/g for aflatoxins B1 and G1 and one ng/g for aflatoxins B2 and G2. Based on the recovery values, aflatoxin levels were corrected. Expanded uncertainties (k = 2, 95% confidence) were evaluated to indicate measurement uncertainties. Main sources of uncertainty were considered for this study: volumetric operations, weighing, purity of standards and reference materials, multi-point calibration (linear regression), and recovery. Samples were analyzed at concentrations of 1, 4, and 8 ng/g to evaluate the uncertainty in high-performance liquid chromatography. Uncertainty values are given in Table 1. Concerning external quality control, Food Control Labs regularly participated in proficiency testing of the Food Analysis Performance Assessment Scheme (FAPAS) in the U.K., obtaining a consistently satisfactory Z-score (-2 ≤ Z score ≤ +2).
| Year | No. | AF Type | LOD (ng/g) | LOQ (ng/g) | Spike Level (ng/g) | Average Recovery (%) | RSDR (%) | Expanded Uncertainty (ng/g) | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Spiked Level 1 (ng/g) | Spiked Level 4 (ng/g) | Spiked Level 8 (ng/g) | ||||||||
| Nov. 2012 - Oct. 2013 | 110 | B1 | 0.078 | 0.26 | 4.0 | 79.50 | 4.10 | 0.083 | 0.273 | 0.430 |
| B2 | 0.032 | 0.11 | 1.0 | 85.60 | 2.40 | 0.072 | 0.090 | 0.120 | ||
| G1 | 0.078 | 0.26 | 4.0 | 92.54 | 2.75 | 0.125 | 0.302 | 0.580 | ||
| G2 | 0.032 | 0.11 | 1.0 | 85.22 | 3.50 | 0.068 | 0.121 | 0.137 | ||
| Nov. 2013 - Oct. 2014 | 145 | B1 | 0.078 | 0.26 | 4.0 | 78.60 | 3.30 | 0.067 | 0.220 | 0.346 |
| B2 | 0.032 | 0.11 | 1.0 | 85.20 | 3.30 | 0.098 | 0.123 | 0.166 | ||
| G1 | 0.078 | 0.26 | 4.0 | 91.30 | 2.20 | 0.100 | 0.242 | 0.464 | ||
| G2 | 0.032 | 0.11 | 1.0 | 87.40 | 1.90 | 0.037 | 0.065 | 0.074 | ||
| Nov. 2014 - Oct. 2015 | 120 | B1 | 0.070 | 0.23 | 4.0 | 91.03 | 6.20 | 0.126 | 0.413 | 0.651 |
| B2 | 0.020 | 0.07 | 1.0 | 86.80 | 3.10 | 0.092 | 0.116 | 0.156 | ||
| G1 | 0.070 | 0.23 | 4.0 | 97.60 | 2.40 | 0.109 | 0.264 | 0.507 | ||
| G2 | 0.020 | 0.07 | 1.0 | 80.76 | 4.90 | 0.095 | 0.169 | 0.192 | ||
| Nov. 2015 - Oct. 2016 | 132 | B1 | 0.066 | 0.22 | 4.0 | 86.76 | 7.80 | 0.159 | 0.519 | 0.819 |
| B2 | 0.033 | 0.11 | 1.0 | 82.69 | 6.50 | 0.194 | 0.243 | 0.326 | ||
| G1 | 0.066 | 0.22 | 4.0 | 98.74 | 5.60 | 0.255 | 0.615 | 1.182 | ||
| G2 | 0.033 | 0.11 | 1.0 | 80.45 | 6.90 | 0.133 | 0.238 | 0.270 | ||
| Nov. 2016 - Oct. 2017 | 146 | B1 | 0.066 | 0.22 | 4.0 | 86.91 | 6.20 | 0.126 | 0.413 | 0.651 |
| B2 | 0.033 | 0.11 | 1.0 | 89.11 | 6.50 | 0.194 | 0.243 | 0.326 | ||
| G1 | 0.066 | 0.22 | 4.0 | 96.07 | 6.40 | 0.292 | 0.703 | 1.351 | ||
| G2 | 0.033 | 0.11 | 1.0 | 92.09 | 2.10 | 0.041 | 0.072 | 0.082 | ||
| Nov. 2017 - Oct. 2018 | 165 | B1 | 0.066 | 0.22 | 4.0 | 87.06 | 8.50 | 0.173 | 0.566 | 0.892 |
| B2 | 0.033 | 0.11 | 1.0 | 93.57 | 4.90 | 0.146 | 0.183 | 0.246 | ||
| G1 | 0.066 | 0.22 | 4.0 | 92.92 | 4.96 | 0.226 | 0.545 | 1.047 | ||
| G2 | 0.033 | 0.11 | 1.0 | 86.75 | 7.20 | 0.139 | 0.248 | 0.282 | ||
Abbreviations: LOD, limit of detection; LOQ, limit of quantification; RSDR, relative standard deviation for reproducibility.
