Voltammetric studies
Cyclic voltammogram of 1.0 mM of acetaminophen (
1) in aqueous solution containing 0.2 M phosphate buffer at pH 7 shows one anodic (A
1) and corresponding cathodic peak (C
1), corresponding to the transformation of acetaminophen to
N-acetyl-
p-benzoquinone-imine (NAPQI) and
vice versa within a quasi-reversible tow-electron process (
Figure 2, curve a). (
13-
15) A peak current ratio
of nearly unity particularly during the recycling of potential can be considered as criteria for the stability of
NAPQI produced at the surface of electrode under the experimental conditions (
14).
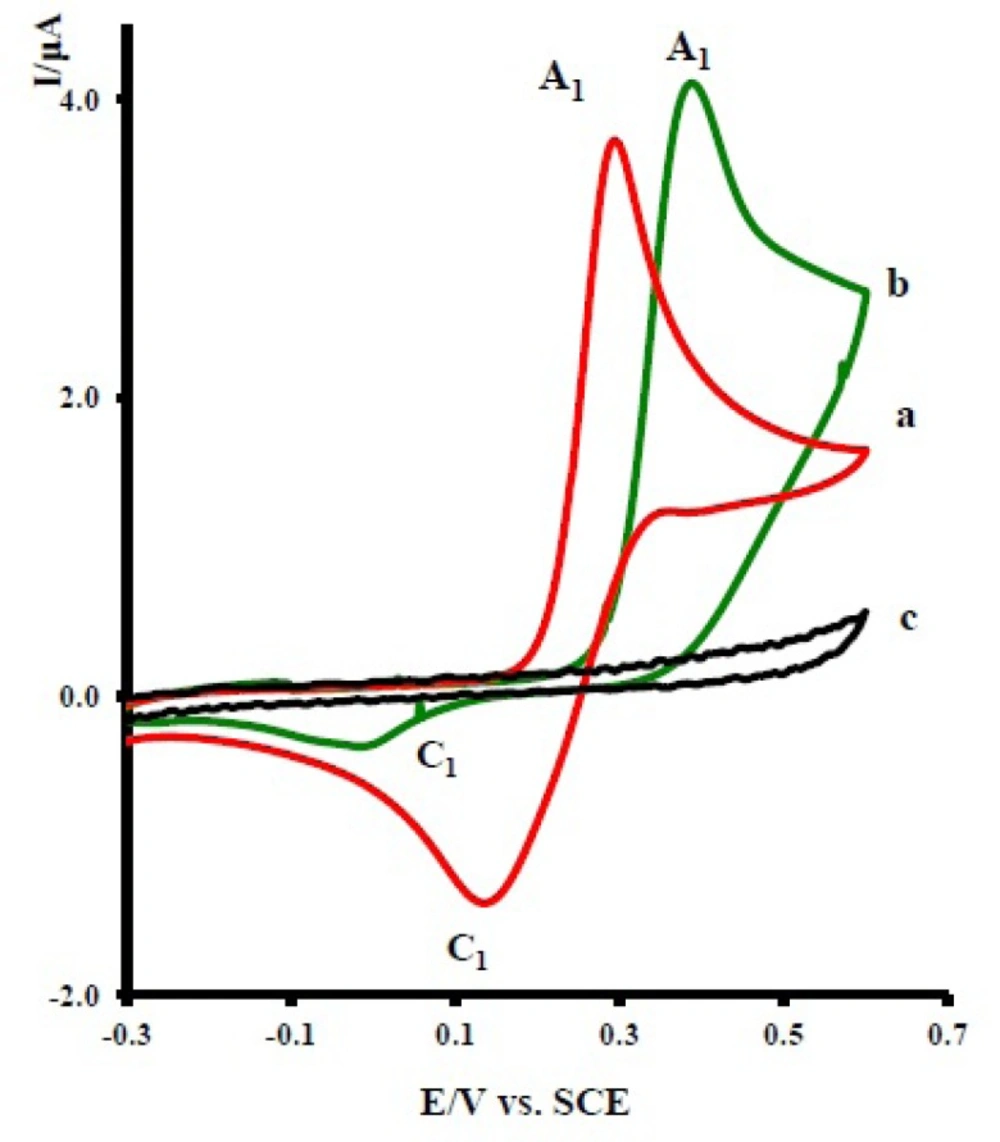
Cyclic voltammograms of 1.0 mM acetaminophen (1): (a) in the absence, (b) in the presence of 10.0 mM nortriptyline (2x), and (c) 10.0 mM nortriptyline (2x) in the absence of acetaminophen at a glassy carbon electrode in water/acetonitrile (70/30) solution containing 0.2 M phosphate buffer (pH = 7.0). Scan rate: 10 mVs-1, T = 25 ± 1ºC
The oxidation of 1 in the presence of 10 mM of nortriptyline (2x) was studied in some detail.
