Introduction
Experimental evidence indicates a critical role of cyclic guanosine monophosphate (cGMP) in antinociception. This evidence is in line with the observation that local administration of L-arginine produces antinociception in rats with carrageenin-induced hyperalgesia, the effect being blocked by nitric oxide (NO) inhibitors and methylene blue (soluble guanyly cyclase inhibitor) (1). Moreover, the possibility of such a role is supported by the fact that intraplantar injection of dibutyryl-cGMP causes antinociception (2). Therefore, cGMP seems to be very important for the functional up-or down-regulation of the nociception.
Intracellular cGMP concentrations are regulated by the action of guanylyl cyclases and the degradation rate induced by cGMP-specific phosphodiesterases (PDEs) (3). It has been reported that PDE-5 seems to be the most relevant enzyme for the cGMP inactivation in cells (4). Vardenafil is a selective and potent inhibitor of cGMP-specific PDE-5 which catalyzes the hydrolyzis of cGMP. Although, previous studies have consistently found that sildenafil, another cGMP-specific PDE-5 inhibitor, produces antinociception in the formalin test in rats, to date the possible role of vardenafil in the nociception has not been defined (5, 6).
On the other hand, Scott et al. have recently shown that A-1048400, a novel, orally active, neuronal calcium channel blocker produced dose-dependent antinociception thus, establishing a link between Ca+2 channels and antinociception (7). The role of this pathway in possible vardenafil-induced antinociception has not been studied extensively and requires further clarification.
In this study, we aimed to investigate the antinociception effects of specific PDE-5 inhibitor vardenafil on carrageenan-induced nociception in rats, and to investigate the possible role of NO/cGMP/Ca+2 channels pathway in this effect.
Experimental
Animals
In this study 64 adult male Wistar-Albino rats, weighing between 200 and 280 g, were used. The care of animals and realization of approaches and procedures within the study framework were realized in strict compliance with the principles stated by The National Institutes of Health in their Guide for Care and Use of Laboratory Animals. This study was performed in the Experimental Medical Research Center of Akdeniz University Medical Faculty, and approved and guidance of Ethics Committee on Animal Research of Akdeniz University Medical Faculty. During the study period, rats were fed a standard laboratory diet, allowed water ad libitum and housed in a light-controlled room with a 12 h light/dark cycle. Animal groups are as follows:
1) Control group (saline injected), 2) Carrageenan group, 3) Vardenafil group, 4) Vardenafil + carrageenan group, 5) Vardenafil + L-NAME group, 6) Vardenafil + ODQ group, 7)Vardenafil + L-arginine, 8) Vardenafil + A23187 group.
Induction and assessment of carrageenan-induced hyperalgesia
Carrageenan is an irritant that induces an acute inflammatory response on injection into tissues (8). It is injected into the footpad of rodents and known to cause physiological and biochemical changes over a short time course and limited to the effected limb. This model results in oedema, pain and hyperalgesia of the injected footpad and limb (9).
In our study, hyperalgesia was induced in the right hind paw of rats by injecting 0.1 ml of a freshly prepared solution of 2% carrageenan. The left paw received 0.1 ml of saline which served as a control. The extent of hyperalgesia was determined by measuring the mechanical nociceptive pressure threshold by the paw pressure test via a commercially available analgesiometer (UGO Basile, Model No: 37215, Italy), which is based on the determination of the animal threshold response to pain that is induced to the paw by the application of a uniformly increasing pressure (10). Briefly, the rats were maintained in a normal/horizontal position in the hand of the researcher. The right hind paw was placed in an analgesiometer, which was composed of a cone-shaped paw-presser with a rounded tip that was used to apply linear increasing force to test paw. The weight in grams that was required to elicit a nociceptive response, such as paw flexion, represented the nociceptive threshold. A maximum pressure force of 400 g was used to prevent damage to the paws. The apparatus was set up to apply a force of 0-400 g, increasing from zero. The nociceptive threshold was taken as the point at which the rat vocalized or struggled vigorously and determined by the average of three consecutive pressure tests recorded before injection of carrageenan (zero time, 100%) and 2 h after the injection of carrageenan, which represented the peak of effect. Vardenafil was injected intraplantarly in the right paw 30 min before the carrageenan challenge and the pressure threshold was observed at 15, 30, 60 and 120 min after carrageenan administration.
The extent of hyperalgesia was calculated as the difference between the pretreatment and the peak of effect pressure test averages (Δ of nociceptive threshold) and expressed as %. To reduce stress, the rats were habituauted to the pressure test apparatus one day prior to the experiments.