2.10. Risk Assessment of Exposure to Aflatoxins B1 via Consumption of Pistachio Nuts
2.10.1. Exposure Estimation
The dietary exposure or estimated dietary intake (EDI) was calculated for pistachio nuts consumers according to following equation: Dietary exposure = [Contamination level AFB1mean or max (ng/kg) × daily pistachio intake (kg/day)]/Average body weight (kg).
The EDI was calculated using the mean or maximum contamination level of AFB1 in pistachio nuts for each year are shown in Table 2, daily pistachio intake, and the average body weight. Italy is considered a country with the highest daily consumption in the E.U., estimated at 0.52 and 2.05 g/person/day in all populations and those who consumed the specific pistachio product in 2016, respectively (20). For Iranians, this figure is 0.82 g/person/day (20, 21). The average body weight of 70 kg for adults was applied (22, 23).
| Year | Consignment No. | AF Type | All consignment ng/g (ppb) | AF Contamination (%) | Consignment Rejection b (%) | AF Contamination of Authorized Consignment | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Median | Max | AFs (ng/g) | Mean | Max | ||||||||
| < LOD | LOD - 5 | 5 - 8 | 8 - 20 | ≥ 20 | |||||||||
| AFT (ng/g) | |||||||||||||
| < LOD | LOD - 10 | 10 - 15 | 15 - 20 | ≥ 20 | |||||||||
| Nov. 2012 - Oct. 2013 | 928 | B1 | 1.78 | < LODa | 181.9 | 68.91 | 25.81 | 0.92 | 2.16 | 2.21 | 6.14 | 0.23 | 7.4 |
| B2 | 0.15 | < LOD | 17.1 | 86.31 | 13.15 | 0.16 | 0.38 | 0.00 | 0.02 | 2.2 | |||
| G1 | 0.06 | < LOD | 37.5 | 98.22 | 1.51 | 0.00 | 0.16 | 0.11 | 0.00 | 1.8 | |||
| G2 | 0.00 | < LOD | 4.2 | 99.73 | 0.27 | 0.00 | 0.00 | 0.00 | 0.00 | 0.0 | |||
| Total | 1.98 | < LOD A | 197.6 | 68.86 | 27.37 | 0.54 | 0.65 | 2.59 | 0.26 | 9.4 | |||
| Nov. 2013 - Oct. 2014 | 992 | B1 | 2.38 | < LOD ab | 345.7 | 66.28 | 25.35 | 2.07 | 2.72 | 3.58 | 9.27 | 0.38 | 7.93 |
| B2 | 0.26 | < LOD | 102.5 | 82.11 | 16.23 | 0.60 | 0.15 | 0.91 | 0.03 | 2.86 | |||
| G1 | 0.05 | < LOD | 25.3 | 97.68 | 2.07 | 0.00 | 0.15 | 0.10 | 0.01 | 3.07 | |||
| G2 | 0.00 | < LOD | 2.8 | 99.80 | 0.20 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | |||
| Total | 2.67 | < LOD A | 384.2 | 66.99 | 27.92 | 1.26 | 0.50 | 3.33 | 0.42 | 8.61 | |||
| Nov. 2014 - Oct. 2015 | 1321 | B1 | 3.76 | < LOD bcd | 340.3 | 64.23 | 27.55 | 1.63 | 2.42 | 4.16 | 10.67 | 0.43 | 7.76 |