Figure 2 curve b, shows the cyclic voltammogram of 1 in the presence of 2x. The voltammogram exhibits one anodic peak (A
1) and its cathodic counterpart (C
1). The comparison of peak C
1 in the absence and presence of 2x shows a decrease in the current for the later. The observed shift of the A
1 peak in curve b, relative to curve a, is probably due to the formation of a thin film of product at the surface of the electrode, inhibiting to a certain extent the performance of the electrode process. In this figure, curve c is the voltammogram of 2x in the same condition and in the absence of 1.
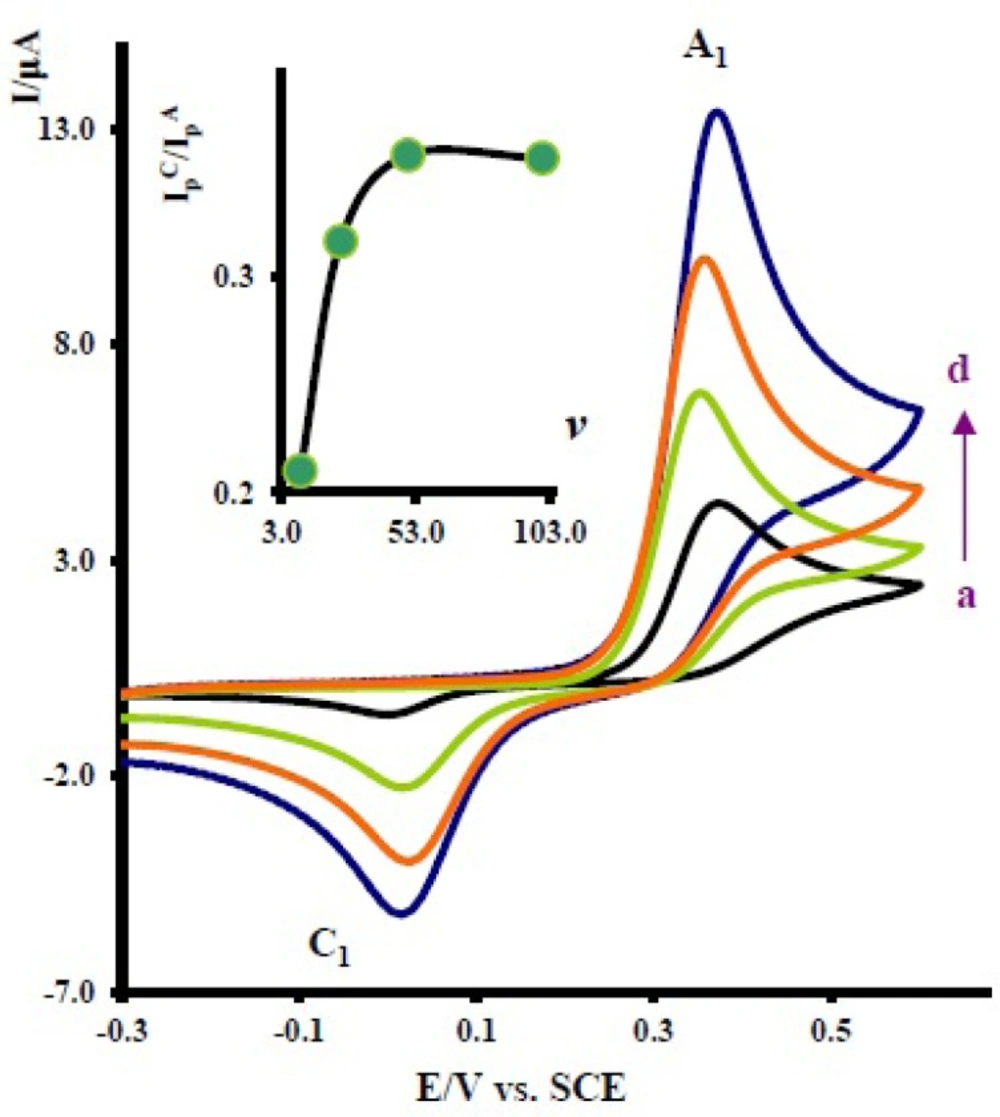