Drugs
Vardenafil, NW-nitro-L-arginine methyl ester (L-NAME), 1H-(1, 2, 4) oxadiazolo (4, 3, a) quinoxalin-1-one (ODQ), L-arginine, A23187 (calcium ionophore) carrageenan and dimethyl-sulfoxide (DMSO) were obtained from Sigma (Sigma Chemical Co., St.Louis, Mo., USA). All test drugs were administered by intraplantar (i.pl). injection. Vardenafil, ODQ and A23187 were dissolved in DMSO whereas carrageenan, L-NAME and L-arginine were dissolved in saline. All solvents were tested alone and showed as ineffective in carrageenan-induced nociception test (DMSO at a final concentration of 0.2% (v/v) did not effect carrageenan-induced nociception test).
Experimental Design
Determination of antinociceptive effect of vardenafil in carrageenan-induced nociception test:
Rats were treated with vehicle (DMSO) or vardenafil (50, 100 µg/kg, i.pl) and 30 min later, carrageenan was injected. Antinociception response was determined before (0 min) and after (15, 30, 60 and 120 min) carrageenan administration with analgesiometer. Same protocol was applied to left paw with DMSO (vehicle of vardenafil) as control.
Effect of pretreatment with L-NAME, ODQ or L-arginine on antinociceptive activity of vardenafil in carrageenan-induced nociception test:
To examine the effect of L-NAME (nitric oxide synthase inhibitor), ODQ (a specific inhibitor of guanylyl cyclase) or L-arginine (nitric oxide donor) on vardenafil activity, animals were pretreated with L-arginin (10 µg/kg, i.pl), L-NAME (100 µg/kg, i.pl) or ODQ (50 µg/kg, i.pl) 10 min before vardenafil (100 µg/kg, i.pl) injection. Carrageenan was administered 30 min after vardenafil injection. Antinociception response was determined before (0 min) and after (15, 30, 60 and 120 min) carrageenan administration with analgesiometer. Same protocol was applied to left paw with DMSO (vehicle of vardenafil and ODQ) or saline (vehicle of L-NAME and L-arginin) as control.
Role of A23187 (calcium ionophore) on vardenafil-induced antinociception
Vardenafil (100 µg/kg, i.pl) was injected to right paw 10 min after A23187 and 30 min before carrageenan administration in order to evaluate the Ca+2-related dynamics in vardenafil’s antinociceptive effect. Antinociception response was determined before (0 min) and after (15, 30, 60 and 120 min) carrageenan administration with analgesiometer. Same protocol was applied to left paw with DMSO (vehicle of A23187) as control.
Data analysis and statistics
All results are presented as means ± S.E.M. for ten animals per group. The significance of the differences in the responses of treatment groups in comparison to the control was determined by Mann-Whitney U test. P < 0.05 was considered statistically significant.
Results and Discussion
Carrageenan response
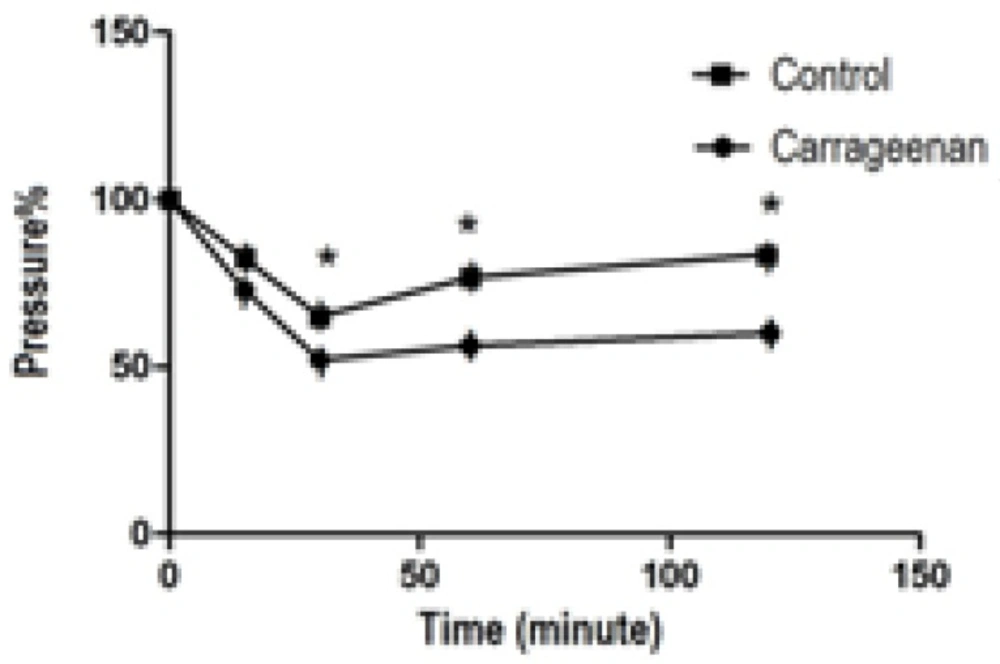
İntraplantar injection of 0.1 mL carrageenan into the right paw of the rats produced a significant decrease in nociception pressure at 15, 30, 60 and 120 min compared to control group (p < 0.05) (0.1 mL saline injected group, Figure 1).