| B2 | 0.37 | < LOD | 146.1 | 78.50 | 19.76 | 0.87 | 0.64 | 0.23 | 0.03 | 0.77 | |||
| G1 | 0.23 | < LOD | 232.1 | 96.71 | 2.80 | 0.08 | 0.15 | 0.26 | 0.01 | 1.29 | |||
| G2 | 0.02 | < LOD | 26.3 | 99.32 | 0.61 | 0.00 | 0.04 | 0.04 | 0.00 | 0.20 | |||
| Total | 4.38 | < LOD B | 367.7 | 64.08 | 29.45 | 1.02 | 0.83 | 4.62 | 0.47 | 8.69 | |||
| Nov. 2015 - Oct. 2016 | 1457 | B1 | 3.94 | 0.1cd | 450.3 | 48.83 | 43.51 | 1.51 | 2.47 | 3.67 | 10.57 | 0.41 | 7.81 |
| B2 | 0.37 | < LOD | 52.6 | 77.45 | 20.69 | 0.69 | 0.82 | 0.34 | 0.02 | 1.45 | |||
| G1 | 0.03 | < LOD | 25.0 | 95.88 | 4.02 | 0.07 | 0.00 | 0.03 | 0.01 | 3.21 | |||
| G2 | 0.00 | < LOD | 3.0 | 99.62 | 0.38 | 0.00 | 0.00 | 0.00 | 0.00 | 0.04 | |||
| Total | 4.34 | 0.1B | 478.9 | 48.83 | 45.30 | 1.10 | 0.86 | 3.91 | 0.44 | 8.32 | |||
| Nov. 2016 - Oct. 2017 | 1240 | B1 | 4.02 | < LOD d | 413.2 | 50.93 | 40.00 | 1.65 | 3.15 | 4.27 | 9.76 | 0.35 | 7.98 |
| B2 | 0.41 | < LOD | 52.5 | 76.29 | 21.57 | 0.97 | 0.85 | 0.32 | 0.02 | 0.71 | |||
| G1 | 0.02 | < LOD | 7.65 | 95.73 | 4.15 | 0.12 | 0.00 | 0.00 | 0.00 | 0.65 | |||
| G2 | 0.00 | < LOD | 0.71 | 99.64 | 0.36 | 0.00 | 0.00 | 0.00 | 0.00 | 0.08 | |||
| Total | 4.45 | < LOD B | 479.8 | 50.93 | 42.22 | 1.37 | 0.93 | 4.56 | 0.38 | 8.46 | |||
| Nov. 2017 - Oct. 2018 | 1357 | B1 | 2.57 | < LOD abc | 422.6 | 67.58 | 27.75 | 0.63 | 1.44 | 2.62 | 5.08 | 0.15 | 6.64 |
| B2 | 0.26 | < LOD | 36.2 | 84.34 | 13.96 | 0.63 | 0.92 | 0.15 | 0.01 | 0.86 | |||
| G1 | 0.03 | < LOD | 50.6 | 97.61 | 2.25 | 0.11 | 0.00 | 0.04 | 0.01 | 5.34 | |||
| G2 | 0.00 | < LOD | 1.7 | 99.56 | 0.44 | 0.00 | 0.00 | 0.00 | 0.00 | 0.11 | |||
| Total | 2.87 | < LOD AB | 458.8 | 67.21 | 28.89 | 0.44 | 0.59 | 2.87 | 0.17 | 7.14 | |||
Abbreviation: LOD, limit of detection.
a Different lowercase and uppercase letter in the column indicate a significant difference (P < 0.05) between the average contamination of AFB1 and AFT, respectively.
b Removal percentage of consignment with AFs contamination exceeding the E.U. (AFB1 < 8, AFT < 10) using the monitoring system.
2.10.2. Risk Characterization
The MOE proposed by the European Food Safety Authority (EFSA) and Population risk for primary liver cancer proposed by the Joint Expert Committee (WHO/FAO) on Food Additives (JECFA) were used to estimate risk assessment.