Cyclic voltammograms of 1 mM acetaminophen in the presence of 10.0 mM nortriptyline at a glassy carbon electrode in water/acetonitrile (70/30) solution containing 0.2 M phosphate buffer (pH = 7.0) at various scan rates. Scan rates from a to d are: 10, 25, 50 and 100 mVs-1. Inset: variation of peak current ratio vs scan rate
The effect of scan rate on the electrochemical behavior of 1 in the presence of 2x was also studied.
Figure 3 shows the typical cyclic voltammograms obtained for 1 mM of 1 in the presence of 10 mM nortriptyline (2x) at various potential scan rates. As can be seen, with increasing scan rate, the peak current ratio,
, increases (
Figure 3 Inset). To gain further information, the electrochemical oxidation of acetaminophen (
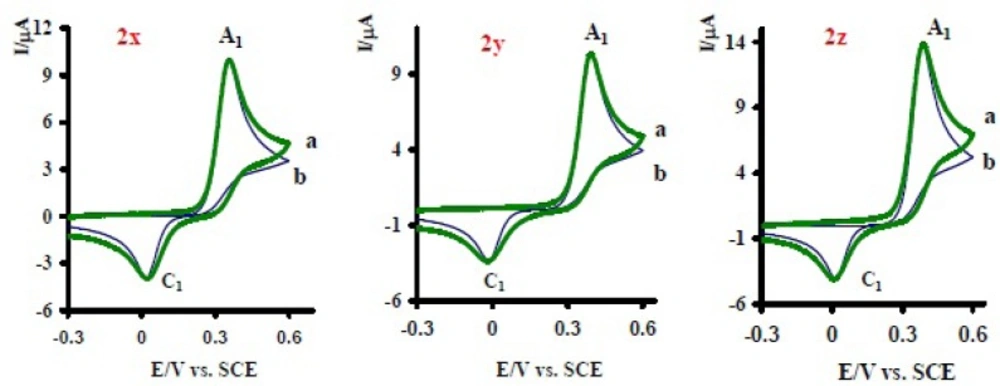
1) was studied in various concentrations of 2x.