Antinociceptive action of vardenafil
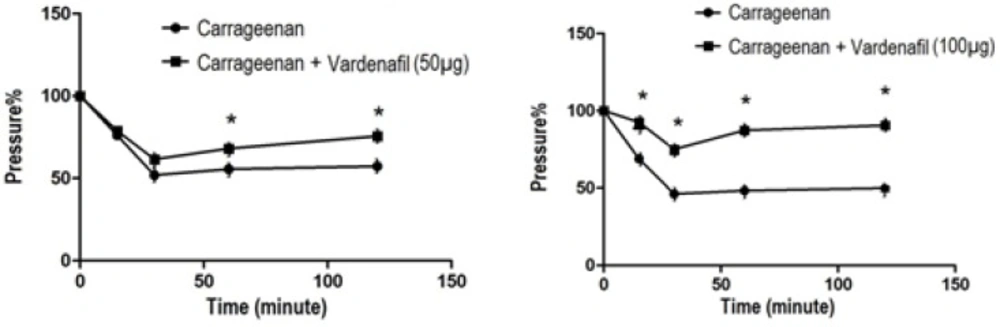
İntraplantar injection of 0.1 mL vardenafil (50, 100 µg/kg,0.1mL, i.pl) produced a significant increase in nociception threshold that had been decreased by carrageenan. This effect is more pronounced at 30, 60 and 120 min in 50 µg/kg vardenafil-injected group, whereas it is significant at all measurement times in 100 µg/kg vardenafil-injected group (p < 0.05) (Figure 2a-b).
Effect of, L-NAME, ODQ, L-arginine and A23187 on the antinociception produced by vardenafil
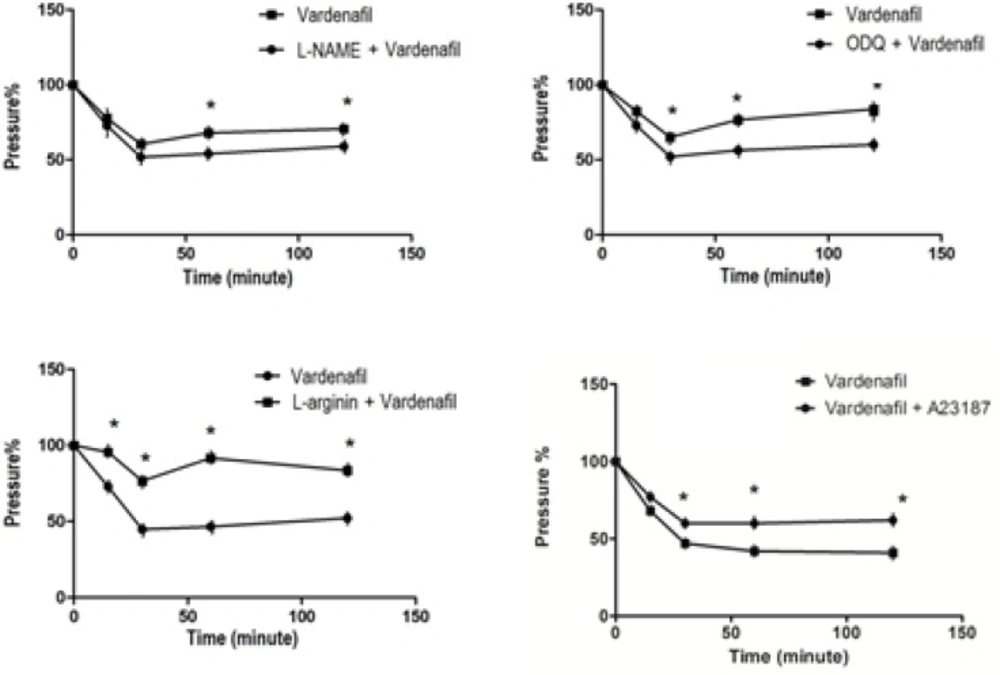
The inhibitor of nitric oxide synthase L-NAME (100 µg/kg, i.pl) and the inhibitor of guanylyl cyclase ODQ (50 µg/kg, i.pl), significantly inhibited the vardenafil-induced antinociception whereas, L-arginine (nitric oxide donor, 10 µg/kg, i.pl) significantly (p < 0.05) enhanced the antinociceptive effect of vardenafil. Moreover, A23187 (calcium ionophore) (1 µg/kg, i.pl) injection 10 min before vardenafil (100 µg/kg, i.pl) administration caused a significant decrease (p < 0.05) in antinociception at 30, 60 and 120 min (Figure 3a-d).