EFSA has proposed the MOE approach for the comparison of estimated risk between compounds and their magnitude. The MOEis the ratio of benchmark dose lower confidence limit 10% (BMDL10), obtained from animal toxicology studies to the estimated human exposure level. Its values were calculated as a ratio between the toxicological reference (BMDL) and the exposure where BMDL10 (rat) = 170 ng/kg.bw/day (17). It is commonly used in the human health risk assessment, which only indicates a level of concern, and its amount less than 10000 would be considered of concern from the public health point of view. MOE was calculated according to the following equation (24):
Population risk for primary liver cancer attributable to aflatoxin consumption through pistachio nuts was calculated according to the following equation (24): Population risk [[(cancers/year)/105] population] = Dietary exposure × Carcinogenic potency
Carcinogenic potency is estimated based on the synergistic HCC effects of AFB1 and hepatitis B virus (HBV) infection. HCC risk for individuals exposed to both aflatoxin and chronic infection with HBV is 0.562 cancer/year/105 population per 1 ng/kg.bw/day which according to the Centers for Disease Control and Prevention (CDC) of Iran and the E.U., the overall prevalence rate of HBV was 1.5% and 0.9%, respectively (25, 26). It was estimated at 0.049 cancers cases/year/105 population per 1 ng/kg.bw/day in individuals who are only exposed to aflatoxin (27). The carcinogenic potency was calculated according to the following equations (24):
2.11. Hazard Index (HI)
According to the below-mentioned formula, the Hazard Index was obtained by dividing the EDI by TD50 of AFB1 (3.2µg/kg bw/day), divided by an uncertainty factor of 50,000. TD50 is a dose that induces tumors in half of the tested animals (28).
2.12. Statistical Analysis
The normality of data was checked by using the Kolmogorov-Smirnov test. According to the normality test, data distribution was not normal; therefore, the difference between AFB1 and AFT contamination levels in different years was compared using Kruskal-Wallis H non-parametric tests. Statistical analysis was performed using IBM-SPSS (version 25), and values of P < 0.05 were considered statistically significant. The graph is drawn in Excel software (version 2013).
3. Results
Internal quality validation parameters such as LOD, LOQ, and recovery percentage are shown in Table 1. The Limitation range was 0.066 - 0.078 ng/g and 0.02 - 0.033 ng/g for aflatoxins B1 & G1 and B2 & G2, respectively. These parameters were acceptable, implying that the generated data and good performance at low statutory limits are reliable. The accuracy and precision of the used analytical method were investigated by calculating the average recovery and relative standard deviation for reproducibility (RSDR). The average recoveries of the spiked blank samples with four ng/g for aflatoxin B1 and G1and one ng/g for aflatoxin B2 and G2 were 78.6% - 97.6% with RSDR below 8.5%, which were within the acceptable range set by the European commission No. 401/2006.
The incidence and distribution of AFs in pistachio for export to the E.U. 2012 - 2018 are detailed in Table 2. According to previous studies, the most prevalent aflatoxin in analyzed pistachio nuts was AFB1, accounting for more than 86% of the total, followed by B2, G1, and G2. The latter was detected only in seven samples (7, 13). The mean contamination level of AFT and AFB1 for all samples was 4.07 ± 20.98 ng/g and 3.64 ± 18.92 ng/g in 14590 sub-samples, analyzed during the years 2012 - 2018, respectively. No aflatoxin contamination (AFT < LOD) was detected in 60.4% of all analyzed samples (8810); however, AFB1 and AFT exceeded the acceptable level set by the E.U. in 789 (5.47%) and 853 (5.84%) sub-samples, respectively. The median contamination level of samples was < LOD, and the mean occurrence level for AFB1 and AFT was lower than the maximum tolerable limits according to the E.U. and Iran standard regulations (AFB1 = 8, AFT = 10 ng/g). There was a significant difference in the average concentration of AFT at years 2012 - 3, 2013 - 4, and 2017 - 8 with the years 2014 - 5, 2015 - 6, and 2016 - 7 (Table 2). The highest and lowest mean and maximum contamination level was observed in 2016 - 7 and 2012 - 3, respectively. With the increase in the average level of contamination, the percentage of rejected cargoes rose in the range of 5.08 - 10.67%, with the highest and lowest rejections in 2014 - 5 and 2017 - 8, respectively. Regarding authorized consignment, the mean aflatoxin contamination level was drastically reduced after removing cargoes with AFs content exceeding the E.U. and Iran standard levels.