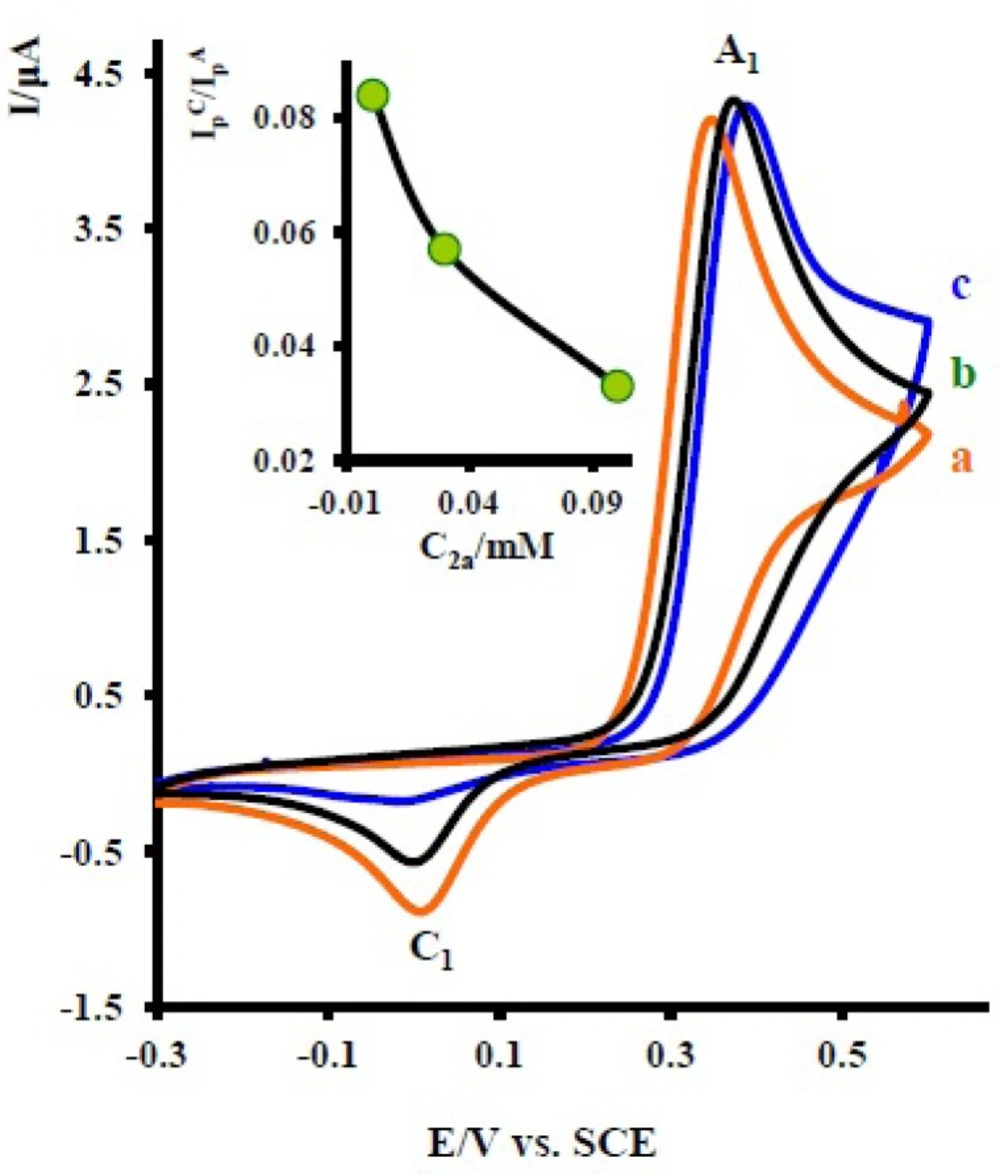
Figure 4 shows the cyclic voltammograms of 1 at various concentrations of 2x. As can be seen the peak current ratio
decrease with increasing of the concentration of 2x. This implies that the chemical reaction (DDI) occurred after the electron transfer process (
19).
Cyclic voltammograms of 1 mM acetaminophen (1) in the presence various concentration of nortriptyline (2x) at a glassy carbon electrode in water/acetonitrile (70/30) solution containing 0.2 M phosphate buffer (pH = 7.0) at 10 mVs-1 scan rate. Concentrations from a to c are: 0.1, 0.01 and 0.03 mM. Inset: variation of peak current ratio vs concentration ratio (nortriptyline (2x) / acetaminophen (1)). T = 25 ± 1 ºC.