As previously reported for sildenafil, we have found that intraplantar administration of vardenafil is also able to produce antinociception in carragenan-induced hyperalgesia test (11). This is the first data about this topic. Based on the fact that vardenafil is a potent, selective, and reversible phosphodiesterase type-5 inhibitor that blocks cGMP hydrolysis, we suggest that vardenafil could produce its antinociceptive effect through the activation of NO-cGMP transduction pathway. Becuse, this suggestion is in line with evidence that showing that this pathway plays an important role in sildenafil-induced analgesia (5, 12, 13).
Recent studies have suggested that L-arginine/NO-cGMP pathway is involved in the modulation of pain perception (14, 15). Nitric oxide and cGMP appears to involve in certain drugs –induced antinociception mechanisms (16, 17). The NO-cGMP pathway depends on the synthesis and release of NO triggered by the activation of nitric oxide synthase (NOS), which then activates the guanylate cyclase enzyme, guanylyl synthase, which is directly responsible for an increase in the intracellular level of the most important messenger of the system, cGMP (18). It has been reported that cGMP plays an important role in the up-or down-regulation of nociceptors and is a key mediator of antinociceptive activity (19). Intracellular concentration of cGMP is regulated with the balance of phosphodiesterase and guanylate cyclase activity. Phosphodiesterase type-5 is the predominant enzyme for hydrolysis of cGMP (3).
In order to examine antinociceptive effect of vardenafil and to explain the participation of the NO-cGMP pathway, carrageenan-induced hyperalgesia model was used. As shown in Figure 1, i.pl. injection of carregeenan (0 1mL) into the right paw of rats evoked a significant hypersensivity when compared with saline group. Carrageenan-induced hyperalgesia model is an example of acute and local inflammation. In this model, physiological and biochemical changes occur over a short time course and limited to the only effected limb. We administered all drugs also, carrageenan, via intraplantar injection (local administration route). So, we suppose to avoid the effects of a systemic inflammation. On the other hand, it has been recently demonstrated that sildenafil has a partial anti-inflammatory effect through iNOS inhibition (20). Also, sildenafil has been known to reduce the expression of proinflammatory cytokines and cyclooxygenase-2 (COX-2). Moreover, in-vitro treatment with tadalafil or vardenafil in human myofibroblast prostatic cells, reduced IL-8 secretion and blunt inflammatory response, induced by inflammatory stimuli, likely via activation of cGMP/PKG signaling (21). But, investigators declared the necessity of further studies to explain the molecular mechanisms of this protection in nervous system.
The results presented here show that vardenafil exhibits antinociceptive activity in dose-dependent manner. This study presents for the first time the interesting fact that vardenafil-induced antinociception is significantly blocked by L-NAME, a NOS inhibitor and ODQ, a guanylyl cyclase inhibitor while, L-arginine (nitric oxide donor, 10 µg/kg, i.pl) enhanced the antinociceptive effect of vardenafil. Together these data suggest major participation of the NO-cGMP pathway in the antinociception produced by vardenafil. We assume that the inhibition of PDE type-5 and therefore the accumulation of cGMP is sufficient to produce antinociception.
There is now a substansial body of evidence indicating the relationship between Ca+2 channels and analgesia (22, 23). It is well known that Ca+2 channels play essential roles in setting the resting membrane potential as well as controlling the excitability of neurons (24). The opening of Ca+2channels leads to depolarization of cell membranes, and that increase cell excitability. The results demonstrated in our study show that the pre-treatment of rat wit a calcium ionophore, A23187 (1 µg/kg, i.pl) significantly decreased the antinociception caused by vardenafil. Such results support that the antinociceptive effect caused by vardenafil in rats seems to involve Ca+2-related dynamics.
Since vardenafil is well documented in the treatment of pulmonary hypertension and erectile dysfunction, our study offers a new approach for its potential therapeutic utility in analgesia. To our knowledge, this is the first one to directly demonstrate that vardenafil exhibit antinociception and this effect thought to be mediated primarily through NO-cGMP signaling pathway and additional Ca+2-related mechanisms.