The calculated dietary exposure to AFB1, based on the mean and maximum contamination levels, is shown in Table 3. The dietary exposure for all European populations and those who consumed the specific pistachio product was in the range of 0.013 - 0.03 (ng/kg.bw/day) and 0.052 - 0.118 (ng/kg.bw/day), respectively. Iranians dietary exposure was 0.021 - 0.049 (ng/kg - bw/day). This value is reduced from 6.1 to 14.8 times after removing cargoes with the AFs contamination exceeding the European limit. This amount increased in the range of 0.3 - 1.98 (ng/kg - bw/day) in consignment rejected using a monitoring system.
| Year | Mean AFB1 µg.kg (ppb) | Dietary Exposure to AFB1 in EU (ng.kg - bw.day) | Dietary AFB1 Exposure (ng.kg - bw.day), MOE BMDL10 for Rat a | Hazard Index | Estimated Liver Cancer Risk (Cases.100,000 Population. Year) | Cancer Incidence Attributable to Dietary AFB1 (%) b | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EU c | EU d | IR e | EU c | EU d | IR e | EU c | EU d | IR e | EU c | EU d | IR e | EU c | EU d | IR e | ||
| All Consignment | ||||||||||||||||
| 2012 - 3 | 1.78 | 0.0132 | 0.0523 | 0.0216 | 12843 | 3254 | 7865 | 0.21 | 0.82 | 0.34 | 0.00071 | 0.00280 | 0.00123 | 0.02 | 0.09 | 0.02 |
| 2013 - 4 | 2.38 | 0.0177 | 0.0699 | 0.0289 | 9605 | 2433 | 5882 | 0.28 | 1.09 | 0.45 | 0.00095 | 0.00375 | 0.00164 | 0.03 | 0.12 | 0.03 |
| 2014 - 5 | 3.76 | 0.0280 | 0.1104 | 0.0457 | 6080 | 1540 | 3723 | 0.44 | 1.72 | 0.71 | 0.00150 | 0.00592 | 0.00259 | 0.05 | 0.19 | 0.04 |
| 2015 - 6 | 3.94 | 0.0293 | 0.1157 | 0.0478 | 5802 | 1470 | 3553 | 0.46 | 1.81 | 0.75 | 0.00157 | 0.00620 | 0.00271 | 0.05 | 0.20 | 0.05 |
| 2016 - 7 | 4.02 | 0.0299 | 0.1180 | 0.0488 | 5687 | 1441 | 3483 | 0.47 | 1.84 | 0.76 | 0.00160 | 0.00633 | 0.00277 | 0.05 | 0.20 | 0.05 |
| 2017 - 8 | 2.57 | 0.0191 | 0754.0 | 0.0312 | 8895 | 2253 | 5447 | 0.30 | 1.18 | 0.49 | 0.00102 | 0.00404 | 0.00177 | 0.03 | 0.13 | 0.03 |
| Authorized Consignment | ||||||||||||||||
| 2012 - 3 | 0.23 | 0.0017 | 0.0068 | 0.0028 | 99394 | 25180 | 60870 | 0.03 | 0.11 | 0.04 | 0.00009 | 0.00036 | 0.00016 | 0.00 | 0.01 | 0.00 |
| 2013 - 4 | 0.38 | 0.0029 | 0.0113 | 0.0047 | 60159 | 15240 | 36842 | 0.04 | 0.17 | 0.07 | 0.00015 | 0.00060 | 0.00026 | 0.00 | 0.02 | 0.00 |
| 2014 - 5 | 0.43 | 0.0030 | 0.0117 | 0.0048 | 57151 | 14478 | 35000 | 0.05 | 0.18 | 0.08 | 0.00016 | 0.00063 | 0.00028 | 0.01 | 0.02 | 0.00 |
| 2015 - 6 | 0.41 | 0.0030 | 0.0117 | 0.0048 | 57151 | 14478 | 35000 | 0.05 | 0.18 | 0.08 | 0.00016 | 0.00063 | 0.00028 | 0.01 | 0.02 | 0.00 |
| 2016 - 7 | 0.35 | 0.0029 | 0.0115 | 0.0048 | 58617 | 14850 | 35897 | 0.05 | 0.18 | 0.07 | 0.00016 | 0.00061 | 0.00027 | 0.01 | 0.02 | 0.00 |