The existence of a subsequent chemical reaction between NAPQI and 2x is supported by the following evidences: (a) decrease in
during the reverse scan, which could be indicative of the fact that electrochemically generated NAPQI is removed by chemical reaction with 2x. (b) Dependency of the peak current ratio
on the potential sweep rate (
Figure 3). In this case, for the highest sweep rate employed, a well-defined cathodic peak C
1 is observed. For lower sweep rates,
become less than one and increases with increasing scan rate (
Figure 3, inset). This is indicative of departure from the intermediate and arrival to the diffusion region with increasing sweep rate (
19). (c) Dependency of
on the concentration of 2x. This is related to the increase of the homogeneous reaction rate of the following chemical reaction. (d) Increasing of
in the presence of 2x (
Figure 2 curve b). This can be related to the increase of apparent number of electrons from two to six (see coulometry section).
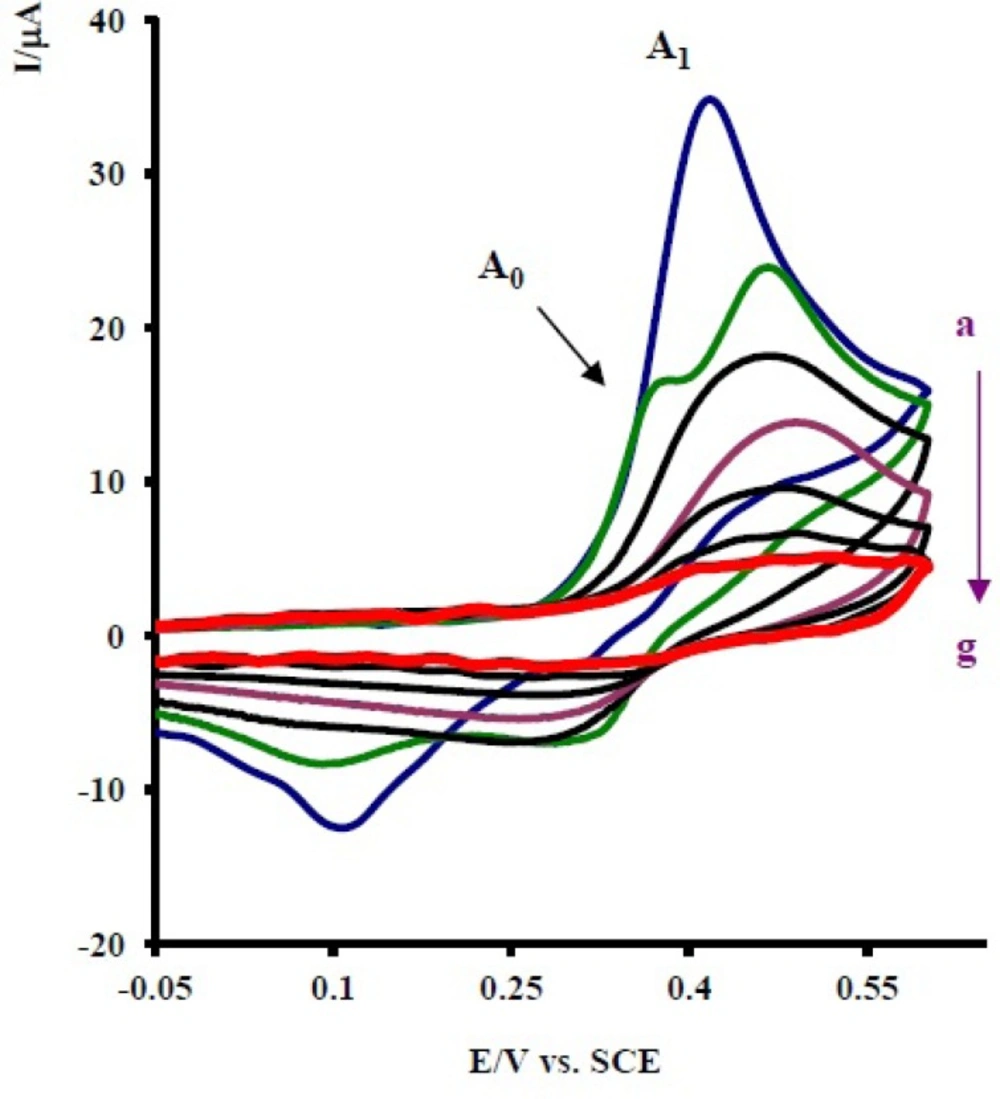
Controlled-potential coulometry was performed in aqueous solution containing 0.1 mmol of 1 and 0.1 mmol of 2x at 0.30 V
versus saturated calomel electrode (SCE). The electrolysis progress was monitored using cyclic voltammetry (
Figure 5). It is shown that, proportional to the advancement of coulometry, the anodic peak A