| 2017 - 8 | 0.15 | 0.0013 | 0.0051 | 0.0021 | 127003 | 32174 | 77778 | 0.02 | 0.08 | 0.03 | 0.00007 | 0.00028 | 0.00012 | 0.00 | 0.01 | 0.00 |
| Rejected Consignment | ||||||||||||||||
| 2012 - 3 | 42.3 | 0.3146 | 1.2417 | 0.5136 | 540 | 137 | 331 | 4.91 | 19.40 | 8.03 | 0.01687 | 0.06658 | 0.02912 | 0.54 | 2.15 | 0.49 |
| 2013 - 4 | 40.84 | 0.3037 | 1.1988 | 0.4959 | 560 | 142 | 343 | 4.75 | 18.73 | 7.75 | 0.01628 | 0.06428 | 0.02812 | 0.53 | 2.07 | 0.48 |
| 2014 - 5 | 56.2 | 0.4179 | 1.6497 | 0.6824 | 407 | 103 | 249 | 6.53 | 25.78 | 10.66 | 0.02241 | 0.08845 | 0.03869 | 0.72 | 2.85 | 0.66 |
| 2015 - 6 | 67.48 | 0.5018 | 1.9808 | 0.8194 | 339 | 86 | 207 | 7.84 | 30.95 | 12.80 | 0.02691 | 0.10621 | 0.04646 | 0.87 | 3.43 | 0.79 |
| 2016 - 7 | 56.22 | 0.4181 | 1.6503 | 0.6827 | 407 | 103 | 249 | 6.53 | 25.79 | 10.67 | 0.02242 | 0.08848 | 0.03870 | 0.72 | 2.85 | 0.66 |
| 2017 - 8 | 64.95 | 0.4830 | 1.9066 | 0.7887 | 352 | 89 | 216 | 7.55 | 29.79 | 12.32 | 0.02590 | 0.10222 | 0.04471 | 0.84 | 3.30 | 0.76 |
| Max Contamination | ||||||||||||||||
| 2012 - 3 | 181.9 | 1.3527 | 5.3395 | 2.2088 | 126 | 32 | 77 | 21.14 | 83.43 | 34.51 | 0.07253 | 0.28629 | 0.12523 | 2.34 | 9.24 | 2.12 |
| 2013 - 4 | 345.7 | 2.5708 | 10.1477 | 4.1978 | 66 | 17 | 40 | 40.17 | 158.56 | 65.59 | 0.13784 | 0.54409 | 0.23799 | 4.45 | 17.55 | 4.03 |
| 2014 - 5 | 340.3 | 2.5306 | 9.9892 | 4.1322 | 67 | 17 | 41 | 39.54 | 156.08 | 64.57 | 0.13568 | 0.53559 | 0.23428 | 4.38 | 17.28 | 3.97 |
| 2015 - 6 | 450.3 | 3.3486 | 13.2182 | 5.4679 | 51 | 13 | 31 | 52.32 | 206.53 | 85.44 | 0.17954 | 0.70872 | 0.31000 | 5.79 | 22.86 | 5.25 |
| 2016 - 7 | 413.2 | 3.0727 | 12.1292 | 5.0174 | 55 | 14 | 34 | 48.01 | 189.52 | 78.40 | 0.16475 | 0.65033 | 0.28446 | 5.31 | 20.98 | 4.82 |
| 2017 - 8 | 422.6 | 3.1426 | 12.4051 | 5.18 | 54 | 14 | 33 | 49.10 | 193.83 | 80.18 | 0.16850 | 0.66512 | 0.29093 | 5.44 | 21.46 | 4.93 |
Abbreviations: MOE, margin of exposure; BMDL10, benchmark dose lower confidence limit 10%.
a Calculated margin of exposure (MOE) via lowest BMDL10 of AFB1 derived from the animal data (170 ng/kg-b.w/day) (17).
b Based on age-standardized incidence rate in Northern Europe for liver cancer 3.1/100000 population/year and 5.9/100000 population/year for Iranian people (29, 30).
c Consumption in total European population (0.52 g/person/day) (20).
d The consumed amount of the pistachio product in the population percentage-wise based on the estimated percentage of the population who consumed the products of pistachio (2.05 g/person/day) (20).
e Consumption in total Iranian population (0.82 g/person/day) (20, 21).
Table 3 presents HI values for the mean exposure to AFB1 for Iranian and European consumers which the highest values are 0.76 and 1.84, respectively. This figure is more than one only for European people who consumed the specific pistachio products, indicating health concerns. The MOE derived from dietary exposure was used for performing the risk assessment (Table 3). This indicator was between 1441 and 12843 for the whole consignment in all six years. MOE's level based on BMDL10 was less than 10000 for Iranian and European people who consumed the specific pistachio products, indicating a high health concern. It increased to more than 14478 after removing the contaminated consignment leading to cargo rejection using the monitoring system, resulting in no concern about pistachio consumption from the public health point of view. MOE measured according to the max and mean AF contamination in Rejected consignment was reported to be significantly less than 10000, indicating a high risk for human consumption. The HI values more than one also confirm this. Such cargos need serious measures to regain their safety criteria via sorting and other similar policies for aflatoxin contamination (4).
As shown in Table 3, the estimated liver cancer risk from dietary exposure to AFB1, based on the mean contamination, was 71 × 10-5 - 633 × 10-5. Based on the maximum contamination exposure, liver cancer risk ranged from 72 × 10-3 to 70 × 10- 2 cancers/100,000 population/year, contributing to 0.02 - 0.2% and 2.12 - 22.86% of the liver cancer incidence attributable to dietary AFB1. This range was reduced to below 0.02% after removing contaminated cargoes by the monitoring system.
4. Discussion
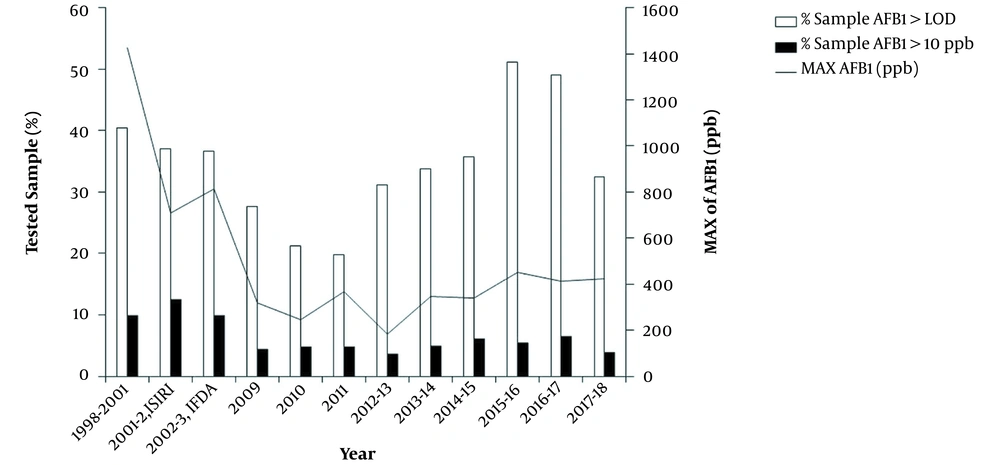
The susceptibility of pistachio nuts to AFs contamination has been identified (21, 31). Factors such as fruiting, harvesting, transport, and storage may impact pistachio contamination rate. Infection of pistachio with the fungus during ripening may result in its contamination with aflatoxin. It forms as pistachios undergo early split and mechanical damage caused by biotic and abiotic factors. Fungal contamination, aflatoxin production, and their incidence are mainly affected by temperature, relative humidity, and the moisture content of pistachio as it is produced and processed during stages. Most factors influencing the growth of Aspergillus and pistachio contamination with aflatoxin can be controlled by implementing good agricultural practice (GAP) in the orchards and hazard analysis and critical control point principles in processing plants and post-harvesting stages (32). However, some climatic factors such as temperature, humidity, and the extent of rainfall during cultivation, especially in a period of ripening and harvest from August through November, are not controllable (33, 34). Despite controlling factors affecting aflatoxin production, fluctuations in its contamination level have been observed due to climate change and its differences in various geographical areas (35-37). Therefore, monitoring mycotoxins has been conducted since 1999 to minimize aflatoxin contamination in pistachio throughout the country, implemented by the Iranian industrial research and standard institute (ISIRI) and Iranian food drug administration (IFDA). As shown in Figure 1, the contamination rate caused by AFB1 and its maximum level observed in cargoes has reduced since 1999.According to the previous study, the mean contamination level of AFB1 decreased from 5.7 ng/g to 1.99 ng/g during 2002 - 2011 (7, 13). The results also revealed that the declining contamination trend continued up to 2012 - 2014.Overall, the mean contamination experienced a fluctuation in 6 years, beginning from Nov. 2012 ending in Oct. 2018. From Nov. 2012 to Oct. 2018, approximately 94% of examined samples had AFB1 contamination rate within the acceptable E.U. and Iran limit with an average of 1.78 - 4.02ng/g.