1 decreases and a new anodic peak (A
0) appears. The anodic peak A
0 is probably related to the oxidation of an intermediate that is produced in the electrolysis cell (7a or 9a in
Scheme 1). Anodic and cathodic peakas (A
1/C
1 and A
0/C
0) disappear when the charge consumption becomes about 6e
- per molecule of 1.
Cycilc voltammograms of 0.1 mmol acetaminophen (1) in the presence of 0.1 mmol nortriptyline (2x), in 0.2 M phosphate buffer, pH 7, during controlled potential coulometry at 0.30 V vs. SCE, after consumption of: (a) 0, (b) 10, (c) 20, (d) 30, (e) 40, (f) 50 and (g) 55 C. Scan rate: 100 mV s -1. Inset: variation of peak current vs charge consumed. T = 25 ± 1 °C
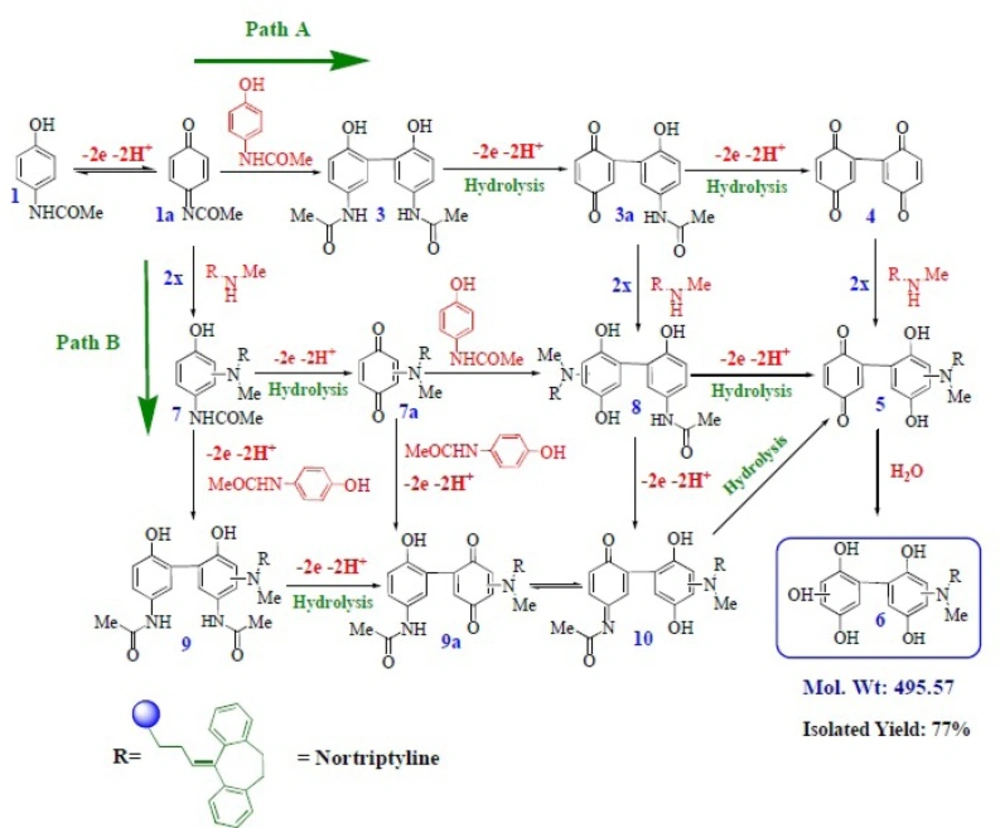
Electrochemical and spectroscopic data in accompanied by previously published data (
12-
15) allow us to propose the mechanism presented in
Scheme 1 for the electrooxidation of acetaminophen (
1) in the presence of 2x-z. It seems that because of the good reactivity (decrease in
during the reverse scan) between NAPQI and 2x at the surface electrode in time window of cyclic voltammetry (10
-7-1 s), so we can expect that Michael addition reaction takes place prior to the hydrolysis reaction in controlled-potential coulometry (a method with time windows, 100–3000 s (
19)) according to the path B (