Compared with the similar research done by Yazdanpanah (38), these findings suggest a lower level of contamination. It should be noted that the mean level of contamination during Nov. 2013 - Oct. 2017, based on the current study, was more significant than the level suggested by Dini et al. in the years 2009 - 2011 (13). Despite increasing pistachio contamination levels from Nov. 2013 to Oct. 2017, there was a decreasing trend of contamination in exported pistachio cargoes in the Nov. 2017- Oct. 2018
In the previous study, the TDI of AFB1 was calculated at 0.11 - 0.19 ng/kg.bw/day by Kuiper-Goodman (39). The dietary exposure based on the mean contamination levels of AFB1 for pistachio nuts in all consignment ranged from 0.013 to 0.118 ng/kg.bw/day, none of which exceeded TDI. This criterion was higher (2 - 12 times) than TDI for all groups and years in rejected consignment using a monitoring system, which perfectly shows the importance of sorting and removing contaminated pistachio cargos.
The calculated values for MOE under 10,000 indicate that the level of AFB1 exposure concerns from a public health perspective. It could be considered a reasonable priority for risk management measures to reduce exposure in humans. The results obtained from the risk assessment of all consignments exporting to the E.U. indicated that the MOE value was below 10000 in those European and Iranian consuming pistachio products in years with a mean contamination level of more than 2.25 ng/g. As a result of the monitoring system, it reached above 14478 in unrejected or authorized consignments and remained within the hazardous range for rejected ones. This indicates that the maximum level for AFB1 set by the E.U. commission is proper, leading to lower public health concerns (40).
Despite the high level of contamination in rejected cargoes and low MOE value, cancer incidence attributable to pistachio consumption was in the range of 0.48 - 3.43% due to pistachios' low per capita consumption. Thus, the estimated liver cancer risk from consumption of pistachio nuts was reduced by nearly 10 times using the monitoring system. The results suggest that consumption of authorized pistachio consignment poses no noticeable health risk of mycotoxins residues for European and Iranian consumers. The findings were in line with the research done by Taghizade et al. on different varieties of Iranian pistachio. They showed that Hazard Index is lower than 1 for Iranian consumers (21). Although Cancer incidence associated with pistachio consumption, based on mean contamination in rejected cargoes, can enhance by 3.43%, and it may reach 22.86% if the consignments with a maximum level of aflatoxin contamination are consumed. Therefore a monitoring system to reduce AFs to an acceptable limit is necessary.
Since HI is more than one in rejected consignments, and the MOE is less than 10,000, so the consumption of these shipments endangers consumers' health. Therefore, eliminating these cargoes or reducing the amount of aflatoxin in them is necessary to minimize health risks and the financial burden on health care. A lot of researchers have tried to find ways to reduce AF in contaminated crops. Recently these AF management strategies have been reviewed by Yazdanpanah and Eslamizad (4), Stoev 41), and Gnonlonfin et al. (42). Although several physical and chemical techniques have been proposed to lower AF, most are not considered safe or suitable for human consumption. Among such techniques, physical methods, including mechanical and electronic sorting and roasting, can be applied. Moreover, chemical treatments whit ozone, H2O2, lemon extract, citric acid, the salt solution can also be used for AF detoxification in pistachio (24, 41, 43).
4.1. Conclusions
Incidence of AFs level showed that the mean contamination of AFB1 and AFT was below the maximum limit recommended by the European Commission regulations. Also, aflatoxin contamination levels in 95% of analyzed samples comply with the legislated limit. The risk assessment results showed that the MOE value for those Europeans who consume the specific pistachio products would be of concern from the public health point of view. In the light of the monitoring system, rejection of aflatoxin-contaminated consignments increased the MOE and HI value to more than 10000 and less than 1, respectively, resulting in lower population risk in primary liver cancer caused by AFB1 contamination. Risk assessment in rejected consignments showed that the consumption of these shipments endangers consumers' health. Therefore, eliminating these cargoes or reducing the amount of aflatoxin in them is necessary to minimize health risks and the financial burden on health care. The monitoring system's performance indicates that the consumption of pistachio nuts poses no health risk for European and Iranian consumers.