Scheme 1) leading to the product 6. Since the oxidation of 6 occurs at more positive potentials, the overoxidation of 6 was circumvented during the controlled potential preparative reaction. The electroorganic synthesis of 6 has been performed using oxidation of 1 in the presence of 2x as described in the experimental section. According to our results, the anodic peak of the voltammograms presented in
Figure 2 (A
1) pertains to the oxidation of acetaminophen (1) to the
N-acetyl-
p-benzoquinone-imine (NAPQI) 1a. Obviously, the cathodic peak C
1 is corresponding to the reduction of
N-acetyl-
p-benzoquinone-imine (NAPQI) 1a.
Proposed mechanisms for the electrochemical oxidation of acetaminophen (1) in the presence of 2x-z
The proposed mechanism was established by using Ms and IR technique. In the mass spectrum of compound 6, molecular ion [M + 1H] was recorded. This mass is related to protonation of the nortriptyline (2x) group (
20-
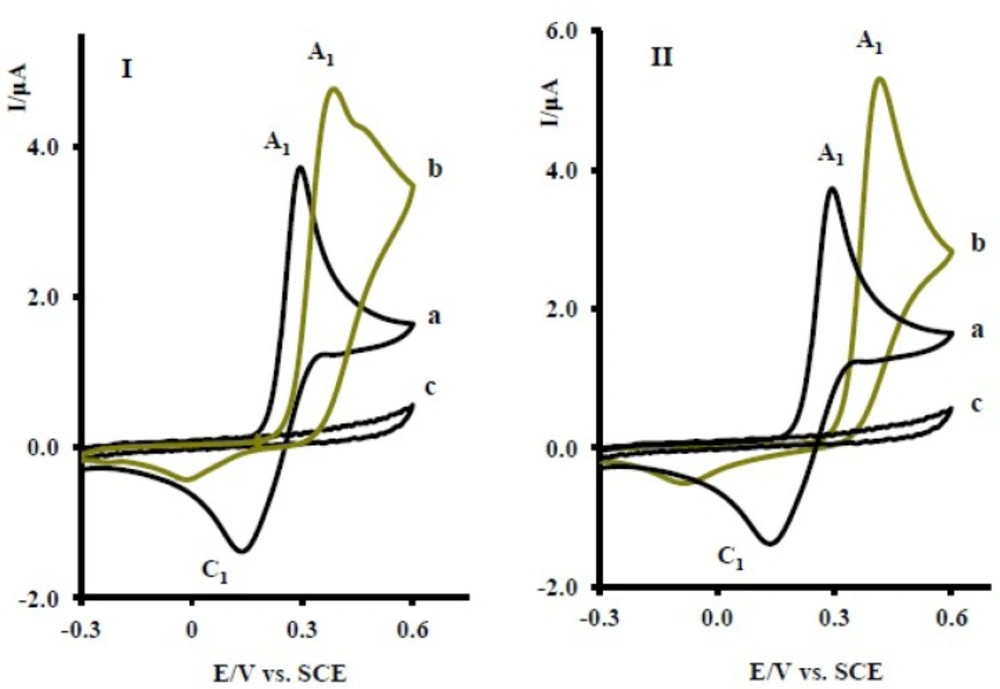
22) and is another proof for production of 6 in electrooxidation of 1 in the presence of 2x. The electrochemical oxidation of acetaminophen (1) in the presence of sertraline (2y) and fluoxetine (2z) was also investigated (
Figure 6). The same behavior has been seen for the electrochemical oxidation of 1 in the presence of 2y, z.
(I) Cyclic voltammograms of 1 mM acetaminophen (1) in the absence (a), presence (b) of 10 mM fluxetin (2z) and (c) 10 mM fluxetin (2z) (II) Cyclic voltammograms of 1 mM acetaminophen (1) in the absence (a), presence (b) of 10 mM serteralin (2y) and (c) 10 mM serteralin (2y) at the glassy carbon electrode in 0.2 M phosphate buffer, pH 7 at scan rate of 10 mVs-1, T = 25 ± 1ºC
Simulation
Electrochemical oxidation of acetaminophen (1) in various pHs was tested by digital simulation. The simulation was carried out assuming semi-infinite one-dimensional diffusion and planar electrode geometry (
14). The experimental parameters entered for digital simulation consisted of the following:
Estart,
Eswitch,
Eend,
t=25
◦C and analytical concentration of acetaminophen (1). The transfer coefficient (
α) was assumed to be 0.5 and the formal potentials were obtained experimentally as the mid point potential between the anodic and cathodic peaks (
Emid). The heterogeneous rate constant (0.002cms
−1) for oxidation of acetaminophen (1) was estimated by use of an experimental working curve (
14,
23). All these parameters were kept constant through out the fitting of the digitally simulated voltammogram to the experimental data. The parameter
was allowed to change through the fitting processes. It should be noted that since in the absence of 2x-z, the peak current ratio
of 1 because of the participation of NAPQI (1a) in side reaction(s), is less than unity, the homogeneous rate constant of side reactions must be calculated, in the absence of nucleophile (2x-z), firstly, then
kobs for the reaction of 1a with 2x-z estimated in the next step according to simplified
Scheme 1.
Table 1 shows the
as a function of pH. As shown in
Table 1,
is strongly dependent to solution’s pH and type of antidepressant and increase with increasing of pH. The observed trend is expected, because of the more deprotonation of donating atom(s) in the structures of 2x-z and more activation of them towards Michael addition reactions with NAPQI (24). Moreover
for these drugs varied in the order of
>
>
. This can be due to the absence of any electron withdrawing group (EWG) (Cl, CF
3) in the structure of 2x (
Figure 1) (
13).
Cyclic voltammogrms of 1mM acetaminophen (1) and 10mM antidepressant drugs: nortriptyline (2x), serteralin (2y) and fluxetine (2z) at pH= 7.4, (a) experimental (b) simulated. Scan rate 70 mVs-1
| pH | kobsa of 1a with 2x | kobsa of 1a with 2y | kobsa of 1a with 2z |
|---|
| 2.9 | 7.61±64 | 5.45±0.16 | 1.35±0.07 |
| 4 | 9.40±1.02 | 7.1±0.44 | 1.95±0.06 |
| 4.8 | 11.43±0.97 | 8.66±1.52 | 3.84±0.22 |
| 7.5 | 14.00±0.84 | 12.13±1.53 | 7.04±0.25 |
| 8.7 | 20.32±2.18 | 18.00±1.73 | 11.03±1.65 |
| 9.7 | 44.00±3.87 | 33.5±3.53 | 21.53±3.13 |
Each second-order rate coefficient is the average of five determinations and is reported together with the standard deviation.